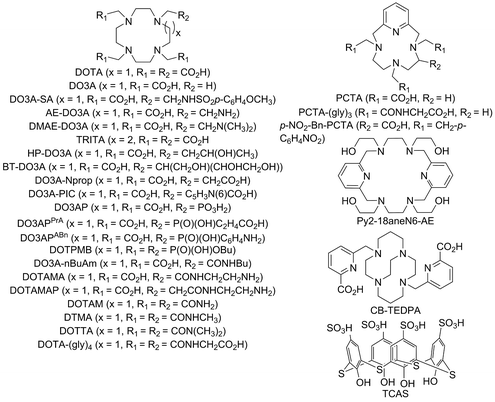
briefly explain stepwise and overall formation constants
|
PG
▫ describe stepwise and overall formation constants; ▫ explain thermodynamic importance of stability constants; ▫ discuss factors affecting stability of |
|
Trends in Stepwise Constants Dalal Institute
The values of stepwise equilibrium constants for the formation of a particular metal-complex decrease successively in most of the cases i e K1 > K2 > K3 |
What is the trend in step wise formation constants?
The order of stepwise formation constants is observed as follows, K1 > K2 > K3 < K4, which is not in agreement with the common trend of K1 > K2 > K3 > K4.
The formation constant is the equilibrium constant for the reaction leading to the formation of a coordination complex.
The greater the formation constant, the more thermodynamically stable the product complex compared to the reactant complex.25 oct. 2022
What is the concept of formation constant?
The formation constant, which can also be referred to as the stability or association constant, occurs when a complex is formed.
A complex is formed when a metal ion reacts with a ligand.
A ligand is a molecule or ion that contains at least one lone pair of electrons.
What is stepwise formation constant?
What are Stepwise Stability Constants? Stepwise stability constants are equilibrium constants given for each step of the process of ligand substitution.
When a transition metal ion complex has water ligands surrounding the metal ion, the ligand substitution takes place as a stepwise process.18 fév. 2018
|
Stepwise and Overall Formation Constants and Their - Dalal Institute
Kn are the equilibrium constants for different steps, which in turn also imparted their conventional label of stepwise stability or the stepwise formation constants The magnitude of these individual equilibrium constants indicates the extent of the formation of different species in a particular step |
|
BSc Chemistry - e-PG Pathshala
The overall constant, β is equal to the product of the stepwise formation constants K1, K2 Kn Also the formation of a complex takes place in various stepwise equilibria The inverse of the formation constant is called the dissociation constant (Kd) or instability constant of the complex |
|
UGC Minor Research Project - Shri RR Lahoti Science College
in human physiology A brief survey of the study of binary complexes of these metal ions with describe stepwise and overall formation constants; ▫ explain |
|
The stability constants of some carboxylate complexes of - CORE
of explaining the general features of the data found by several other workers The step formation constant of the complex MAn Is defined as follows : Kn : [Mnl |
|
Coordination Compounds - NCERT
measurements in solution can be explained if (i) six groups in all, either chloride The stepwise and overall stability constant are therefore related as follows: β4 9 25 What is meant by stability of a coordination compound in solution? State |
|
Automatically generated PDF from existing images
b) What is the general trend in the relative magnitudes of stepwise stability constants of a complex ? What are the reasons for the trend ? c) Explain the |
|
Ligands - Chemistry 332 Basic Inorganic Chemistry II
The coordination number of a metal center is defined as the number of We start with a Co2+ salt what is the oxidation state of Co in the product? Why do we use the Typically expressed in terms of an overall formation or stability constant (This is K st are the stepwise formation constants and provide insight into the |