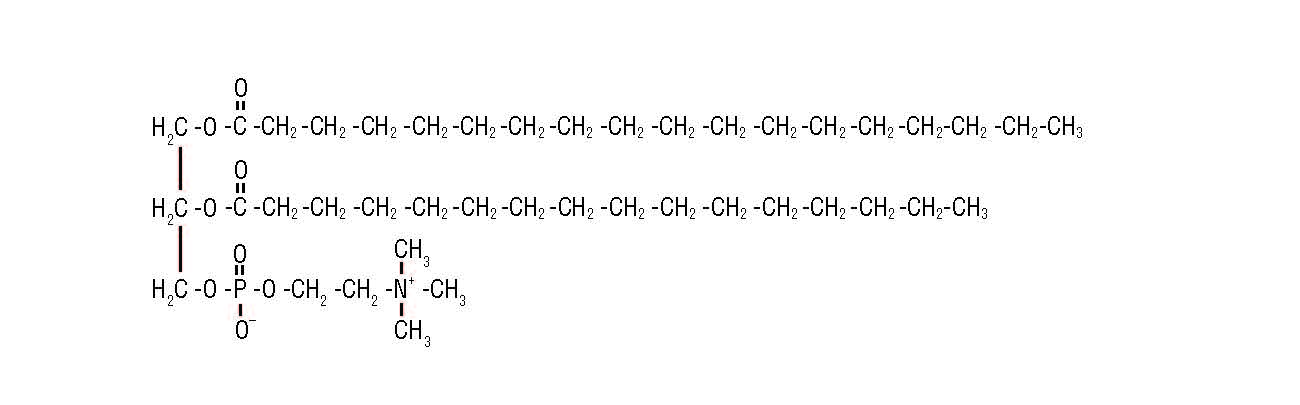
colligative properties making ice cream lab answers
|
Lab 24: Colligative Properties and Ice Cream
Some colligative properties are: boiling point elevation; freezing point depression; osmotic pressure; and vapor pressure depression In this lab we will utilize the colligative property of freezing point depression in order to make a tiny tasty treat – ice cream! Learning Objectives: Observe the effect of adding salt to freezing water |
|
Laboratory Exercise Ice Cream
In this exercise we will take advantage of one of the colligative properties of solutions freezing point depression to make homemade ice cream Then we will evaluate another property of ice cream taste to evaluate how changes to the ingredients used to make ice cream impact flavor |
|
Make Ice Cream in a Baggie
point is referred to as a colligative property In this experiment you will use the lowered freezing point of water to chill another mixture (ice cream) to the solid state Materials Subsitutions 1 quart Ziploc™ bag 1 gallon Ziploc™ bag 1/2 cup milk 1/2 cup whipping cream 1/4 cup sugar 1/4 teaspoon vanilla flavoring |
|
Experiment: Colligative Properties Do it at Home Lab
Thomas Cahill and John Wang Objective: The objective of this experiment is to determine the freezing point depression and boiling point elevation of water by addition of sodium chloride and to explore the osmosis process pass through the membrane of deshelled un-cooked eggs |
How do I know if my Deli Creamer is colligative?
Remove the creamer and, without opening it, check for firmness either by squeezing the sides or shaking the cup near your ear. If you feel or hear liquid sloshing, place the creamer back in the deli cup and swirl for 5 more minutes. Open and enjoy! Freezing-point depression is an example of a colligative property.
Is Creamer a colligative property?
Enjoy this tasty application of a colligative property. The creamer itself consists of water as a solvent and contains particles of various types. While its own freezing point is not depressed to the extent of the ice-water-salt mix, it is depressed.
What are colligative properties of a solution?
The colligative properties of a solution depend on only the total number of dissolved particles in solution, not on their chemical identity. Colligative properties include vapor pressure, boiling point, freezing point, and osmotic pressure.
What are colligative properties of ice cream?
Question: ACTIVITY REPORT: ICE CREAM MAKING Colligative properties are properties of solutions that depend on the number of solute particles dissolved in the solution. There are four colligative properties of solutions, namely, vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
Preparation and Procedure
Pour the 50 g of sodium chloride (salt) into the deli cup. If you do not have a scale, simply pour a layer of salt from 0.5 to 1 cm deep.Place the unopened creamer cup in the deli cup.Fill the deli cup with iceFill the deli cup with water, leaving just enough room to attach the lid. knowledge.carolina.com
Conclusion
Freezing-point depression is an example of a colligative property. A colligative property of a solution is one that depends on the number of solute particles but not on the solute’s identity. For example, the freezing point of an aqueous 1 molal sodium chloride solution is the same as the freezing point of a 1 molal potassium chloride solution beca
Extension
The creamer itself consists of water as a solvent and contains particles of various types. While its own freezing point is not depressed to the extent of the ice-water-salt mix, it is depressed. Your students may enjoy developing experiments to find the freezing point of the creamer or to compare the freezing point among creamers with different ing

13.3 Colligative Properties General Chemistry

Practice Problem: Colligative Properties

Colligative Properties Chemistry Matters
|
Title: Colligative Properties Lab - ICE CREAM Purpose: To
This makes the ice colder than it was before which is how your ice cream freezes. Ideally |
|
Ice Cream Lab.pdf
D.. Your Guide to Chemistry. Freezing Point Depression and Colligative Properties. Materials. 1/2 cup milk. 1/2 cup whipping cream |
|
Ice Cream Lab & Application Questions
Overview. Have you ever wondered what it is about throwing salt on ice that makes it melt? And just why does it melt? Where does the heat come from to melt |
|
Using the Principle of Colligative Properties in Food Science
nents of an ice cream sundae and how the colligative properties Depending on the instructor and their familiarity with the ice cream making process an online. |
|
COLLIGATIVE PROPERTIES ICE CREAM LAB
Colligative Properties: Physical properties determined by the concentration of dissolved particles in a mixture and not affected by the type of dissolved |
|
Chem 1B Objective 4: Predict solution properties based on
Predict solution properties based on colligative properties. Key Ideas: Important in making ice cream melting ice |
|
Experiment: Colligative Properties Do it at Home Lab
This is also the same trick used in making home-made ice cream since ice cream solidifies below 0°C. Explain your answer. (1 pt). 3. Arrange the aqueous ... |
|
HNR Chemistry 1AB 1/12/99 17.3.1 Newport-Mesa Unified School
Ene 12 1999 and answer questions involving concepts of periodic properties ... • Make ice cream. • Colligative properties laboratory Effects of Antifreeze on ... |
|
Colligative Properties: Freezing Point Determination
Hun 22 2020 After the experiment |
|
KAP Chemistry Syllabus Westerville Central High School 2008-09
Laboratory Exercises: The students will make ice cream and determining the Kb value for water. The method is set up as an inquiry in order for the student |
|
Title: Colligative Properties Lab - ICE CREAM Purpose: To
Title: Colligative Properties Lab - ICE CREAM. Purpose: To investigate the colligative property of freezing point depression through the creation of ice |
|
Colligative Properties: Freezing Point Determination
Jun 22 2020 After the experiment |
|
Experiment: Colligative Properties Do it at Home Lab
same trick used in making home-made ice cream since ice cream solidifies below 0°C. Many salts and solutes will change the properties of the solvent in |
|
Untitled
chemistry that helps explains the making of ice cream is thermodynamics. Again what you know about colligative properties provides the answer: you make ... |
|
Biology - Ice Cream . . . & Chemistry Un
May 25 2020 Freezing point depression is a colligative property |
|
Chem 1B Objective 4: Predict solution properties based on
Important in making ice cream melting ice |
|
Chemistry of Ice-Cream Making: Lowering the Freezing Point of Water
Sep 6 2013 We care about molality because freezing point depression is a colligative property |
|
Untitled
the dissolved solute particles are called colligative properties. ice and water mixture is used to cool milk and cream to make homemade ice cream. |
|
Freezing Point Depression: Determining CaCl2 Vant Hoff Factor ?T
solute particles and not their specific type are known as colligative ice cream maker (figure at right) consists of two containers: the inner steel ... |
|
Chemistry - The Science of Ice Cream Week 05/25/20 Reading
May 25 2020 Homemade Ice Cream Directions. Writing: ... Answer the writing prompt at the end of the article. ... if you have ever made ice cream |
|
Title: Colligative Properties Lab - ICE CREAM Purpose: To
Title: Colligative Properties Lab - ICE CREAM Purpose: To investigate the colligative property of freezing point depression through the creation of ice cream |
|
Make Ice Cream in a Baggie
Freezing Point Depression and Colligative Properties Countertop Chemistry Experiment 15 Ice Cream Adding a solute to a solvent Answers to Questions tub with a mixture of one (1) part Morton® Rock Salt for Making Ice Cream and |
|
Freezing Point Depression and Colligative Properties
This makes the ice colder than it was before, which is how your ice cream freezes When you use ice to cool the ingredients for ice cream, the energy is absorbed |
|
Ice Cream - University of San Diego Home Pages
How does understanding colligative properties help us with making ice cream? In this experiment we will taste test a series of vanilla ice cream products that The questions you have just answered should help you think about the different |
|
Ice Cream Lab & Application Questions - Thirteen
Have you ever wondered what it is about throwing salt on ice that makes it melt? And just why does it melt? Where does the heat come from to melt the water? Why |
|
Ice Creamer Lab Answers
27 déc 2020 · chemistry alliance chemistry, ice cream lab example yahoo answers, chemistry of ice cream making lowering the freezing point, colligative properties ice cream |
|
Freezing point depression - University of Waterloo
Freezing point depression: Making ice cream in a Ziploc® bag Gil Abisdris Freezing point depression is one of four colligative properties of water, the others being boiling should carry this out in the classroom, not the lab ) 1 Place the |
|
Ice Cream Lab Answers
LAB LAB REPORT ICE CREAM SCRIBD COLLIGATIVE PROPERTIES MAKING ICE CREAM CHEMISTRY OF ICE CREAM MAKING LOWERING THE |
|
Ice Cream Lab Answers
Colligative Properties Making Ice Cream Freezing Point Depression Ice Cream Lab Answers pehawe de mandipayne Ice Cream Lab Countertop Chemistry |
























/glass-saucepan-on-a-gas-burner-with-boiling-water-dor961844-58fced4e3df78ca159b1f51c.jpg)