colligative properties of electrolytes pdf
|
Colligative Properties
If colligative properties depend on the amount of the solute in the solvent then the equations defining them must include a concentration term and sure enough they do Over the next few pages you will be introduced to the equations in the context of the specific properties but for now simply note the similarities in structure for the equat |
|
CHAPTER 14 Solutions
Colligative Properties and Dissociation of Electrolytes • Electrolytes have larger effects on boiling point elevation and freezing point depression than nonelectrolytes –This is because the number of particles released in solution is greater for electrolytes • One mole of NaCl dissolves in water to produce two moles of aqueous ions: |
|
Colligative Properties of Solutions
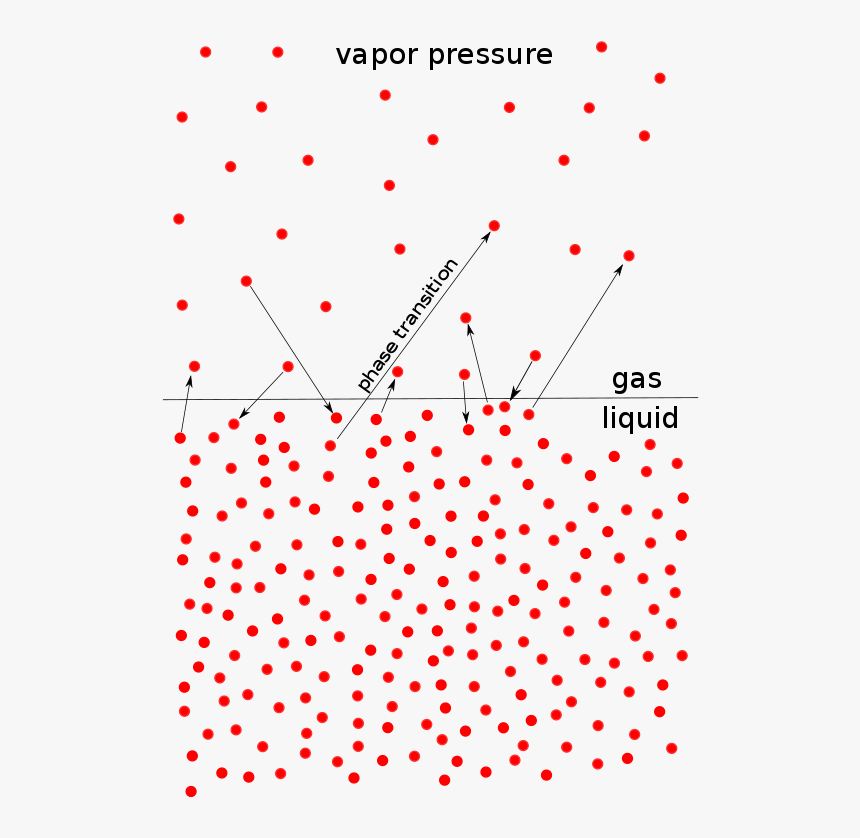
Liquids solutions experience the following four colligative properties: Vapor Pressure Reduction Boiling Point Elevation Freezing Point Depression and Osmotic Pressure over that of the pure solvent Top View Vapor Pressure Reduction Side View Pure Solvent Solvent and Non-Volatile Solute Vapor Pressure Reduction |

Colligative Properties of Electrolyte Solutions

Molality and Colligative Properties

13.3 Colligative Properties General Chemistry
|
Colligative properties of solutions
1 févr. 2021 Boiling-point elevation. ? Freezing-point depression. ? Osmosis. ? Colligative properties of electrolyte solutions ... |
|
ACTIVITY COEFFICIENTS AND COLLIGATIVE PROPERTIES OF
ACTIVITY COEFFICIENTS AND COLLIGATIVE PROPERTIES OF. ELECTROLYTES. By Herbert S. Harneo. Received November 9 1921. As a result of considerable study in |
|
Colligative properties of solutions
In solutions of electrolytes number of solute particles increases due to dissociation. KCl ? K+ + Cl. ?. Page 10. Osmotic pressure of. |
|
13.5 Colligative Properties of Solutions
Colligative properties – depend on the Strong electrolytes – dissociate completely (soluble ... Weak electrolytes – dissociate partially (weak acids. |
|
CHAPTER 14 Solutions
7. Determination of Molecular Weight by Freezing. Point Depression or Boiling Point Elevation. 8. Colligative Properties and Dissociation of. Electrolytes. |
|
THE INTERPRETATION OF THE COLLIGATIVE PROPERTIES OF
activity coefficients12 of the components has been most emphasized in the field of strong electrolytes |
|
Electrolyte Solutions Electrolytes are substances that form ions in
Properties of Solutions of Electrolytes. ? Electrolysis: When a direct electric Colligative Properties of Electrolytic Solutions and Concentrated. |
|
No Slide Title
Weak electrolytes provide few ions in solution. • Even in high concentrations solutions of contributes to the colligative properties of the solvent. |
|
Colligative properties of solutions. 1. Importance Colligative
2. Colligative properties of diluted solutions of electrolytes. Isotonic coefficient. Hypo- hyper- and isotonic solutions in medical practice. 3. Role |
|
12.6 Colligative Properties
This is known as reverse osmosis a method for purifying water. Page 6. 12.7 Colligative Properties of Electrolytes. NaCl lowers the freezing point of water by |
|
Colligative Properties of Solutions
7 Determination of Molecular Weight by Freezing Point Depression or Boiling Point Elevation 8 Colligative Properties and Dissociation of Electrolytes 9 |
|
Physical Chemistry Laboratory
Experiment 1 Freezing Point Depression of Electrolytes Revised, 01/25/06 Colligative properties are properties of solutions that depend on the concentrations |
|
CHAPTER 13
acid is always considered a weak electrolyte, even in a 10 M solution—a very concentrated solution Ions in Aqueous Solutions and Colligative Properties 427 |
|
Electrolyte Solutions: Thermodynamics, Crystallization - CORE
18 déc 2017 · Publisher's PDF, also known as Version of record Colligative properties are properties that according to physical chemistry textbooks are |
|
135 Colligative Properties of Solutions - Web Pages
Colligative properties – depend on the Strong electrolytes – dissociate completely (soluble salts, strong Weak electrolytes – dissociate partially (weak acids |
|
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1
Colligative properties of electrolyte solutions ∆Tbp = iKbm and ∆Tfp = iKfm where "i" is called the van't Hoff factor As a solution is made more dilute, the value of |
|
Chapter 13 Properties of Solutions Classification of Matter
Describe what a colligative property is and explain the van't Hoff factor ➢ Calculate the vapor Use colligative properties of solutions to calculate molar masses of solutes ➢ Explain the solutions of electrolytes show greater changes than |
|
COLLIGATIVE PROPERTIES OF IONIC SOLUTIONS
Our immediate interests here are the applications of the foregoing representations of Raou1t's law in determining the degree of ionization of weak electrolytes, and |
|
Freezing-Point Depression
A Simple Picture for Strong Electrolytes 1 Colligative Property: depends only on amount of solute, not activity; all quantities refer to solvent properties |





![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](https://image.slidesharecdn.com/3-solutions-160318182004/95/3-solutions-its-colligative-propertiesphysical-pharmacy-3-638.jpg?cb\u003d1458325676)







