co2 ka molar mass
What is the density of carbon dioxide?
At standard temperature and pressure, the density of carbon dioxide is around 1.98 kg/m 3, about 1.53 times that of air. Carbon dioxide has no liquid state at pressures below 0.51795 (10) MPa (5.11177 (99) atm ).
What is the molar mass of CO2?
CO 2 has one carbon atom and two oxygen atoms. The molar mass of carbon dioxide is 12.01 + (2 × 16.00) = 44.01 g/mol. Weights of atoms and isotopes are from NIST article. Molar mass calculator computes molar mass, molecular weight and elemental composition of any given compound.

Molar Mass of CO2 Chemistry

How to Find the Mass of One Molecule of Carbon dioxide (CO2)

How to find the Molar Mass of CO: Carbon monoxide
|
Monomodal Ultrahigh-Molar-Mass Polycarbonate Homopolymers
Nov 21 2019 Monomodal Ultrahigh-Molar-Mass Polycarbonate Homopolymers and Diblock Copolymers by Anionic Copolymerization of Epoxides with CO2. |
|
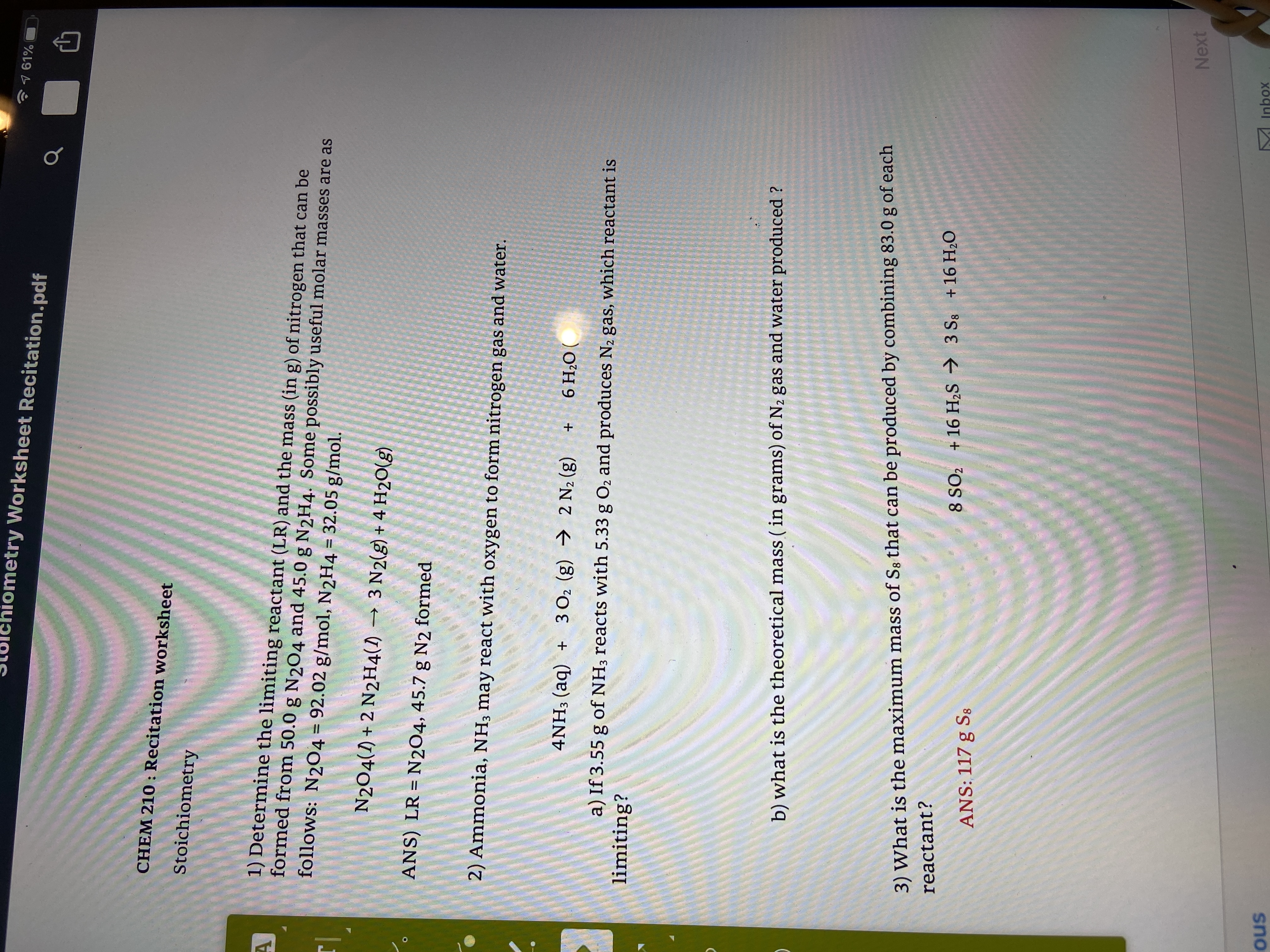
AP Chemistry 2021 Free-Response Questions
m = mass. M = molar mass. D = density. KE = kinetic energy v = velocity (Assume that the amount of CO2(g) dissolved in the solution is negligible.). |
|
Untitled
The molar solubility of CaCO3(s) in a saturated solution can be decreased by: 10) a) What is the mass of one mole of cobalt atoms in grams? Ans! |
|
Chemical Formulae
%20%25.pdf |
|
Chemistry – A Molecular Sciences Appendices
To calculate a molar mass we simply sum up the contributions of each element or atom. For carbon dioxide |
|
CH2 hmwk - CHEM 1411
The molar mass of the compound is 78.1 g/mol. What is the molecular formula of the compound? 3.385 g CO2 |
|
Tutorial 6
molar mass. 44.0 g CO2 / mol. D = = = 1.96 g/L molar volume. 22.4 L CO2 / mol. EXAMPLE: Calculate the density of carbon dioxide at 100oC and 850 mm Hg. |
|
CaCO3(s) ? ? CaO(s) + CO2(g) When heated calcium carbonate
CaO(s) + CO2(g). When heated calcium carbonate decomposes according to the equation above. In a study of the decomposition of calcium carbonate |
|
Chapter 4 - Stoichiometry of Chemical Reactions
Write and balance chemical equations in molecular total ionic |
|
Problem Set 1
From the periodic table, we see that the molar mass of gold is 196 967 g/mol We need to convert the given mass of carbon dioxide generated to moles of CO2 |
|
Calculating the Molar Volume of Carbon Dioxide
Determine the molar volume of carbon dioxide by measuring the mass and vol- ume of a carbon dioxide gas sample at laboratory temperature and pressure |
|
How much volume does 1 kg of CO2 occupy at room temperature
CO2 has a molecular weight of 44 g/mol 1 kg CO2 = 1000 g × (1 mol/44 g) = 22 7 mol CO2 V=nRT/P, V=(22 7)(0 0821)(300)/1 = 559 L CO2 at 27°C (300K), |
|
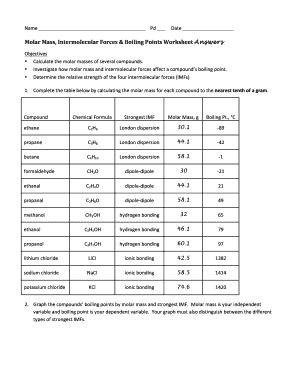
Mole & Molar Mass - Montgomery College
How many molecules are in 2 50 moles of CO2? 5 Calculate the mass , in grams , of 0 433 mol of calcium nitrate Hint: Formula Weight of Ca(NO3)2 |
|
Carbon dioxide, CO2, is usually a gas It is exhaled by - Cal State LA
Carbon dioxide is a chemical compound that consists of two oxygen atoms and one The molecular formula for carbon dioxide is CO2 The molar mass for it is |
|
Mole Calculations - UTC
Terms used: Molar mass ~ Molecular weight ~ Formula weight This means the terms are interchangeable: For 1 mole of CO2 = 44 0 g/mol For 1 average |
|
Student Academic Learning Services - Durham College
26 juil 2013 · What is the molar mass for each of the following compounds? CO2 H2SO4 Mg3( PO4)2 Find the atomic mass of one atom for each element |
|
Calculations with Chemical Reactions - George Brown College
Molar mass– the mass of 1 mole of a substance (molecule or atom) 2 Fe(s) + 3 CO2(g) Solution 1: Using the Step-by-Step Proportion Technique uqn: mass |
|
122 Practice Final
(E) The average kinetic energy of the CO2 molecules is greater than that of the H2 molecules Molar Mass MgCl2 95 2 g mol-1 9 If 1 L of a gas at 30 °C and |
|
Download pdf - Commack Schools
A compound has a molar mass of 90 grams per mole and the empirical If 4 mol of CoH1206 is produced by the process, how many moles of CO2 were used? |












![WS - Molar Mass - [PDF Document] WS - Molar Mass - [PDF Document]](https://online.fliphtml5.com/nzsh/daqo/files/large/1.jpg)

:max_bytes(150000):strip_icc()/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)













/chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)