colligative properties of electrolytes ppt
Which molecule is dissolved in an electroyte?
Each molecule that dissolves yields one dissolved solute molecule. The dissolution of an electroyte, however, is not this simple, as illustrated by the two common examples below: dissociation NaCl(s) →Na+(aq)+Cl−(aq) NaCl ( s) → Na + ( a q) + Cl − ( a q)
What is the effective concentration of ions in a dilute solution?
Thus, in extremely dilute solutions, the effective concentrations of the ions (their activities) are essentially equal to the actual concentrations. Note that the van’t Hoff factors for the electrolytes in Table 12.6.1 are for 0.05 m solutions, at which concentration the value of i for NaCl is 1.9, as opposed to an ideal value of 2.
|
PowerPoint Presentation - Chapter 13 Properties of Solutions
e.g. NaCl dissociates to form 2 ion particles; its limiting van?t Hoff factor is 2. Page 43. Colligative Properties of. Electrolytes. However a 1 M solution of |
|
Colloidal dispersion ppt.pdf
Characteristics of dispersed phase: 1.Particle size: a)Colloidal dispersions + electrolytes ? Stable colloids ... viscosity colligative properties. |
|
CHE 1302 – Basic Principles of Modern Chemistry II
01?/02?/2021 Colligative Properties of Electrolyte Solutions. Important concepts from CHE 1301 required ... Also ppm |
|
No Slide Title
Weak electrolytes provide few ions in solution. • Even in high concentrations solutions of contributes to the colligative properties of the solvent. |
|
Chapter 10 Effect of Electrolytes on Chemical Equilibria
The activity of a species is a measure of its effective concentration as determined by colligative properties. If we substitute a. X for [X] in any equilibrium- |
|
Properties of Solutions
Colligative Properties of Electrolyte Solutions. A solute which dissociates into ions has depressions and black from our colligative property formulas. |
|
15 Theory of Dilute Solutions
POINT DEPRESSION. (1) Beckmann's Method. (2) Rast's Camphor Method. COLLIGATIVE PROPERTIES OF. ELECTROLYTES. ABNORMAL MOLECULAR MASSES. OF ELECTROLYTES |
|
K to 12 BASIC EDUCATION CURRICULUM SENIOR HIGH
Solubility. 6. Colligative Properties of Nonelectrolyte and electrolyte solutions properties of solutions solubility |
|
BSc Chemistry
The activity ('a') of an electrolyte is a property that can be measured by experiment e.g.: by measuring colligative properties such as osmotic pressure or |
|
Class - XII Multiple Choice Question Bank [MCQ ] Term – I & Term-II
gases in liquids solid solutions |
|
PowerPoint Presentation - Chapter 13 Properties of Solutions
e g NaCl dissociates to form 2 ion particles; its limiting van‟t Hoff factor is 2 Page 43 Colligative Properties of Electrolytes However, a 1 M solution of |
|
Colligative Properties Of Solutions Ppt File Type - Unhaggle
Chemistry, Class 12, SolutionsSolutions and colligative properties - SlideShareColligative Properties of Electrolyte Solutions by Amber Colligative properties |
|
Colligative Properties Of Solutions Ppt File Type - Unhaggle
Solutions PowerPoint Colligative Properties_ Boiling Electrolyte Solutions Colligative Properties- Page 1 molality ppt - Solutions Colligative Properties of |
|
Lecture 4: Colligative Properties
By definition a colligative property is a solution property (a property of If colligative properties depend on the amount of the solute in the solvent, then the in cells There is an elaborate process by which water and various electrolytes cross |
|
Colligative properties LinkedIn SlideShare - Free eBooks in the
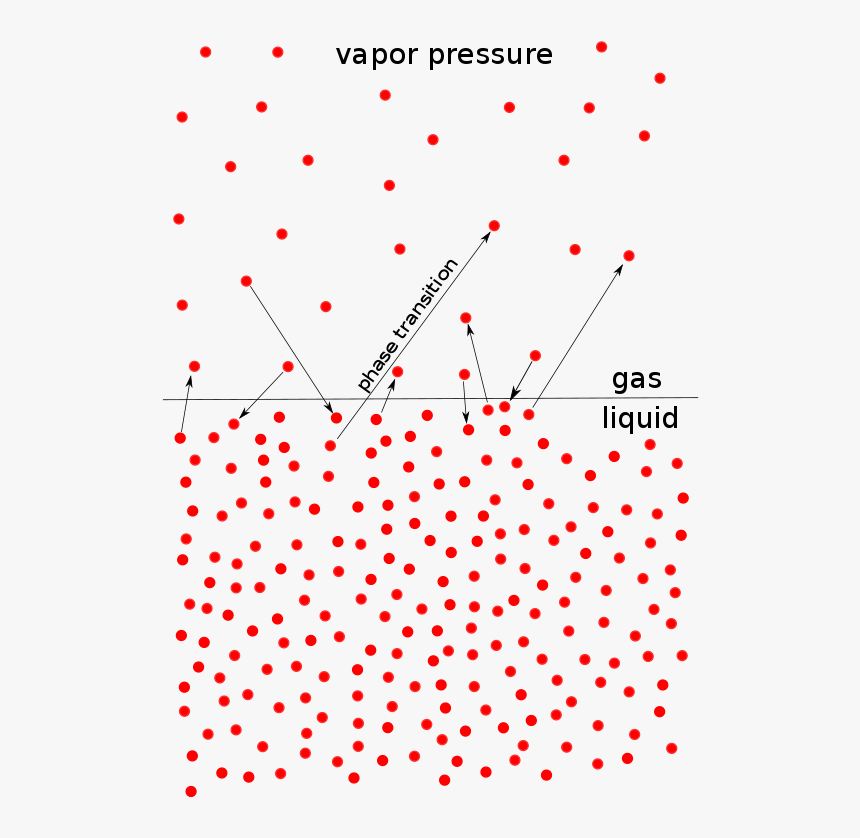
colligative properties, solutions, vapor pressure, boiling and freezing point changes, osmotic pressure Slideshare uses cookies to improve functionality and |
|
Colligative Properties Of Solution Ppt
Properties Of Solution Ppt colligative properties are Vapor pressure lowering Boiling point elevation depression of freezing point of solvent in solution |
|
Chapter 10 Effect of Electrolytes on Chemical Equilibria
The activity of a species is a measure of its effective concentration as determined by colligative properties If we substitute a X for [X] in any equilibrium-constant |





![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](https://image.slidesharecdn.com/3-solutions-160318182004/95/3-solutions-its-colligative-propertiesphysical-pharmacy-3-638.jpg?cb\u003d1458325676)




















![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](http://slideplayer.com/slide/4509159/15/images/1/Colligative+Properties+%28solutions%29.jpg)




![19 152-3-21-2011 colligative properties - [PDF Document] 19 152-3-21-2011 colligative properties - [PDF Document]](https://i1.rgstatic.net/publication/285629114_Protolytic_Equilibrium_Constant_and_Colligative_Properties_of_Mono-_and_Polyelectrolyte_Solutions/links/5ea8914d299bf18b95821f75/largepreview.png)



