chem guide hydrolysis of halogenoalkanes
|
33 Halogenoalkanes chemrevise
Hydrolysis is defined as the splitting of a molecule ( in this case a Ozone Chemistry The naturally occurring ozone (O3) layer in the upper atmosphere is |
|
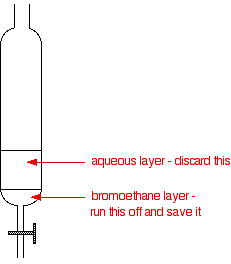
Core practical 4: Investigate the hydrolysis of halogenoalkanes
Learning tips ○ The hydrolysis of halogenoalkanes is a nucleophilic substitution reaction ○ In this investigation the nucleophile is water ○ If NaOH is |
What is the trend in hydrolysis of halogenoalkanes?
Testing for halogenoalkanes
Silver nitrate solution can be used to find out which halogen is present in a suspected halogenoalkane.
The most effective way is to do a substitution reaction which turns the halogen into a halide ion, and then to test for that ion with silver nitrate solution.How do you hydrolysis halogenoalkanes?
Hydrolysis of halogenoalkanes is a chemical reaction where a halogenoalkane (also known as a haloalkane or alkyl halide) reacts with water, resulting in the formation of an alcohol and a halide ion.
What is the hydrolysis reaction of haloalkanes?
Rate of hydrolysis increases down group 7 because the bond enthalpy of the carbon-halogen bond decreases down the group meaning less energy is needed to break this bond.
This means hydrolysis of iodoalkanes will form a precipitate the fastest.
|
Answers HALOGENOALKANES: REACTIONS WITH - Chemguide
HALOGENOALKANES: REACTIONS WITH SILVER NITRATE. SOLUTION. 1. a) You get a substitution reaction in which the halogen atom is displaced as a halide ion which |
|
OCR AS and A Level Chemistry A Delivery Guide - Organic Chemistry
(a) hydrolysis of haloalkanes in a substitution reaction: (i) by aqueous alkali. (ii) by water in the presence of AgNO3 and ethanol to compare |
|
Chemrevise
This guide includes details about the required practicals for A-level chemistry. Comparing the rate of hydrolysis of halogenoalkanes reaction. |
|
Practical Guide EDEXCEL - chemrevise
Practical Guide EDEXCEL chemical being tested may vary between samples. ... Comparing the rate of hydrolysis of halogenoalkanes reaction. |
|
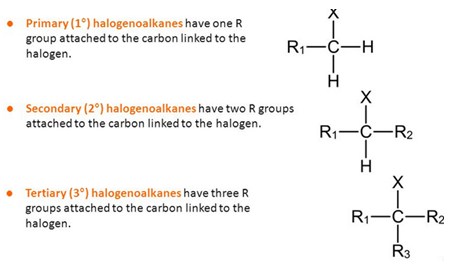
4.2.2 revision guide haloalkanes
N Goalby chemrevise.org Haloalkanes can be classified as primary secondary or tertiary depending on ... Comparing the rate of hydrolysis reactions. |
|
Chemrevise
3 Nov 2018 Comparing the rate of hydrolysis reactions. Water is a poor nucleophile but it can react slowly with halogenoalkanes in a. |
|
Core practical 4: Investigate the hydrolysis of halogenoalkanes
Classify each halogenoalkane as primary secondary or tertiary. Page 5. EDEXCEL. Chemistry. Teacher Resource Pack 1. |
|
Chemrevise
Comparing the rate of hydrolysis reactions. Water is a poor nucleophile but it can react slowly with halogenoalkanes in a substitution reaction. |
|
Mechanism Summary for A-level AQA Chemistry :
Mechanism Summary for A-level AQA Chemistry. Nucleophilic Substitution of Nucleophilic Substitution of Halogenoalkanes ... hydrolysis. KOH aqueous. |
|
Chemistry guide
This guide can be found on the subject page of the online curriculum centre Writing the equation for the substitution reactions of halogenoalkanes with. |
|
Core practical 4: Investigate the hydrolysis of halogenoalkanes
The hydrolysis of halogenoalkanes is a nucleophilic substitution reaction In this investigation the nucleophile is water If NaOH is used to hydrolyse the halogenoalkanes, then any excess NaOH has to be neutralised by HNO3 before adding AgNO3 |
|
Answers HALOGENOALKANES: REACTIONS WITH - Chemguide
Chemguide – answers HALOGENOALKANES: REACTIONS WITH SILVER NITRATE SOLUTION 1 a) You get a substitution reaction in which the halogen |
|
Questions HALOGENOALKANES: INTRODUCTION - Chemguide
The next diagram, also taken from the same Chemguide page, shows the polarity of the various carbon-halogen bonds in a simple series of halogenoalkanes |
|
33 Halogenoalkanes - chemrevise
Comparing the rate of hydrolysis reactions Water is a poor nucleophile but it can react slowly with haloalkanes in a substitution reaction Hydrolysis is defined as |
|
422 revision guide haloalkanes - chemrevise
Haloalkanes can be classified as primary, secondary or tertiary depending on the number of carbon Hydrolysis is defined as the splitting of a molecule ( in this |
|
A Level Chemistry B (Salters) Explaining Observations: rates - OCR
reaction of halogenoalkanes' activity which supports OCR A Level Chemistry B on to investigate the rates of hydrolysis of 1-chlorobutane, 1-bromobutane and |
|
OCR AS and A Level Chemistry A Delivery Guide - Theme: Rates
The hydrolysis of some haloalkanes is believed to take place by a two-step mechanism For example, with 2-bromo-2-methylpropane: Step 1 (CH3)3CBr |
|
Organic Chemistry - SQA
to form alkenes Alkaline hydrolysis of a bromoalkane Experiment on nucleophilic substitution reactions of haloalkanes — See 'Chemistry in Context Laboratory |
|
Haloalkane to alkene chemguide - f-static
Haloalkane to alkene chemguide Continue Hydrolysis of halogenoalcans can give alcohols in the presence of foundation Halogenoalcans (or Halogenoalkanes classes can be classified according to their structure The name is given |
|
Chemistry Year 12 - Ark Globe Academy
3 chemguide co uk 4 physicsandmathstutor co uk Haloalkanes o Revise using the online support (b) M1 hydrolysis For M1 give credit for 'hydration' on |











![Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry](https://cdn.masterorganicchemistry.com/wp-content/uploads/2020/01/1-reaction-map-for-alcohols-alkenes-alcohols-ethers-carbonyls-oxidation-formation-of-alkyl-halides-tosylates-elimination-roadmap.gif)
![Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry](https://img.yumpu.com/55954865/1/500x640/ib-chem-syllabus-2016.jpg)