colligative properties definition solution
|
Colligative properties of solutions
Definition 2: Osmotic pressure is the pressure of the solute molecules if they were in gaseous state at the same temperature and in the same volume. van 't |
|
Solutions
studying the properties of the solutions like vapour colligative properties (colligative: from Latin: co means together |
|
Solutions
studying the properties of the solutions like vapour colligative properties (colligative: from Latin: co means together |
|
Colligative Properties
Historically colligative properties have been one means In an ideal solution |
|
Solutions and Colligative Properties
Solvation: It is defined as the process of interaction of solvent molecules with solute particles to form aggregates. When water is used as solvent it is |
|
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES. 1. Definitions: (a). Solvent and solute. (b). Solubility and miscibility. (c). Dilute and concentrated solutions. |
|
SOLUTIONS & COLLIGATIVE PROPERTIES
This is because volume depends on temperature and the mass does not. 2. VAPOUR PRESSURE. 2.1 Definition. Vapour pressure of a liquid/solution is the pressure |
|
A Lecture on Colligative Properties in an Undergraduate Curriculum
In pharmaceutical solutions the solute is usually non- volatile and it does not contribute directly to the vapor pressure of the solution. However |
|
Student misconception analysis on the concept of colligative
Colligative Properties of Solutions Using a Digital Three- (7) that there is a misconception due to misunderstanding of the definition of a solution. |
|
The Science of a Sundae: Using the Principle of Colligative
Boiling point is defined as the temperature at which a illustrate how molecules in solution change colligative properties like boiling point. |
|
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1 Definitions: (a) Solvent and solute (b) Solubility and miscibility (c) Dilute and concentrated solutions |
|
Colligative Properties
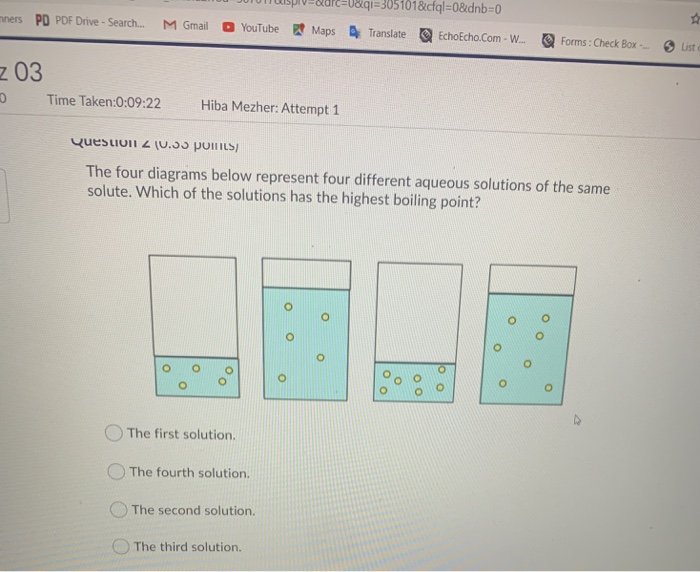
Properties of solutions that depend on the number of molecules present and not on the kind of molecules are called colligative properties These properties include boiling point elevation, freezing point depression, and osmotic pressure |
|
Solutions - NCERT
For example, 10 ethanol solution in water means that 10 mL of ethanol is colligative properties (colligative: from Latin: co means together, ligare means to |
|
Solutions and Colligative Properties
sugar(s)+ H2O( l) →sugar solution Solubility- It is defined as the maximum amount of solute which dissolves completely in given amount of solvent at a constant |
|
Colligative Properties of Foods - Encyclopedia of Life Support Systems
Keywords: Colligative properties, freezing-point depression, boiling point, osmotic A two-component ideal solution can be defined as one in which the total |
|
Colligative Properties ofa Solution - Science
other colligative properties are discussed below Even when the solute molecules dissociate into ions or hydrate, these equations remain valid ifN2 is defined as |
|
Chapter 13 Properties of Solutions
We define the enthalpy change in the solution process as: ∆Hsoln = ∆Hsolute + Colligative properties depend on number of solute particles • There are four |
|
Lecture 4: Colligative Properties
By definition a colligative property is a solution property (a property of mixtures) for which it is the amount of solute dissolved in the solvent matters but the kind of |