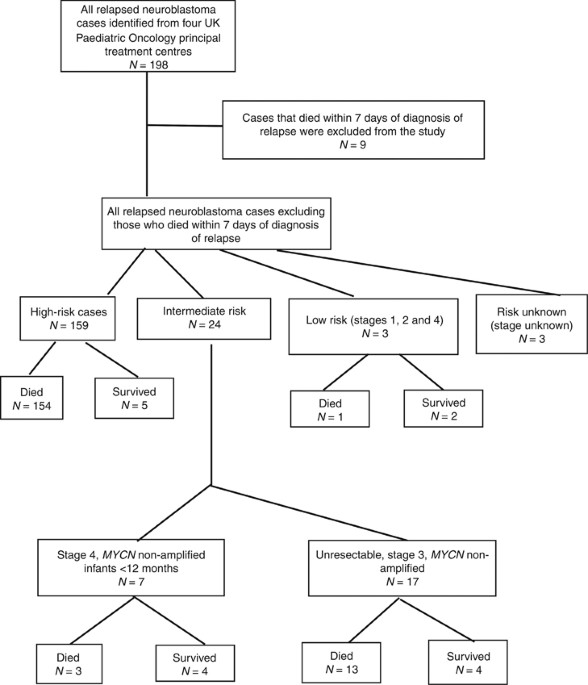
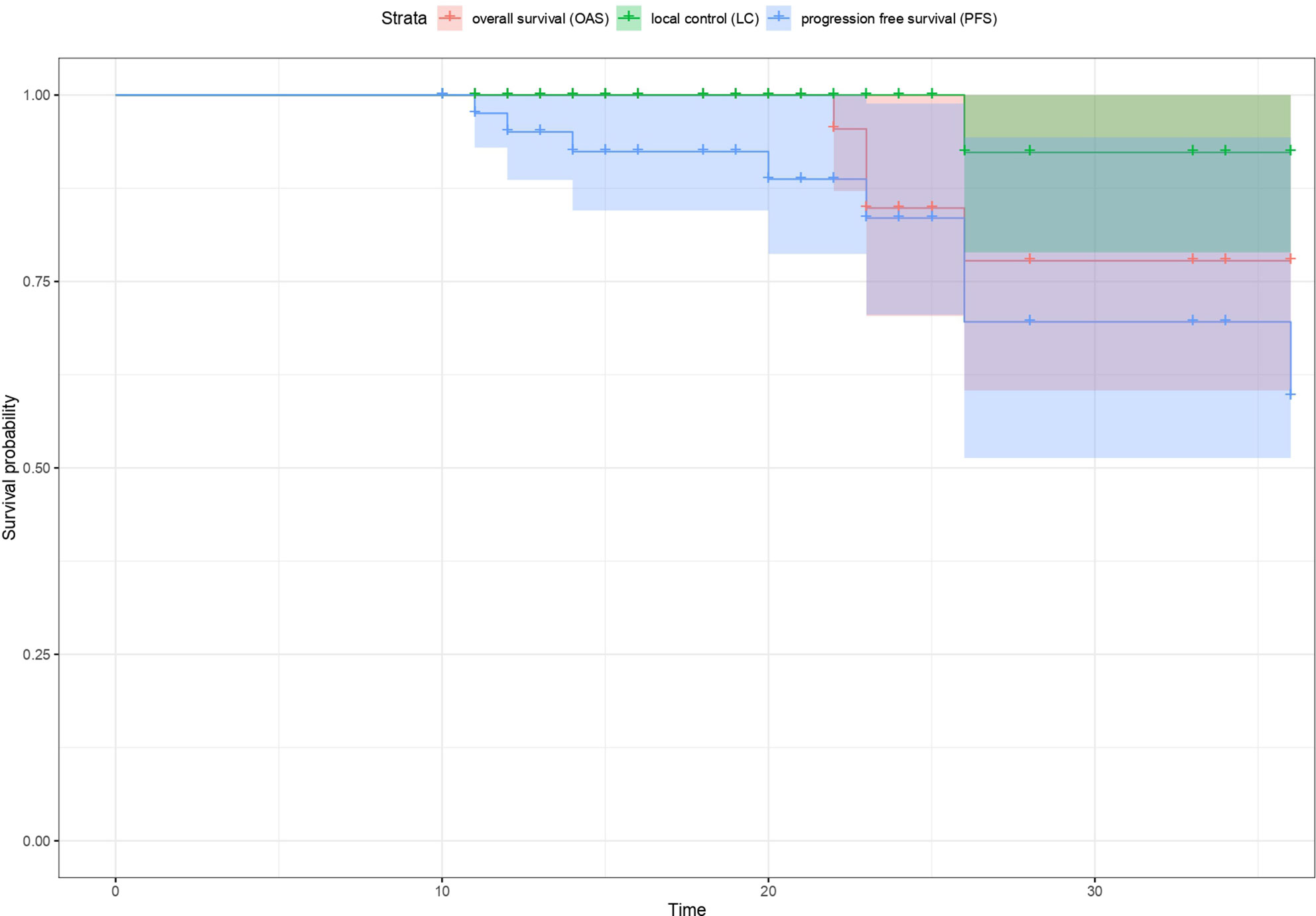
high risk neuroblastoma study 1 of siop-europe
|
HIGH-RISK NEUROBLASTOMA STANDARD CLINICAL PRACTICE
conducted by the European Neuroblastoma Study Group (ENSG) between 1990 and 1999 (ENSG5) investigated the effect of dose intensity of induction therapy on EFS in patients over the age of 1 year with metastatic disease |
Should induction therapy be randomized in high-risk neuroblastoma?
To our knowledge, this is only the fourth randomized study of induction therapy in high-risk neuroblastoma.3,12,31 Randomized comparisons have many advantages as they mitigate many of the differences in supportive care and assessment that change over time. When designing the trial, one option was to compare the current Children s
What percentage of neuroblastoma patients have high-risk disease?
Patients with high-risk disease account for approximately half of all patients with newly diagnosed neuroblastoma.
How is high-risk neuroblastoma treated?
Kletzel M, Katzenstein HM, Haut PR, et al. Treatment of high-risk neuroblastoma with triple-tandem high-dose therapy and stem-cell rescue: results of the Chicago Pilot II Study. J Clin Oncol 2002;20:2284-2292.
Does risk stratification improve neuroblastoma outcomes?
Prospective studies of risk stratification and risk-adapted therapy have sustained the excellent outcomes in patients with low-risk and intermediate-risk neuroblastoma while also reducing the incidence of adverse events; improved outcomes have also been achieved in those with high-risk disease.
|
Escp-high-risk-neuroblastoma-standard-clinical-practice
These guidance documents have been made publicly available by SIOP Europe – the. European Society of Paediatric Oncology and the European Reference Network for |
|
HIGH-RISK NEUROBLASTOMA: STANDARD CLINICAL PRACTICE
22 sept. 2022 If any doubt please share and ask for advice! Ideal world. Reality. Page 31. ESCP Webinars. Thanks to SIOP EUROPE Neuroblastoma Group. 31. |
|
Radiotherapy quality assurance in paediatric clinical trials: first
23 févr. 2023 than 3 years. 6 High risk neuroblastoma study 1 of SIOP-Europe (SIOPEN). 7 European Low and Intermediate Risk Neuroblastoma. 8 Molecular ... |
|
Heterogeneous MYCN amplification in neuroblastoma: a SIOP
14 mai 2018 Europe High-Risk Neuroblastoma Study 1 SIOPEN HR-NBL1 |
|
Nom de la direction sur 2 lignes maximum
9 mai 2022 ... high-risk neuroblastoma: a new approach to neuroblastoma therapy consortium study. ... Cancer-SIOP-European Neuroblastoma study. Eur J Cancer ... |
|
A European Cancer Plan for Children and Adolescents
8 sept. 2021 Similar initiatives were planned by SIOPEN (SIOP Europe Neuroblastoma Group) and the SIOP Europe Brain ... (FaR-RMS) and High-risk Neuroblastoma-2 ... |
|
Untitled
From 2002 to date: National Coordinator of the SIOP-Europe Neuroblastoma Protocol for high- risk Neuroblastoma (Study 1 - NBL-HR-01-SIOPEN). From May 2004 |
|
SIOP-PODC adapted risk stratification and treatment guidelines
Neuroblastoma is the most common extracranial solid tumor in childhood in high-income countries (HIC) where it accounts for. 10% of pediatric cancers [1]. |
|
Second Paediatric Strategy Forum for anaplastic lymphoma kinase
HR-NBL2: High-Risk Neuroblastoma Study 2 of SIOP-Europe-Neuroblastoma (SIOPEN). (NCT04221035)c. B7461036: Real World Data Collection Pediatric Neuroblastoma |
|
QUARTET: A SIOP Europe project for quality and excellence in
31 déc. 2021 neuroblastoma study 1 of SIOP-Europe ... paediatric oncology (Europe) neuroblastoma group's high-risk neuroblastoma trial: a SIOPEN study. |
|
Autologous stem cell transplant for high?risk neuroblastoma
Mar 2 2020 Patients were treated on the backbone of the high- risk neuroblastoma study-1 of SIOP-Europe (HR-NBL1/SIOPEN) protocol with ASCT. Adaptations. |
|
QUARTET: A SIOP Europe project for quality and excellence in
Jun 30 2022 Retrospective Radiotherapy Quality Assurance. HR-NBL1. High risk neuroblastoma study 1 of SIOP-Europe. (SIOPEN). Neuroblastoma. |
|
Paediatric Cancer
School of Cancer Sciences University of Birmingham |
|
A European Cancer Plan for Children and Adolescents
Sept 8 2021 the high-quality clinical and basic research carried out in Europe. SIOP Europe is committed to strengthening the. |
|
Rapid COJEC versus standard induction therapies for high?risk
induction schedules in patients with high-risk neuroblastoma (as defined by the International ENSG-5 stands for European Neuroblastoma Group Fi h Study. |
|
QUARTET: A SIOP Europe project for quality and excellence in
Jun 30 2022 Retrospective Radiotherapy Quality Assurance. HR-NBL1. High risk neuroblastoma study 1 of SIOP-Europe. (SIOPEN). Neuroblastoma. |
|
HIGH-RISK NEUROBLASTOMA STANDARD CLINICAL PRACTICE
These guidance documents have been made publicly available by SIOP Europe – the Approximately one third of children with high-risk neuroblastoma do not ... |
|
Survival in patients with High-Risk Neuroblastoma without
Jul 20 2020 High-risk neuroblastoma study 1 of SIOP-Europe (SIOPEN)/UK version/ August 2002/amended July. 2007. 16. Brodeur GM |
|
Paediatric Cancer
School of Cancer Sciences University of Birmingham |
|
Slaop - Molecular Assessment of Minimal Residual Disease in
Mar 21 2013 tic in different points of high-risk neuroblastomas (HR-NB) treatment [1 ... according to the HR-NB study of SIOP-Europe clinical protocol. |
|
O-001 High Risk Neuroblatsoma RESULTS OF AN - SIOP
This study aimed analysing high-risk neuroblastoma recurrence after high dose Median age at diagnosis was 39 5 mos (3 |
|
Recommendations for neuroblastoma in low - SIOP
21 mar 2015 · only 1–3 of cancers, and in most LMIC its true incidence is unknown [2–4] and high-risk neuroblastoma and applied these strategies during Results of the LNESG1 study by the SIOP Europe Neuroblastoma Group |
|
APN311 for high risk neuroblastoma in children and adolescents
12 mai 2015 · Journal of Clinical Oncology 2009;27(1):85-91 3 ClinicalTrials gov High risk neuroblastoma study 1 7 of SIOP-Europe (SIOPEN) |
|
High-Risk Neuroblastoma: Current and Future - JSciMed Central
27 oct 2015 · patients with high risk neuroblastoma Report from study 1 of SIOP Europe ( SIOPEN) Advances in Neuroblastoma Research Abstract Book |
|
Siop europe neuroblastoma (siopen)
1 jui 2014 · Amendment 1; 2005 November: R0 closed and protocol send out for approval High Risk Neuroblastoma Study 1 5 of SIOP-Europe (SIOPEN) |
|
Melphalan in high-risk neuroblastoma - Nature
4 avr 2016 · From 1980 to 2009, 215 patients aged >1 year with stage 4 NBL were treated with HD been reported, and three randomized studies showed that HDC European SIOP Neuroblastoma (SIOPEN)/HR-NBL randomized trial |
|
Survival of High-Risk Pediatric Neuroblastoma Patients In a - CORE
9-1-2016 Survival of High-Risk Pediatric Neuroblastoma Patients In a study population comprised 39 high-risk neuroblastoma patients, of whom 39 were aged N7(5) or European SIOP Neuroblastoma (SIOPEN HR-NBL1(6)) protocols |




















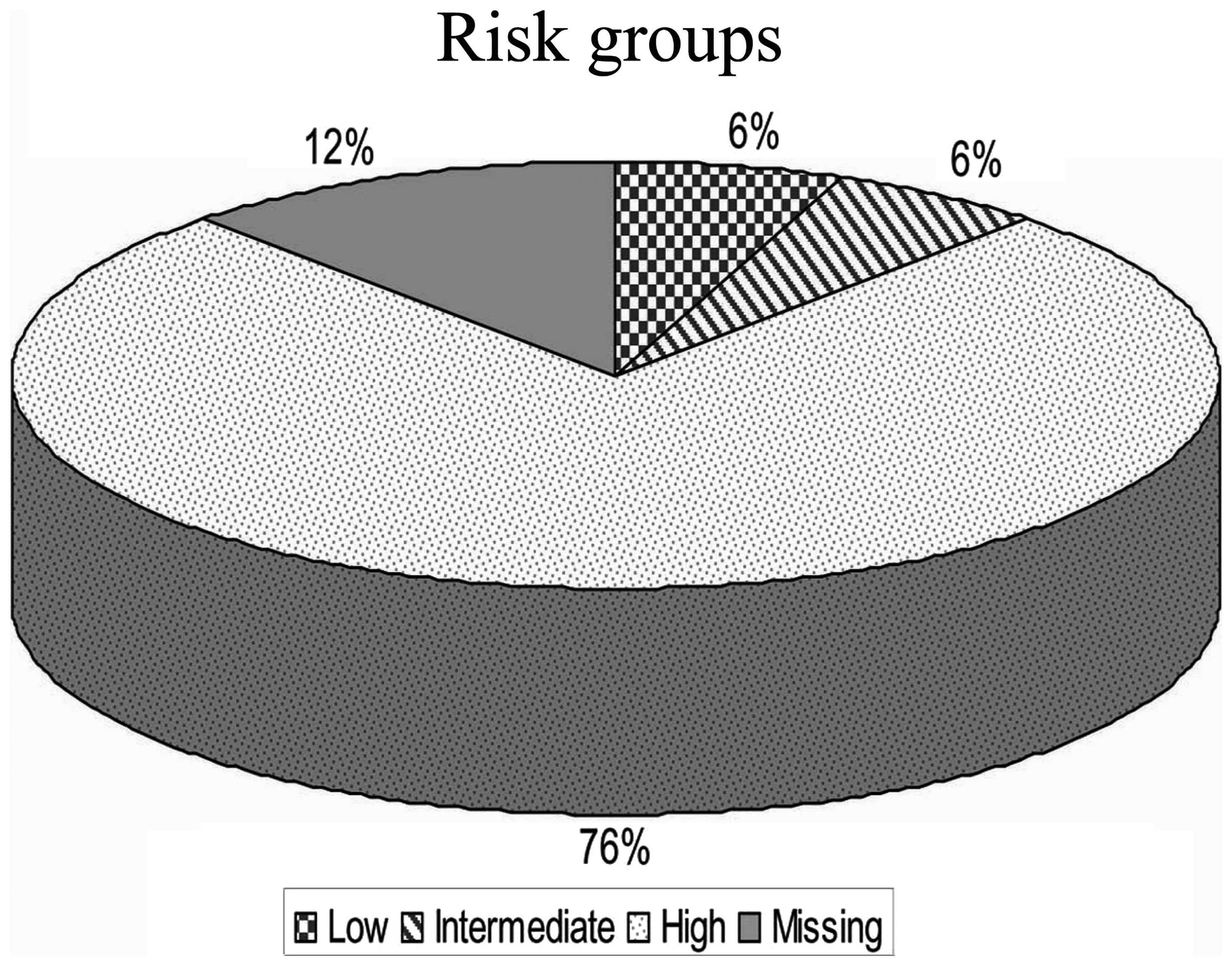











![PDF] Topotecan-Vincristine-Doxorubicin in Stage 4 High-Risk PDF] Topotecan-Vincristine-Doxorubicin in Stage 4 High-Risk](https://www.mdpi.com/cancers/cancers-13-00572/article_deploy/html/images/cancers-13-00572-g001.png)