colligative properties definition and examples
What are ligative properties?
The word “colligative” comes from the Latin word colligatus (“bound together”), referring to the way solvent properties are bound to solute concentration in a solution. Ostwald actually proposed three categories of solute properties: Colligative properties are properties that only depend on solute concentration and temperature.
When should the equations for colligative properties be applied?
In practice, this means the equations for colligative properties should only be applied to dilute real solutions when a nonvolatile solute is dissolved in a volatile liquid solvent. For any given solute to solvent mass ratio, any colligative property is inversely proportional to the molar mass of the solute.
What are colligative properties of a dilute solution?
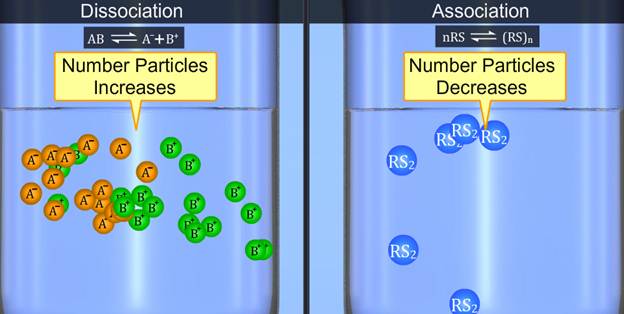
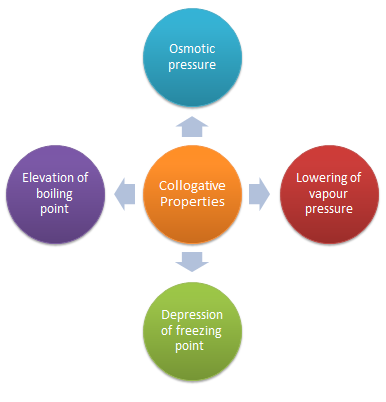
A dilute solution containing non-volatile solute exhibits some properties which depend only on the number of solute particles present and not on the type of solute present. These properties are called colligative properties, and they are mostly seen in dilute solutions.
What are colligative properties?
Definition and Examples Colligative properties depend on the number of solute particles, not their identity. In chemistry, colligative properties are characteristics of chemical solutions that depend on the number of solute particles compared to solvent particles, not on the chemical identity of the solute particles.
|
Properties Of Solutions Chemistry
27 août 2022 Colligative properties – definition and examples and its use in determination of molecular mass. THE PROPERTIES AND STRUCTURE OF MATTER. |
|
Colligative properties of solutions
Definition 2: Osmotic pressure is the pressure of the solute molecules if they were in gaseous state at the same temperature and in the same volume. van 't |
|
CHEMISTRY (862)
(iii) Colligative properties – definition and examples and its use in determination of molecular mass. (a) Relative lowering of vapour pressure: Definition and |
|
Solutions
pressure and colligative properties. We will begin with For example qualitatively we can say that the solution ... 2.3 Define the following terms:. |
|
CHEMISTRY (862)
Azeotropic mixtures – definition and types. (iii) Colligative properties – definition and examples and its use in determination of molecular mass. |
|
ISC Class 12 Chemistry Reduced Syllabus for 2020-21
(iii) Colligative properties – definition and examples and its use in determination of molecular mass. (a) Relative lowering of vapour pressure: Definition |
|
Formulas for Colligative Properties
i = the van't Hoff factor kf = the freezing point depression constant. (a book value CRC reference tables) for the solvent. Example: What is the boiling point |
|
Solutions
pressure and colligative properties. For example if a solution is described by 10% glucose in water ... 2.3 Define the following terms:. |
|
SOLUTIONS & COLLIGATIVE PROPERTIES
Draw vapour pressure vs composition (in terms of mole fraction) diagram for an ideal solution. 4. Define azeotropes with one example of each type. 5. Draw the |
|
The Science of a Sundae: Using the Principle of Colligative
Colligative Properties in Food Science Outreach including definitions which extend to varying depths and examples of concepts. |
|
Lecture 4: Colligative Properties
By definition a colligative property is a solution property (a property of mixtures) for which it is the amount of For example, think about what happens when you |
|
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1
Any of the colligative properties (vapor pressure, freezing point, boiling point or osmotic pressure) can be used to determine the molar mass of nonvolatile molecular substances |
|
Chemistry Colligative Properties Of Solutions Section - Scrumptious
Colligative Properties - Definition, Types, Examples Colligative properties depend only on the number of dissolved particles (that is, the concentration), not |
|
Experiment 1: Colligative Properties - ULM
Background: Colligative properties are properties of a solvent, such as freezing point depression and boiling point Now substituting Eq 3 into the unit definition of molality yields An example of a cooling curve is shown below in Figure 1 30 |
|
Colligative Properties of Foods - Encyclopedia of Life Support Systems
Keywords: Colligative properties, freezing-point depression, boiling point, osmotic This equation combined with Equation (9) generates the definition for ideal entropy as If the solution is described by Raoult's law, for example, the solute is |















/glass-saucepan-on-a-gas-burner-with-boiling-water-dor961844-58fced4e3df78ca159b1f51c.jpg)