alcohols esters aldehydes and ketones
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
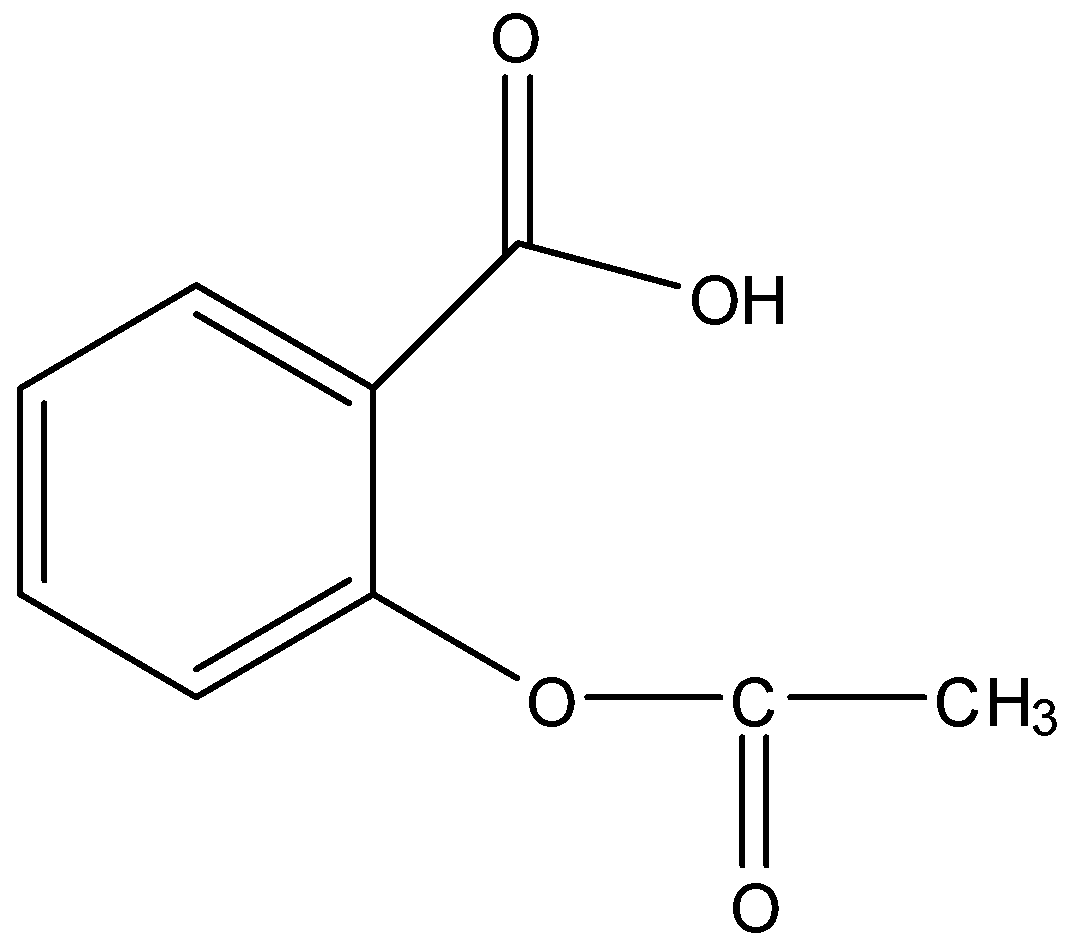
Esters and anhydrides are derivatives of carboxylic acids The general formulas of these classes of compounds are given below: Rationalised 2023-24 Page |
In alcohols, the functional group is OH, which does not have any C in it and hence cannot be considered as a terminal group.
Thus an aldehyde has -CHO and a carboxylic acid has -COOH and both are terminating groups, while -OH has no Carbon and is therefore not a terminal group.
What is relation between aldehyde and ketone or alcohol and ether?
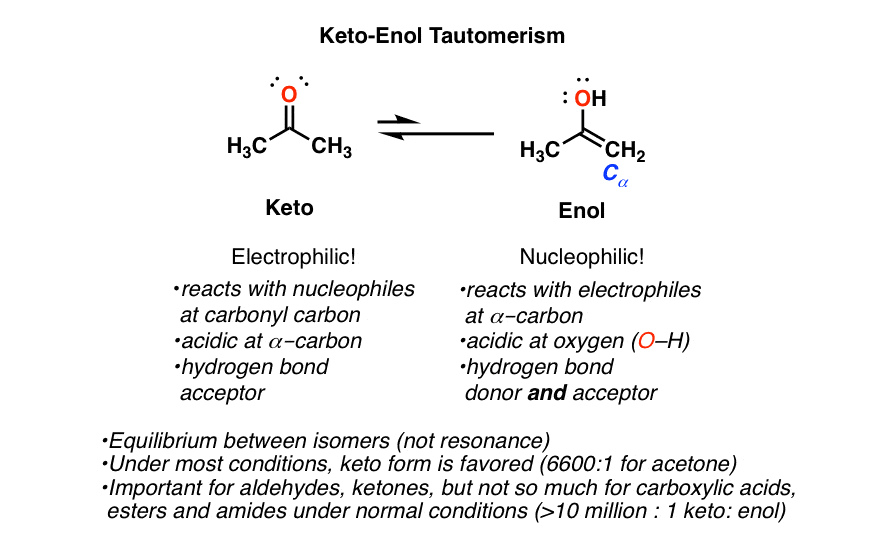
Overall, aldehydes and ketones are related due to their similar carbonyl group but differ in their position within the molecule.
Similarly, alcohols and ethers are related due to their oxygen atom but differ in the presence of a hydroxyl group and the polarity of the molecule.
What is the difference between an aldehyde and a ketone and an ester?
The carbonyl group, a carbon-oxygen double bond, is the key structure in these classes of organic molecules: Aldehydes contain at least one hydrogen atom attached to the carbonyl carbon atom, ketones contain two carbon groups attached to the carbonyl carbon atom, carboxylic acids contain a hydroxyl group attached to 27 oct. 2022
|
Alcohols, Ethers, Aldehydes, and Ketones
Aldehydes and ketones don't form hydrogen bonds to each other, however, they form hydrogen bond with water using the electron lone pairs on oxygen Aldehydes and ketones have lower boiling points compared to alcohols of similar size Aldehydes and ketones with fewer than five carbons are soluble in water |
|
Alcohols, Ethers, Aldehydes, and Ketones
Given the structure of an alcohol, ether, thiol, sulfide, aldehyde, or ketone reduction of aldehydes and ketones carboxylic acids, esters, and amides |
|
Volatile Alcohols, Aldehydes, Ketones and Esters
Volatile alcohols, aldehydes, ketones and esters occur widely in plants, and they are not restricted to the fruits, flowers and leaves though often most concen- |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
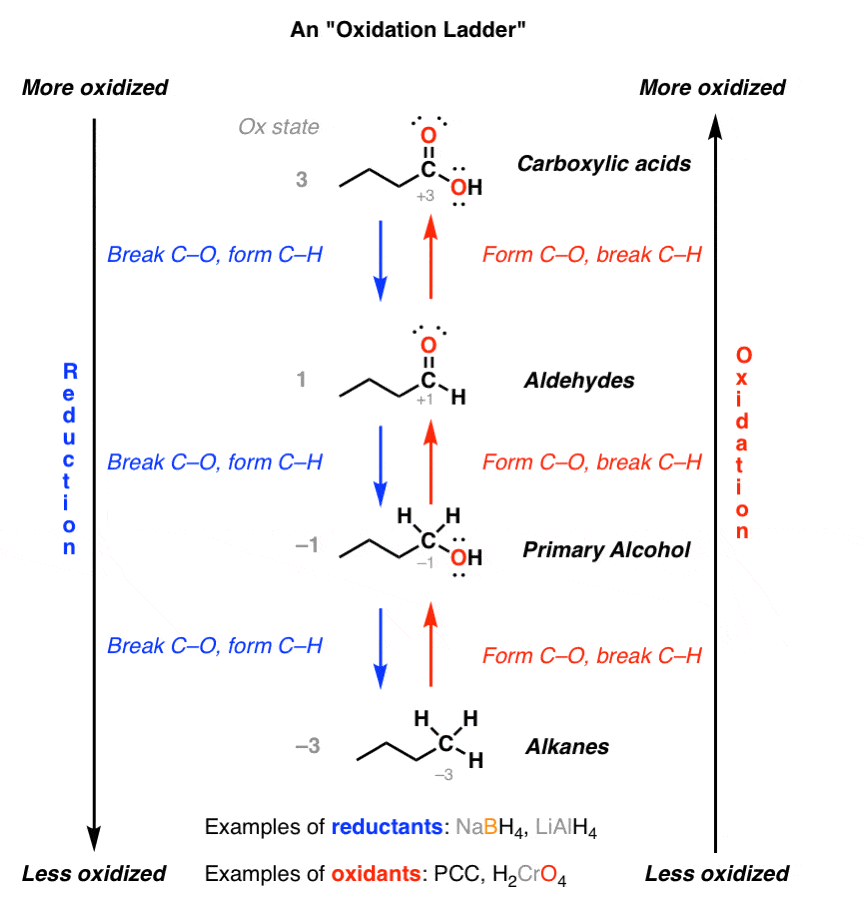
Ketones • Carboxylic acids R C H O Carboxylic acid derivatives: • Esters • Anhydrides OH oxidation • Alcohols are oxidized to aldehydes and ketones |
|
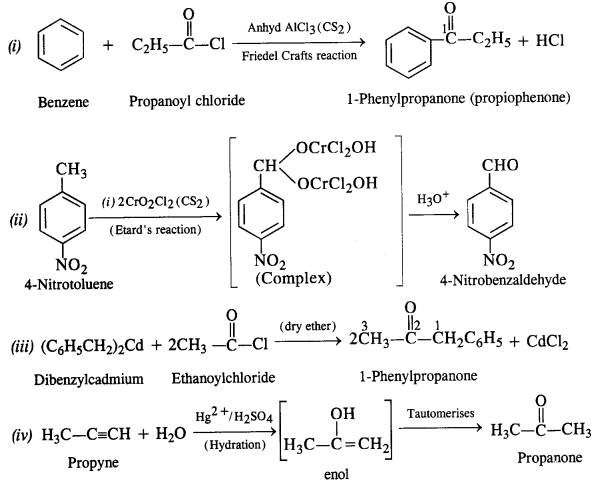
Aldehydes, Ketones and Carboxylic Acids - NCERT
carboxylic acids, and their derivatives (e g esters, anhydrides) while in (i) Reduction to alcohols: Aldehydes and ketones are reduced to primary and |
|
1 Chapter 15: Alcohols, Diols, and Thiols 151: Sources of Alcohols
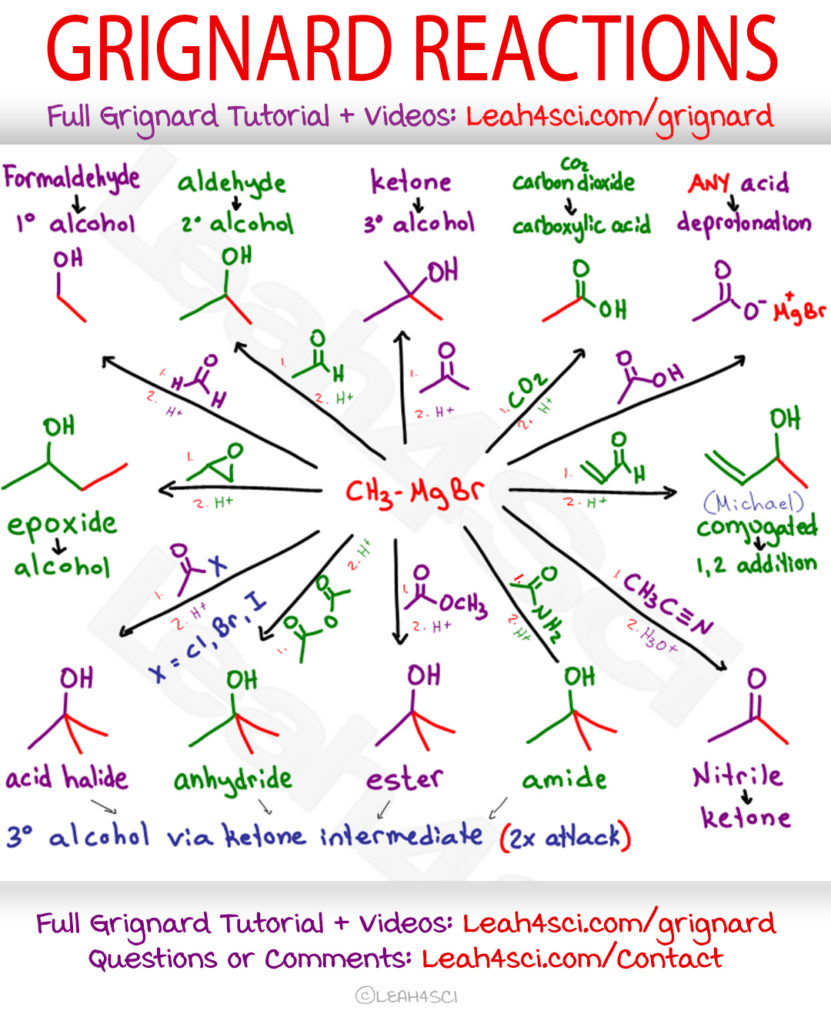
NaBH4 reduces aldehydes to primary alcohols NaBH4 reduces ketones to secondary alcohols NaBH4 does not react with esters or carboxylic acids |
|
Chapter 15: Alcohols, Diols, and Thiols 151: Sources of Alcohols
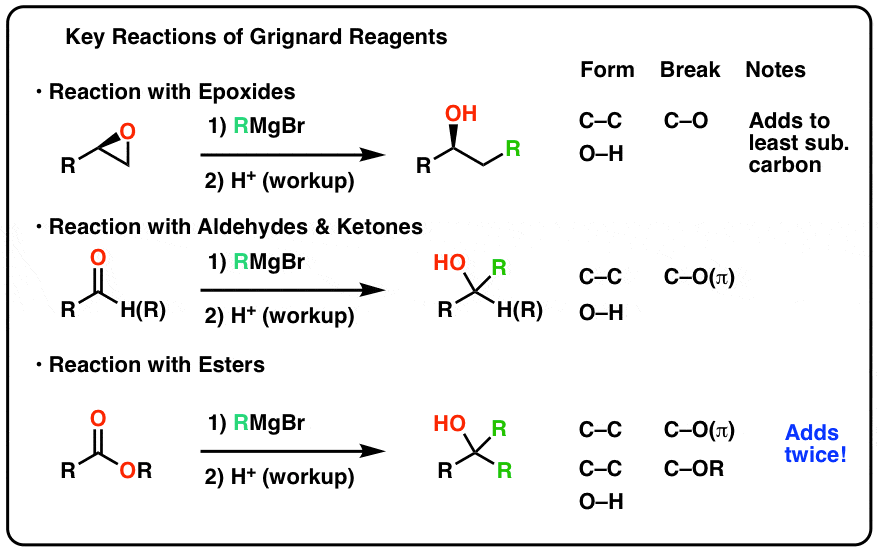
2) H3O+ ketones 2° alcohols aldehydes 1° alcohols 78 15 3: Preparation of Alcohols By Reduction of Carboxylic Acids (and Esters) - LiAlH4 (but not NaBH4 |
|
Reduction
Diazene-Mediated Deoxygenation ester aldehyde alcohol alkane R OH O R Aldehydes, ketones, epoxides, and several other functional groups can also be |
|
27 ALDEHYDES, KETONES AND CARBOXYLIC ACIDS - NIOS
such as aldehydes and ketones as well as carboxylic acids and their derivatives aldehydes and secondary alcohols can be oxidised to ketones 2 acid Carboxylic Alcohol Ester Note that the acid catalysed esterification is an equilibrium |
|
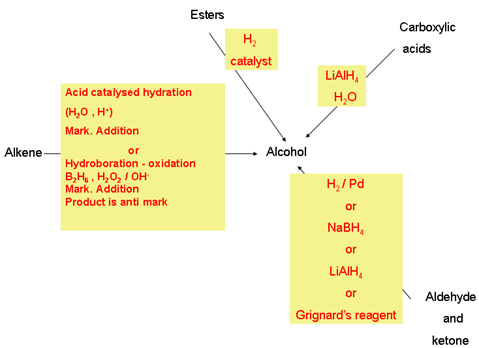
Chapter 12 - Alcohols from Carbonyl Compounds1
other from the alkoxyl part of the ester - Aldehydes and ketones are easily reduces by NaBH4 12 4 - Oxidation of Alcohols - 1◦ alcohols can be oxidized to |
![Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry](https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/2-oxidation-ladder-showing-oxidation-states-of-alkane-to-secondary-alcohol-ketone-ester-going-from-less-oxidized-to-more-oxidized.gif)