classification of colloids
What are 4 types of colloids give its meaning?
Types of Colloids
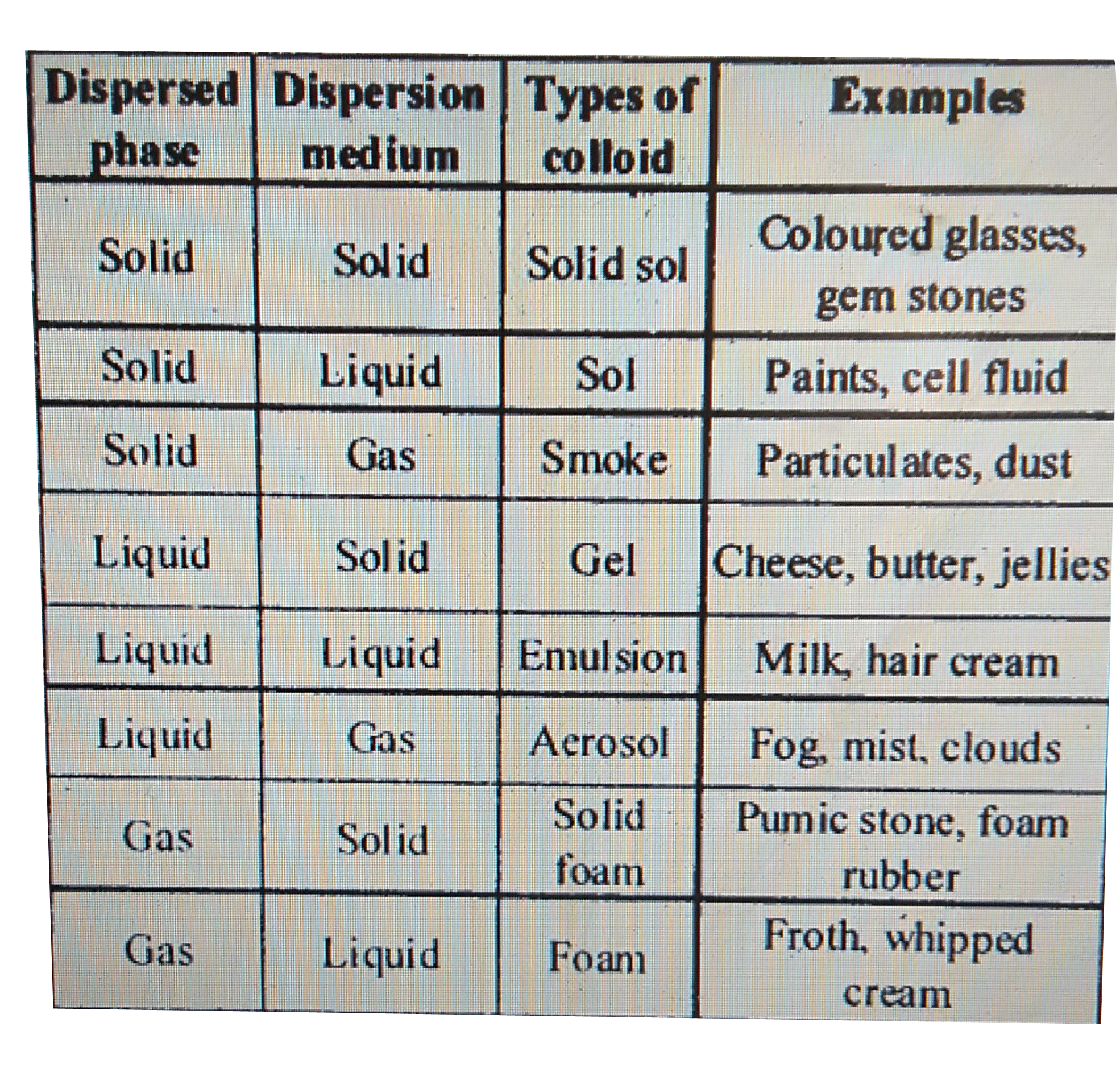
Sol – It is a suspension of minute solid particles in a liquid.
Emulsion – It is a colloid between two or more liquid with one consisting a dispersion of another liquid.
Foam – It consists of gas dispersed in solid or liquid.
Aerosol – It consists of a minute liquid or solid particles in a gas.Colloids are classified on the basis of physical states of components, that is, on the basis of physical states of the dispersed phase and dispersion medium.
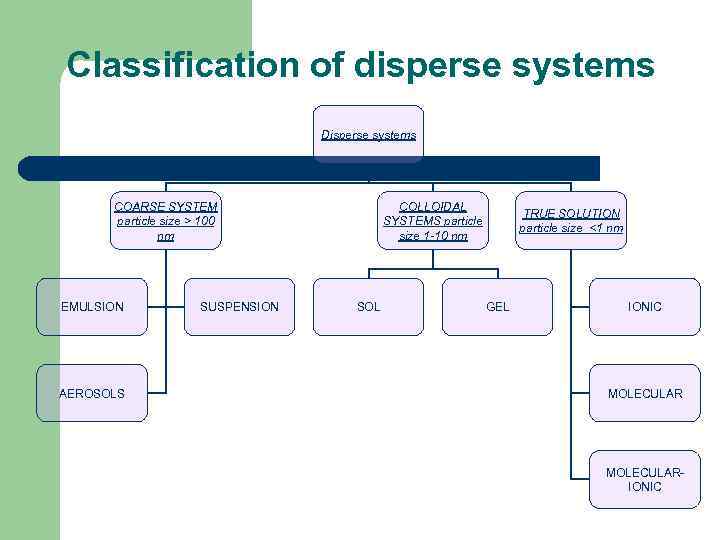
Colloidal solution is classified into eight types of colloidal systems.
What is the classification of colloids on the basis of nature of interaction?
When the dispersion phase and dispersion medium interact then colloids are classified into lyophilic sols and lyophobic sols.
The dispersed phase particles have a great affinity for the dispersion medium in lyophilic sols, Which are reversible.
Examples are gum, gelatin, starch, proteins, etc.
What classifies a colloid?
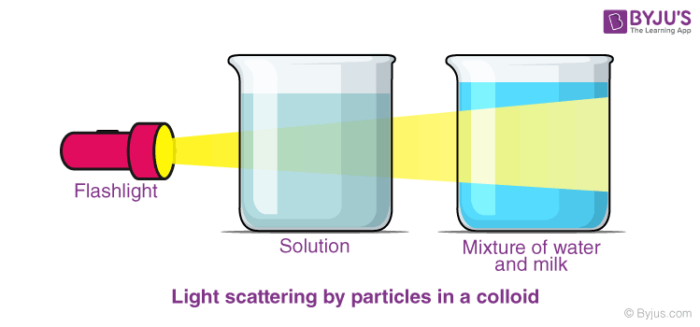
colloid, any substance consisting of particles substantially larger than atoms or ordinary molecules but too small to be visible to the unaided eye; more broadly, any substance, including thin films and fibres, having at least one dimension in this general size range, which encompasses about 10−7 to 10−3 cm.
|
B. Sc. II-Sem Colloidal state (1) The foundation of colloidal chemistry
(ii) Classification based on Nature of interaction between dispersed phase and dispersion medium: Depending upon the nature of interactions between dispersed |
|
Colloids Classification of Colloids
Colloids are classified on the basis of the following criteria: (i) Physical state of dispersed phase and dispersion medium. (ii) Nature of interaction between |
|
Surface Chemistr face Chemistr face Chemistry
enumerate the nature of colloidal state;. • describe preparation properties and purification of colloids;. • classify emulsions and describe their preparation |
|
Phases of colloids and their classification.
For example in the colloidal solution of silver in water (silver acts as a dispersed phase). (ii) External phase or Dispersion medium (continuous phase):It is. |
|
BP 403T. Physical Pharmaceutics-II (Theory) Unit1 (Part I) Prepared
& their general characteristics size & shapes of colloidal particles |
|
ANNUAL ACADEMIC PLAN 2023-24
4.3 Colloids. 4.4 Classification of Colloids. 4.5 Emulsions. 4.6 Colloids Around Us. PRACTICALS: B. Chemical Kinetics. C. Solutions. 5. GENERAL PRINCIPLES OF |
|
COLLOIDS:
CLASSIFICATION OF COLLOIDS: AgNO. 3. + CuCl. 2. Page 8. A solid suspended in another solids are SOLID. SUSPENSIONS. CLASSIFICATION OF COLLOIDS: There are no GAS |
|
The Composition of Soil Colloids in Relation to Soil Classification
Introduction. For a period of approximately fifteen years investigators in the Bureau of. Chemistry and Soils have been engaged in the accumulation of |
|
Colloids and Surfaces Prof. Basavaraj Madivala Gurappa
I have defined what is a colloid okay we are going to look at what is a colloidal dispersion to begin with |
|
HANDOUT CLASSIFICATION AND PREPARATION OF COLLOIDS
colloidal particles are dispersed. Classification of Colloids. Classification in terms of the state of the disperse phase and dispersion medium. Dispersed. |
|
B. Sc. II-Sem Colloidal state (1) The foundation of colloidal chemistry
For example in the colloidal solution of silver in water. Water act as a dispersion medium. (2) Classification of colloids: The colloids are classified on |
|
The Composition of Soil Colloids in Relation to Soil Classification
This view regards the colloid complex as essentially a group of acids organic and inorganic |
|
Hermann Staudinger - Nobel Lecture
stance can be broken down into particles of colloid size by appropriate. Table 3. Classification of colloidal solutions of organic substances. |
|
Lech105.pdf
enumerate the nature of colloidal state;. • describe preparation properties and purification of colloids;. • classify emulsions and describe their preparation |
|
Colloidal dispersion ppt.pdf
Colloids are dispersions where in dispersed particles are Classification of colloidal dispersion: 1.Basing on charge- (+) (-). |
|
COLLOIDS Subject: Physical Pharmacy Subject code:PHCY102
and suspensions red blood cells. * 1 nm (nanometer) = 10-9 m; 1 µm (micrometer) = 10-6 m. CLASSIFICATION OF COLLOIDAL SYSTEMS ACCORDING TO PARTICLE SIZE. |
|
The Colloidal State Introduction: A colloid is one of the three primary
Classification. Lyophilic colloids (solvent loving): They are so called because of affinity of particles for the dispersion medium. |
|
Nature vol 138-n3489.indd
also needed for the 'educated classes' and an studied soil formation and classification from the ... The two great classes of colloids found in the. |
|
A classification scheme for marine organic colloids in the Adriatic
A classification scheme for marine organic colloids in the Adriatic Sea: colloid speciation by transmission electron microscopy. |
|
Sedimentation classification of silver colloids for surface-enhanced
Sedimentation Classification of Silver Colloids for. Surface-Enhanced Raman Scattering. Rong-Sheng Sheng1 Ling Zhu |
|
B Sc II-Sem Colloidal state (1) The foundation of colloidal chemistry
7 avr 2020 · For example, in the colloidal solution of silver in water Water act as a dispersion medium (2) Classification of colloids: The colloids are |
|
COLLOIDAL SOLUTIONS
CLASSIFICATION OF COLLOIDAL SYSTEM : Size of colloidal particles: ➢ monodispersive (particles of dispersed phase have the same dimensions) |
|
COLLOIDAL DISPERSIONS Dispersed systems consist of - IGNTU
Based on the interaction between dispersed phase and dispersion medium, colloidal systems are classified as (a) Lyophilic colloids (solvent-loving) (When the |
|
The Composition of Soil Colloids in Relation to Soil Classification
“The Chemical Composition of Soil Colloids,” U S D A Bulletin No 1311 (19241 Page 5 SOIL COLLOIDS AND SOIL CLASSIFICATION 3 |
|
Classification of Matter
following Foldable to help you classify heterogeneous and homogeneous mixtures, colloids, and suspensions Heterogeneous Mixture Colloid Suspension |
|
8 COLLOIDS - NIOS
identify phases of colloidal solution; ○ classify colloidal solutions; ○ describe methods of preparation of colloids; ○ explain some properties of colloidal |
|
Types of disperse systems Dispersed phases What colloids - IFM
Macromolecular radius R? A historical example: Classification of bacteria Three primary bacterial forms: coccus - spherical bacillus – rod-like spirillum – spiral |
|
Details of Module and its structure - NROER
Colloidal systems are classified in three ways depending upon (i) physical states of the dispersed phase and dispersion medium (ii) nature of interaction between |
|
1 Introduction - Wiley-VCH
For example, a clay dispersion in water could be classified as a lyophobic colloid with hydrophilic surfaces Simple colloidal dispersions are two-phase systems, |





![Colloidal disp[1] ppt [compatibility mode] Colloidal disp[1] ppt [compatibility mode]](https://image.slidesharecdn.com/colloidsppt-161019033051/95/colloids-presentation-slides-41-638.jpg?cb\u003d1476847880)