activity coefficient table
|
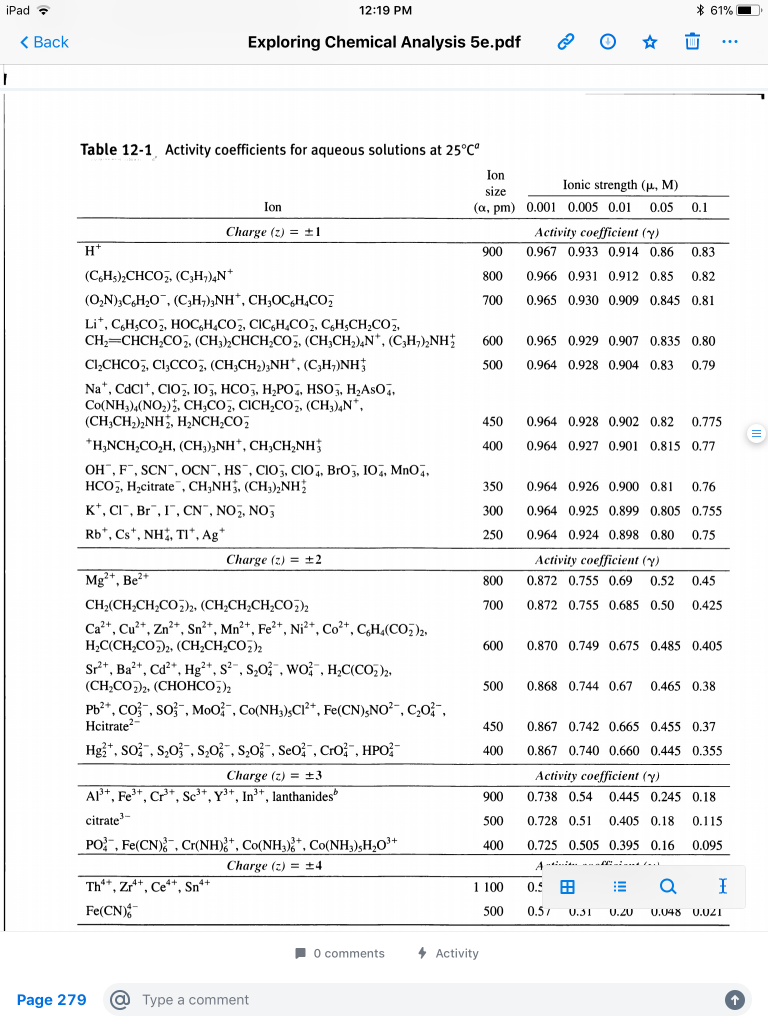
TABLE 7-1 Activity coefficients for aqueous solutions at 25 C
TABLE 7-1 Activity coefficients for aqueous solutions at 25 C Ion size Ionic Activity coefficient ( ) 900 0 967 0 933 0 914 0 86 0 83 800 0 966 |
|
Chem 355 Activity Coefficients
activity coefficient = 1 the activity = Molar concentration in expressions ○ As We look up in the table the activity coefficients based on this ionic |
|
Individual Activity Coefficients of Ions in Aqueous Solutions
It is the purpose of the present paper to present a revised and extended table of ionic activity coefficients which has largely been computed by |
|
Chem 355 Activity Coefficients
○ Activities activity coefficient ionic strength concepts Page 3 Effects of We look up in the table the activity coefficients based on this ionic |
How do you find the activity coefficient?
Activity coefficients may be determined experimentally by making measurements on non-ideal mixtures.
Use may be made of Raoult's law or Henry's law to provide a value for an ideal mixture against which the experimental value may be compared to obtain the activity coefficient.When activity coefficient is greater than 1?
Conversely, when activity coefficient is greater than 1.0, it implies that molecules have strong repelling force and exhibits positive deviation from Raoult's law.
In this case, lesser energy is needed to separate the constituent molecules.In solutions, the activity coefficient is a measure of how much a solution differs from an ideal solution—i.e., one in which the effectiveness of each molecule is equal to its theoretical effectiveness and thus the activity coefficient would be 1.
What is the activity coefficient of H3O+?
c = a/gamma and in the case of H3O+ (very dilute solution), the activity coefficient gamma is very close to 1.
Finally, the concentration of H3O+ in solution with pH = 7.4 is very close to 10^(7.4) = 39.8 nmol/L.
|
TABLE 7-1 Activity coefficients for aqueous solutions at 25 C Ion size
450. 0.867. 0.742. 0.665. 0.455. 0.37. 400. 0.867. 0.740. 0.660. 0.445. 0.355. Charge. 3. Activity coefficient ( ). 900. 0.738. 0.54. 0.445. 0.245. |
|
Chem 35.5 Activity Coefficients
Activity coefficient. Varies with ionic strength ai = [Xi]?i We look up in the table the activity coefficients based on this ionic. |
|
Chem 321 Lecture 11 - Chemical Activities
3 Oct 2013 be gotten from a table of activity coefficients such as shown below which lists ? as a function of ion charge |
|
Individual Activity Coefficients of Ions in Aqueous Solutions
table and no corrections were made even by a revised and extended table of ionic activity ... activity coefficient of the ith ion with valence q |
|
Theoretical mean activity coefficients of strong electrolytes in
Table. _No. 1. Relations between mean molality mean activity coefficient |
|
Activity coefficients of bicarbonates and carbonates in seawater
20 Feb 1975 Table 1. Total activity coefficients of bicar- bonate and carbonate ions in seawater at 25°C versus the salinity. Three significant. |
|
THE ACTIVITY COEFFICIENT OF STRONG ELECTROLYTES.1
Table VIII. Activity Coefficients of Sodium Chloride at 25° from Electromotive Force. m. 0.1. 0.784. |
|
Chapter 6 Activity Scales and Activity Corrections
11 Oct 2004 6.1 Total Activity Coefficient: Electrostatic Interactions and Ion ... activity coefficients (Table 6-1) (e.g. Table 5.2 of Libes). |
|
Osmotic Coefficients and Mean Activity Coefficients of Uni-univalent
29 Oct 2009 to evaluate activity coefficients of a selected electrolyte over a range of concentrations is illustrated here (table 1). |
|
Activity Coefficient Ratios of Nitric Acid and Silver Nitrate and their
Table II. Equilibrium Constant and Resin Activity Coefficients in Na+-H + Exchange. Ionic strength 1.0 M. Ionic strength 0.3 M |
|
Individual Activity Coefficients of Ions in Aqueous Solutions - UFSCar
Lewis and Randall2 in 1923 published a table of 26 individual ionic activity coefficients, which has subsequently been of frequent use to chemists The authors |
|
Chem 321 Lecture 11 - Chemical Activities - CSUN
3 oct 2013 · Suppose, for example, you require the activity coefficient for a solution that has an ionic strength of 0 007 M In the table of activity coefficients you |
|
Theoretical mean activity coefficients of strong electrolytes in
List of Tables Table _No 1 Relations between mean molality, mean activity coefficient, solute (or electrolyte) activity, and ionic strengt tor various valence |
|
TABLE 7-1 Activity coefficients for aqueous solutions at 25 C Ion size
TABLE 7-1 Activity coefficients for aqueous solutions at 25 C Ion size Ionic strength ( , M) Ion ( , pm) 0 001 0 005 0 01 0 05 0 1 Charge 1 Activity coefficient |
|
Activity coefficient - Chem 355
Activity of An Ion is “effective concentration” Activity coefficient Varies with ionic strength We look up in the table the activity coefficients based on this ionic |
|
Chapter 6 Activity Scales and Activity Corrections
11 oct 2004 · 6 1 Total Activity Coefficient: Electrostatic Interactions and Ion Complexing The goal activity coefficients (Table 6-1) (e g Table 5 2 of Libes) |
|
Ionic strength
activity coefficients and ionic strength up to μ ≈ 0 1 M In theory, α is the diameter of the hydrated ion However, sizes in Table 7-1 (see next slide) cannot be |
|
TEMPERATURE EFFECTS ON THE ACTIVITY COEFFICIENT OF
Finally, the Ion Interaction Model is utilized to solve various problems of water chemistry where bicarbonate is present in solution Page 5 v TABLE OF |
|
Research Article New Data on Activity Coefficients of - CORE
Table 1: Individual activity coefficient and mean activity coefficient values of KNO3 solution m (mol/kg) γK+ γNO3 − γ± (Expt ) γ± (Hamer and Wu) [10] 0 0012 |