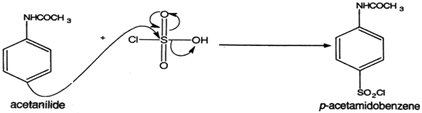
chlorosulfonation of acetanilide mechanism
|
204-210 Research Article Chlorosulfonation of Acetanilide to Obtain
The third step was recrystallization of sulfathiazole to obtain purer product All the identification of samples was done using IR Spectroscopy with FT-IR [4] |
What is the chlorosulfonation reaction?
Chlorosulfonation is introducing the chlorosulfonyl (- SO into organic molecule 2Cl sulfonic acid chloride (R-SO) is formed 2Cl mistake) Journey.It is a kind of substitution reaction, and a hydrogen atom in reactant is changed into chlorosulfonyl after the reaction.
Which chlorosulfonation of acetanilide to obtain an intermediate for the preparation of a sulfa drug?
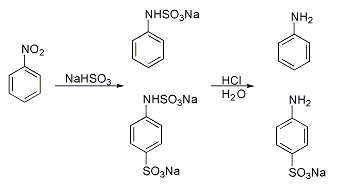
Materials and Methods: Sulfathiazole is prepared by chlorosulfonation of acetanilide (at 114°C) to obtain an intermediate which is reacted with 2-aminothiazole.
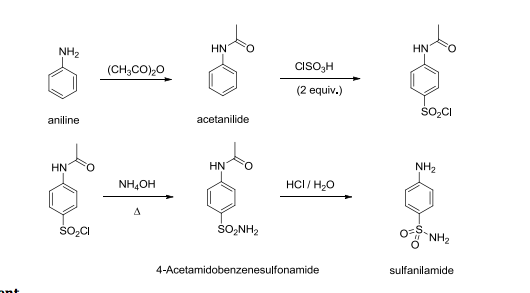
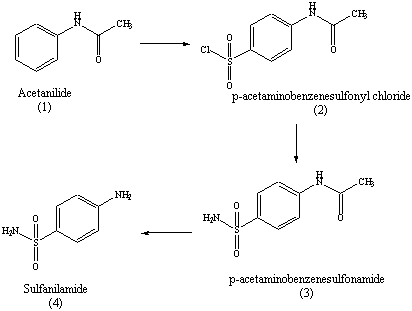
Take Acetanilide as raw material, through chlorosulphonic acid, prepare p-acetaminobenzenesulfonyl chloride, then obtain the acetamido benzsulfamide through ammonolysis reaction, through hydrolysis, recrystallization obtains the finished product sulfanilamide (SN).
|
Chlorosulfonation of Acetanilide to Obtain an Intermediate for the
17 févr. 2017 Conclusions: Among the reaction ratios studied for sulfathiazole sample with 3:1 ratio of p-acetamidobenzenesulfonyl chloride to 2- ... |
|
Chlorosulfonation of Acetanilide to Obtain an Intermediate for the
PROCESS REACTION. Material and methods. Materials and equipment: Acetanilide (Hy-chem labs) Chlorosulfonic acid (Uvsientifics) |
|
REVIEW. CHLOROSULFONATION OF AROMATIC AND HETERO
In the benzene-chlorosulfonic acid reaction the quantity of diphenyl Chlorosulfonic acid is particularly useful for the conversion of acetanilide (30). |
|
THE SYNTHESIS OF SULFA DRUGS
Reaction of chlorosulfonic acid with water: HOSO2Cl + H2O ----> H2SO4 + HCl. Place 6 g of dry acetanilide into a dry 100-mL Erlenmeyer flask. |
|
1. La molécule de benzène. 1.1.1. Formule brute : C 1.1.2
L'acide chlorosulfonique donne un électrophile bien plus puissant : Ainsi la chlorosulfonation de l'acétanilide est-elle une réaction violente : Cette réaction |
|
1. La molécule de benzène. 1.1.1. Formule brute : C 1.1.2
L'acide chlorosulfonique donne un électrophile bien plus puissant : Ainsi la chlorosulfonation de l'acétanilide est-elle une réaction violente : Cette réaction |
|
Sulfa Antibiotics - Synthesis of Sulfanilamide INTORODUCTION
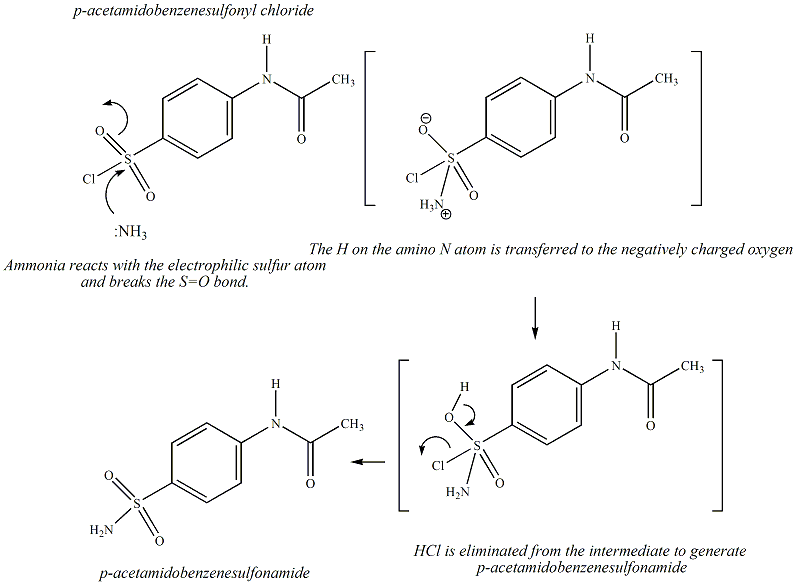
using the electrophilic aromatic substitution reaction known as chlorosulfonation: HN. O. Acetanilide. ClSO3H. HN. O. 4-Acetamidobenzene- sulfonyl chloride. |
|
ORIGINAL PAPER An alternative synthetic process of Ô
Reaction mechanism. Traditional chlorosulfonation process to synthe- size P-ASC from acetanilide actually contains two sequential reactions (Li et al. |
|
M.Sc (ORGANIC CHEMISTRY) LABORATORY MANUAL
HCl to form p-bromo aniline. Reaction: Hydrolysis (deacylation). Chemicals: Para bromo acetanilide – 4gms. Conc HCl-5mL. Ethyl alcohol – 8mL. |
|
Standard Operating Procedure (SOP) for Utilization of Spent
The chlorosulfonation of acetanilide using Chloro sulphonic acid process (i.e. filtration Centrifuge |
|
Chlorosulfonation of Acetanilide to Obtain an Intermediate for the
PROCESS REACTION Material and methods Materials and equipment: Acetanilide (Hy-chem labs), Chlorosulfonic acid (Uvsientifics), P- acetamido benzene |
|
Sulfa Antibiotics - Synthesis of Sulfanilamide - Chemwiscedu
using the electrophilic aromatic substitution reaction known as chlorosulfonation: HN O Acetanilide ClSO3H HN O 4-Acetamidobenzene- sulfonyl chloride S |
|
Honors Cup Synthesis Proposal [231-HC-III]
Quickly add the chlorosulfonic acid and begin stirring the reaction mixture The mixture should darken as the reaction proceeds After the acetanilide is completely |
|
ORIGINAL PAPER An alternative synthetic process - ResearchGate
Reaction mechanism Traditional chlorosulfonation process to synthe- size P- ASC from acetanilide actually contains two sequential reactions (Li et al , 2007; |
|
Synthesis of sulphanilamide from acetamide من ملف
Chlorosulfonation reactions are widely used in organic (in particular, pharmaceutical) mechanism of action) by inhibiting the synthesis of folic acid in bacteria Bacteria synthesize their another series of acetanilide sulfonyl hydrazides and |
|
SYNTHESIS AND MICROBIOLOGICAL STUDY OF NEW
reaction of acetanilide (5gm, 37mmoles) with chlorosulfonic acid (12 5ml, 188 1 mmoles) in 125ml conical flask on an ice-bath for 5-10 minutes to yield p- |





![Honors Cup Synthesis Proposal [231-HC-III] chem215/231-HC-IIIpdf Honors Cup Synthesis Proposal [231-HC-III] chem215/231-HC-IIIpdf](http://www.organicchem.org/oc2web/lab/exp/sulfa/sulfanilamide.gif)