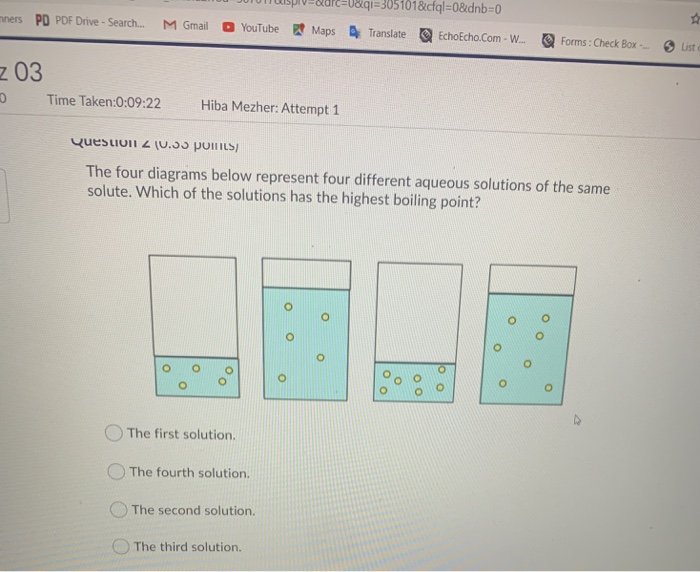
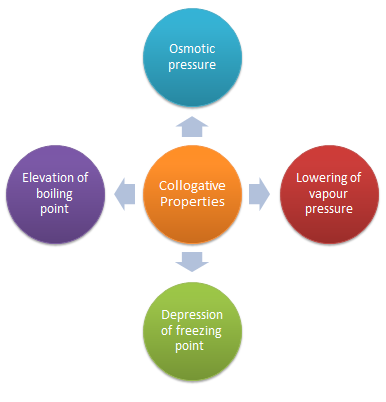
colligative properties lab answers
|
Experiment 1: Colligative Properties
Background: Colligative properties are properties of a solvent such as freezing point depression and boiling point elevation which depend on the concentration of solute particles dissolved in the solvent The decrease in freezing point ΔTf (freezing point depression) for a near ideal solution can be described by the equation: ΔTf = kf · m Eq 1 |
|
Experiment: Colligative Properties Do it at Home Lab
Many salts and solutes will change the properties of the solvent in which they are dissolved This phenomenon is called colligative properties of a solution Most often colligative properties take the form of lowering the freezing point of the solvent or raising the boiling point of the solvent |
What are ligative properties of a solvent?
Background: Colligative properties are properties of a solvent, such as freezing point depression and boiling point elevation, which depend on the concentration of solute particles dissolved in the solvent. The decrease in freezing point, ΔTf (freezing point depression) for a near ideal solution can be described by the equation: ΔTf = kf · m Eq 1
Is osmotic pressure a colligative property?
Osmotic pressure is a colligative property. If two solutions separated by a semipermeable membrane have the same osmotic pressure, no osmosis will occur. Isotonic solutions have same osmotic pressure; solvent passes the membrane at the same rate both ways.

Colligative Properties

Colligative properties lab

13.3 Colligative Properties General Chemistry
|
Experiment 1: Colligative Properties
Background: Colligative properties are properties of a solvent In your lab report briefly discuss the theory behind why the freezing point of a ... |
|
Experiment: Colligative Properties Do it at Home Lab
features of colligative properties is that the change in freezing/boiling points Take a picture of both eggs together and submit with your lab report. |
|
Title: Colligative Properties Lab - ICE CREAM Purpose: To
Period: ______. Title: Colligative Properties Lab - ICE CREAM. Purpose: To investigate the colligative property of freezing point depression through the. |
|
Experiment on Colligative properties Colligative properties are the
The list of colligative properties includes: a) lowering vapor pressure above a solution; b) freezing temperature depression; c) boiling temperature elevation;. |
|
Colligative Properties: Freezing Point Determination
Jun 22 2020 In this at-home experiment |
|
Ice Cream Lab.pdf
Freezing Point Depression and Colligative Properties. Materials In this experiment you will use the lowered freezing ... Answers to Questions. |
|
Molar Mass by Freezing Point Depression
In this experiment you will determine the freezing point of cyclohexane and the The colligative properties are: vapor pressure lowering |
|
Colligative Properties Lab Report Sheet
Colligative Properties Lab Report Sheet. Name: Date: Lab Summary. Colligative properties are those properties of a solution that depend on the number of |
|
Chemistry Investigating Colligative Properties Lab Answers
6 days ago Thank you unquestionably much for downloading chemistry investigating colligative properties lab answers.Most likely you have knowledge that ... |
|
EXPERIMENT 15
Explain how the data support the assertion that freezing point depression is a colligative property. Page 7. EXPERIMENT 15. REPORT SHEET. Name: Date: |
|
Experiment 1: Colligative Properties - ULM
Do not try to pull the thermometer and stirrer from the frozen t-butanol Doing so may break the thermometer Freezing point of solutions: Place the test tube in the |
|
Colligative Properties Lab Report Sheet
Colligative Properties Lab Report Sheet Name: Date: Lab Summary Colligative properties are those properties of a solution that depend on the number of |
|
Title: Colligative Properties Lab - ICE CREAM Purpose: To
Title: Colligative Properties Lab - ICE CREAM Purpose: To investigate the colligative property of freezing point depression through the creation of ice cream |
|
CHM130 Colligative Properties Experiment - classes link 1
Experiment: Determination of Molar Mass by Freezing Point Depression everyday applications of colligative properties save the file for your lab report |
|
Colligative Properties and Freezing-Point Depression - Chemistry
in a laboratory; this week we examine the phenomenon of freezing-point (melting -point) The four colligative properties are freezing-point depression, boiling- point In your lab report, describe the possible sources of error in this experiment, |
|
The Colligative Properties of Solutions - All Bottled up: The Perfect
This discrepant event helps students realize that density is correlated to, but is not the cause of, the changes in the boiling and freezing points This laboratory |
|
Pre Lab: Freezing Point Experiment 1) Define: Colligative properties
Pre Lab: Freezing Point Experiment 1) Define: Colligative properties 2) List four Colligative properties than, or same as) 6 00 °C ? Answer: ______ Explain: |
|
Colligative Properties of Solutions: A Study of Boiling Point Elevation
OBJECTIVES: In this experiment, you will: a) Determine the boiling point elevation constant for water and the molecular weight of an unknown You will do this |
|
Colligative Properties: Freezing Point Depression
15 nov 2004 · CH3511: PHYSICAL CHEMISTRY LAB I Lab 6: Colligative Properties: Freezing Point Depression PRELIMINARY REPORT November 15 |