co2 molar mass calculator
How do you find the molar mass of carbon?
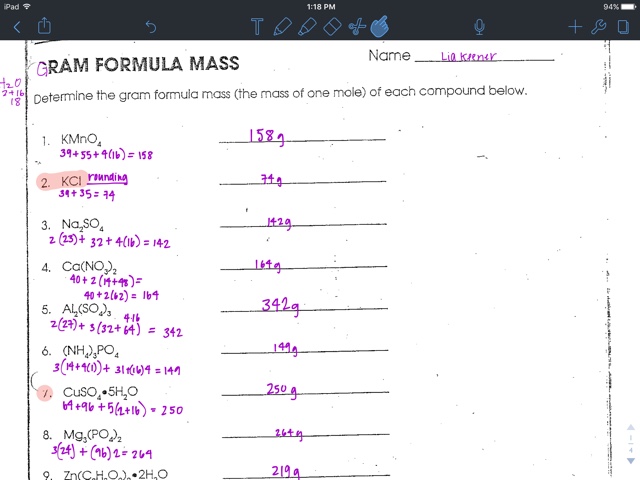
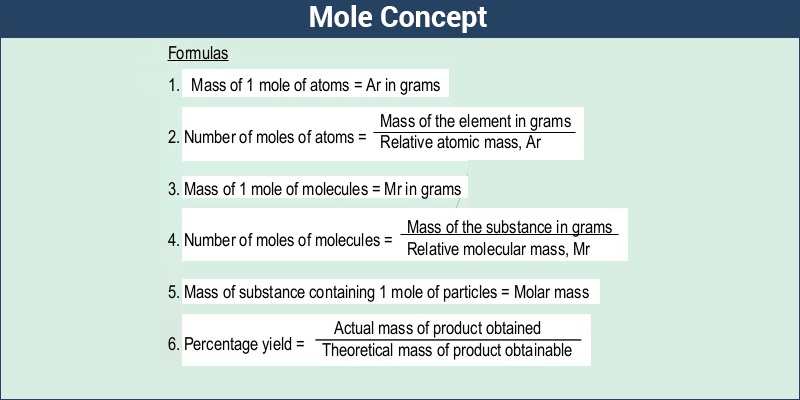
For example, if you want to find the molar mass of carbon, you would find the atomic mass of carbon on the periodic table, and this is equal to the molar mass in grams per mole. So, in our example, carbon has a molar mass of 12.01 grams per mole. There are a few exceptions to this rule.
What is the molar mass of a compound?
The molar mass of any compound is the mass in grams of one mole of that compound. One mole of carbon dioxide molecules has a mass of 44.01g 44.01 g, while one mole of sodium sulfide formula units has a mass of 78.04g 78.04 g. The molar masses are 44.01g/mol 44.01 g/mol and 78.04g/mol 78.04 g/mol respectively.

How To Calculate The Molar Mass of a Compound

Molar Mass of CO2 Chemistry

Molar Mass / Molecular Weight of CO2: Carbon dioxide
|
Chapter 3: Stoichiometry
?Calculate formula weights. ?Convert grams to moles and moles to grams using molar masses. ?Convert number of molecules to moles and moles to number of |
|
AP® CHEMISTRY 2009 SCORING GUIDELINES - Question 2 (10
(d) Using the information above calculate the value of the molar mass of the unknown gas. Occurrence 1: The flask was incompletely flushed with CO2(g) |
|
Mole & Molar Mass
How many molecules are in 2.50 moles of CO2? 5. Calculate the mass in grams |
|
Problem Set 1
Notice that three compounds SO2 |
|
Chemistry 51 ASNWER KEY 1 REVIEW QUESTIONS Chapter 6 1
Calculate each of the following quantities: Molar mass = [9(12.0)+8(1.01)+4(16.0)] = 180.1 g/mol ... Number of molecules in 1.75 moles of CO2. |
|
Tutorial 6
molar mass. 44.0 g CO2 / mol. D = = = 1.96 g/L molar volume. 22.4 L CO2 / mol. EXAMPLE: Calculate the density of carbon dioxide at 100oC and 850 mm Hg. |
|
Calculations with Chemical Reactions
The molar mass of carbon monoxide is 28.01 g/mol and the molar mass of iron is 55.85 g/mol. Fe2O3(s) + 3 CO(g) ? 2 Fe(s) + 3 CO2(g). |
|
Calculating the Molar Volume of Carbon Dioxide
– ma. So we can calculate the mass of CO2 in the flask using Equation 4. m. CO 2. = m2 – m1 + ma = 82.237 g – 82.151 g + 0.198 g = 0.284 |
|
Sample Exercise 13.1 Predicting Solubility Patterns
Calculate the molality of glucose in the solution. Water has a density of 1.00 g/mL. Plan We use the molar mass of C6H12O6 to convert grams to moles. We use |
|
Equations included in the Irrigated Cotton Calculator under the
Cg =Conversion factor of 44/28 to convert elemental mass of N2O to molecular mass (t N2O. t. N2O-N-1). Annual direct emissions in CO2-e from synthetic |
|
Problem Set 1
conversions and the formula for volume from surface area and depth: We need to convert the given mass of carbon dioxide generated to moles of CO2, then to |
|
Calculating the Molar Volume of Carbon Dioxide
From VSTP and the mass of the collected gas, we can calculate the molar volume of the sample at STP Example Problem You determine that in a laboratory |
|
Calculations with Chemical Reactions - George Brown College
Molar mass– the mass of 1 mole of a substance (molecule or atom) 2 Fe(s) + 3 CO2(g) Solution 1: Using the Step-by-Step Proportion Technique uqn: mass Step 2: Calculate the amount of Fe, n, produced from 5 448054266 mol of CO |
|
Mole Calculations - UTC
Terms used: Molar mass ~ Molecular weight ~ Formula weight This means the terms are interchangeable: For 1 mole of CO2 = 44 0 g/mol For 1 average |
|
Mole & Molar Mass - Montgomery College
How many molecules are in 2 50 moles of CO2? 5 Calculate the mass , in grams , of 0 433 mol of calcium nitrate Hint: Formula Weight of Ca(NO3)2 = (AW of |
|
NCERT Solutions for Chapter 1 Some Basic Concept - Studymate
(ii) Molecular mass of CO2 = 12 01 amu + 2 ×16 00 amu = 44 01 amu Determine the molecular formula of an oxide of iron in which the mass per cent of iron |
|
Download pdf - Commack Schools
What mass of carbon dioxide occupies a volume of 22 4 L = I mole 22 4 liters at STP? Imole = Gram formula mass A) 22 0 g B) 44 09 C) 66 0 g CO2 = 12 + 32 - |
|
CONCENTRATION ATOM MOLECULE MASS VOLUME MOLAR
b) If you have 0 25 moles of carbon dioxide, how many molecules do you have? Atoms and molecules Calculate the molar mass of the following substances: |
|
How much volume does 1 kg of CO2 occupy at room temperature
CO2 has a molecular weight of 44 g/mol 1 kg CO2 = 1000 g × (1 mol/44 g) = 22 7 mol CO2 V=nRT/P, V=(22 7)(0 0821)(300)/1 = 559 L CO2 at 27°C (300K), |
|
CHAPTER 5 GASES - ResearchGate
If we can determine the moles of CO2, we can calculate Strategy: We can calculate the molar mass of a gas if we know its density, temperature, and pressure |





/chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)
/the-periodic-table-of-the-elements-in-a-school-in-san-francisco--california--75471038-5a77c1650e23d90036d6c27c.jpg)