co2 molar mass g/mol
|
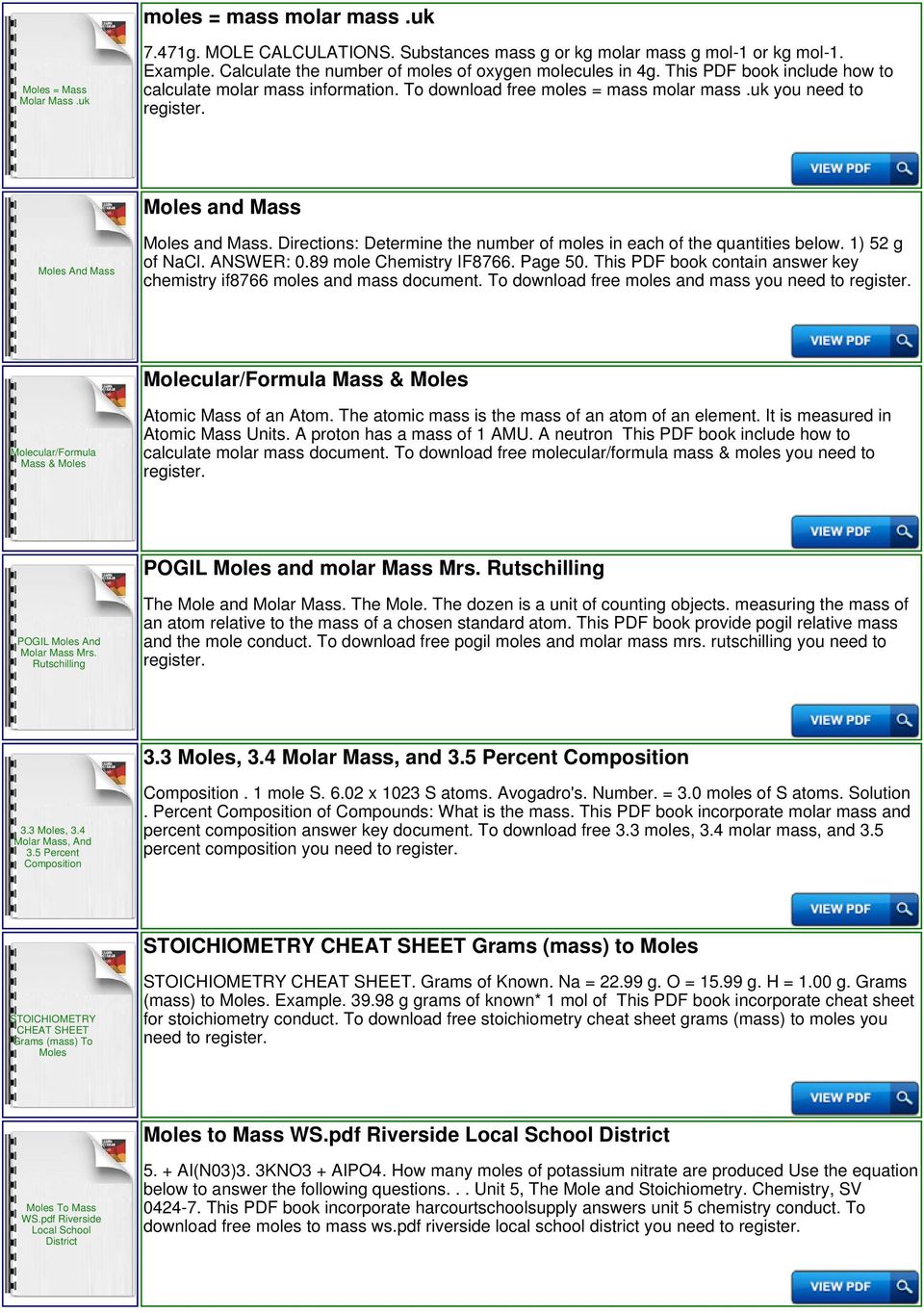
Calculations with Chemical Reactions
Molar mass– the mass of 1 mole of a substance (molecule or atom). The molar mass of carbon monoxide is 28.01 g/mol and the ... Fe : 3 mol CO2. |
|
Chapter 3: Stoichiometry
?Convert grams to moles and moles to grams using molar masses. composition and molecular weight. ... Adjust the coefficient of CO2 to balance C. |
|
Mole & Molar Mass
How many molecules are in 2.50 moles of CO2? 5. Calculate the mass in grams |
|
Chemical Formulae
%20%25.pdf |
|
Selected answers. P. 1 Chapter 1—The Atomic Nature of Matter
5.80 g of dioxane (C4H8O2) is how many moles of dioxane? a. 0.0658 mol How many molecules are present in 36.5 g of CO2 (molar mass = 44.01 g/mol)?. |
|
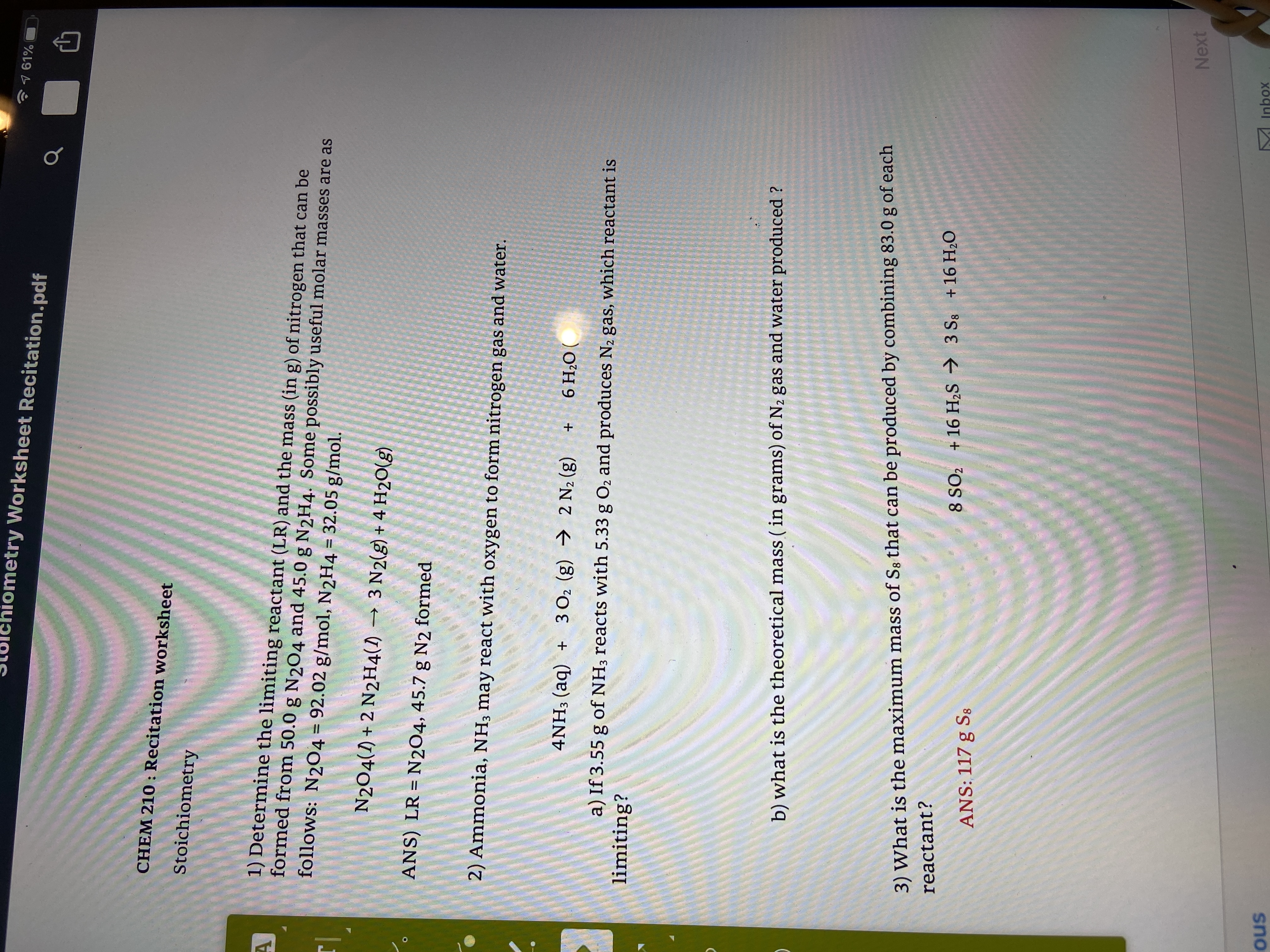
Chemistry 51 ASNWER KEY 1 REVIEW QUESTIONS Chapter 6 1
Mass = 3.82 mol. 166.9 g x. 1 mol. = 638 g (3 sig figs) c). Number of molecules in 1.75 moles of CO2. Molar mass = 12.0 +2(16.2) = 44.0 g/mol. |
|
Problem Set 1
To find the mass of carbon present in the CO2 in grams we need only to multiply the moles of carbon by the molar mass of carbon. Solve. (a) 27 ×109 t CO2 ×. |
|
Physics 2
18.16 Three moles of an ideal gas are in a rigid cubical box with sides of 18.36 The atmosphere of Mars is mostly CO2 (molar mass 44 g/mol) under a. |
|
Answers to worked examples
The molar mass of helium is. He = 4.00 g mol?1 = 4.00 × 10?3 kg mol?1 and of carbon dioxide is. CO2. = [12.01 + (2 × 16.00)] g mol?1 = 44.01 × 10?3 |
|
Untitled
A compound has a molar mass of 90. grams per mole and the empirical formula CH?O. mass = 44 g/mol) present in 11 grams of CO2. x moles = 11 9. 44 g/mol. |
|
Problem Set 1
We can then find the number of moles of carbon dioxide in this mass by dividing by the molar mass of CO2 Because 1 mol of carbon is contained in 1 mol of |
|
Calculating the Molar Volume of Carbon Dioxide
Determine the molar volume of carbon dioxide by measuring the mass and vol- (5) Calculate the number of moles of CO2 in the sample, n, using Equation 5 |
|
Mole Calculations - UTC
Mole abbreviation is mol Molecular and formula weight Suppose we want to make CO2, we burn coal because mainly carbon C + O2 → CO2 Carbon + |
|
Mole & Molar Mass - Montgomery College
How many molecules are in 2 50 moles of CO2? 5 Calculate the mass , in grams , of 0 433 mol of calcium nitrate Hint: Formula Weight of Ca(NO3)2 |
|
Student Academic Learning Services - Durham College
26 juil 2013 · 22 99 g/mol 32 07 g/mol Example: solve for MA of a compound What is the molar mass for each of the following compounds? CO2 H2SO4 |
|
Calculations with Chemical Reactions - George Brown College
Molar mass– the mass of 1 mole of a substance (molecule or atom) 2 Fe(s) + 3 CO2(g) Solution 1: Using the Step-by-Step Proportion Technique uqn: mass |
|
How much volume does 1 kg of CO2 occupy at room temperature
CO2 has a molecular weight of 44 g/mol 1 kg CO2 = 1000 g × (1 mol/44 g) = 22 7 mol CO2 V=nRT/P, V=(22 7)(0 0821)(300)/1 = 559 L CO2 at 27°C (300K), |
|
Download pdf - Commack Schools
A compound has a molar mass of 90 grams per mole and the empirical If 4 mol of CoH1206 is produced by the process, how many moles of CO2 were used? |
|
Carbon dioxide, CO2, is usually a gas It is exhaled by - Cal State LA
In addition, carbon dioxide is an important greenhouse gas due to its ability to absorb many infrared wavelengths of the Sun's light The molecular formula for |
|
Concentrations and Other Units of Measure Unit conversion
[A] = moles of A / volume of air used with CO2: MW = 1x12 + 2x16 = 12 + 32 = 44 grams per mole Mass in grams = Molecular weight x Number of moles |















![WS - Molar Mass - [PDF Document] WS - Molar Mass - [PDF Document]](https://online.fliphtml5.com/nzsh/daqo/files/large/1.jpg)

:max_bytes(150000):strip_icc()/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)













/chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)