colligative properties of nonelectrolyte solutions pdf
|
Colligative Properties
• By definition a colligative property is a solution property (a property of mixtures) for which it is the amount of solute dissolved in the solvent matters but the kind of solute does not matter • Coming to grips with this concept should immediately remind you of kinetic molecular theory of gases—in that case we |
|
Colligative Properties
Colligative Properties 5 1 Introduction Properties of solutions that depend on the number of molecules present and not on the kind of molecules are called colligative properties These properties include boiling point elevation freezing point depression and osmotic pressure Historically colligative properties have been one means |
|
135 Colligative Properties of Solutions Nonvolatile
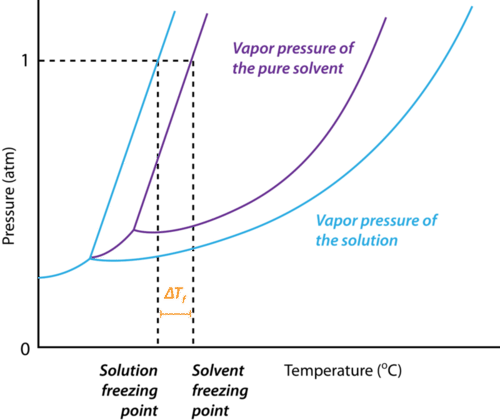
Nonvolatile Nonelectrolyte Solutions – No dissociation; no vapor pressure (glucose sugar ) • Vapor pressure lowering (∆P) – the vapor pressure of the solvent over the solution (Psolv) is always lower than the vapor pressure over the pure solvent (P°solv) at a given temperature Pure solvent Vapor Solution ⇒The solution has a |
|
Colligative Properties of Nonelectrolyte Solutions
Colligative Properties of Nonelectrolyte Solutions Colligative Properties Depends only on number of particles of a solute in solution and not on the nature of the solute Vapor pressure lowering Boiling point elevation Freezing-point depressing Osmotic Pressure Vapor Pressure The pressure exerted by a vapor in equilibrium with its liquid volatile |
Can colligative properties measure the molecular weight of polymers?
In this chapter we discuss using colligative properties to measure the molecular weight of polymers. Because colligative properties depend on the number of molecules, we expect, and will show, that colligative property experiments give a number average molecular weight.

13.3 Colligative Properties General Chemistry

Colligative Properties Explained

Colligative Properties
|
(Nonelectrolytes)
on the number of particles in a solution. The colligative properties of solutions are osmotic pressure vapor pressure lowering |
|
Pre-service Chemistry Teachers Mental Model of Colligative
pre-service chemistry teachers in the colligative properties of non-electrolyte solutions. The study was a descriptive research. |
|
Colligative properties of solutions
1 févr. 2021 Colligative properties of solutions. ? Vapor pressure lowering. ? Boiling-point elevation. ? Freezing-point depression. ? Osmosis. |
|
Developing multiple representations teaching materials assisted by
Abstract. Electrolyte-nonelectrolyte and Colligative properties of solutions are materials which oriented by multiple representation of chemistry. |
|
13.5 Colligative Properties of Solutions
Colligative properties – depend on the Nonvolatile Nonelectrolyte Solutions ... pressure of the solvent over the solution (Psolv). |
|
Using Colligative Properties to Find the Solute Molar Mass Volatile
measuring one of the colligative properties of the solution the mass of the solute and the mass of Volatile Nonelectrolyte Solutions. |
|
General Chemistry
Colligative Properties of Nonelectrolyte Solutions. Vapor-Pressure Lowering. Raoult's law. If the solution contains only one solute:. |
|
General Chemistry
11 avr. 2017 Colligative Properties of Nonelectrolyte Solutions. Vapor-Pressure Lowering. Raoult's law. If the solution contains only one solute:. |
|
GENERAL CHEMISTRY 2
the fundamental concepts on the colligative properties of solutions. solution than weak electrolytes or nonelectrolytes because of the degree or extent ... |
|
Mental Model of Prospective Chemistry Teachers on Electrolyte and
mental models on electrolyte and nonelectrolyte solutions are classified as acid-base and salt theory colligative properties of solutions |
|
Colligative Properties of Solutions
Electrolytes have larger effects on boiling point elevation and freezing point depression than nonelectrolytes One mole of NaCl dissolves in water to produce two |
|
CHAPTER 13
Ions in Aqueous Solutions and Colligative Properties 419 SECTION 1 Nonelectrolyte solutions of the same molality have the same concentration of particles |
|
Chapter 13 Properties of Solutions Classification of Matter
Calculate the concentration of a solution in terms of molarity, molality, mole fraction, percent composition Use colligative properties of solutions to calculate molar masses of solutes ➢ Explain the nonvolatile nonelectrolyte Calculate the |
|
135 Colligative Properties of Solutions - Web Pages
Colligative properties – depend on the concentration of Nonvolatile Nonelectrolyte Solutions pressure of the solvent over the solution (Psolv) is always |
|
Solutions of Nonelectrolytes
In considering the colligative properties of solid-in-liquid solutions, it is ·assumed that the solute is nonvolatile and that the pressure of the vapor above the solution |
|
Solutions - NCERT
studying the properties of the solutions, like vapour pressure and colligative properties A non-volatile, non-electrolyte solid weighing 0 5 g when added to |
|
Physical Chemistry Laboratory
Colligative properties can be described reasonably well by a simple equation for solutions of non-electrolytes The “abnormal” colligative properties of electrolyte |
|
Chapter 7 Mixtures Colligative properties Debye-Hückel
Colligative properties are the properties of dilute solutions that depend only on the number of solute particles present They include: 1 The elevation of boiling |
|
Colligative properties of solutions
1 fév 2021 · Colligative properties of solutions are several important properties that depend If a solute is nonvolatile, vapor pressure of the solution is always less than that by three times as much as a nonelectrolyte solution with the concentration manual / Kh : V N Karazin Kharkiv National University, 2018 196 p |
|
Example Problems on Solutions and Colligative Properties
Solutions and Colligative Properties To be taken up in class or solutions will be posted NA = 6 022 x this solution Sucrose is a non-volatile, non-electrolyte |
![Colligative Properties of Nonelectrolyte Solutions - [PDF Document] Colligative Properties of Nonelectrolyte Solutions - [PDF Document]](https://imgv2-1-f.scribdassets.com/img/document/56212722/149x198/bcbd953555/1306301280?v\u003d1)
![Colligative Properties of Nonelectrolyte Solutions - [PDF Document] Colligative Properties of Nonelectrolyte Solutions - [PDF Document]](https://reader015.docslide.net/reader015/html5/20170819/5571f3ad49795947648e6d32/bg2.png)
![Colligative Properties of Nonelectrolyte Solutions - [PDF Document] Colligative Properties of Nonelectrolyte Solutions - [PDF Document]](https://s3-us-west-2.amazonaws.com/courses-images/wp-content/uploads/sites/1941/2017/05/30162948/osmotic-pressure.png)



![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](https://demo.vdocuments.mx/img/378x509/reader024/reader/2021010409/55cf97c5550346d03393834e/r-2.jpg)
![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](https://imgv2-1-f.scribdassets.com/img/document/290696375/original/fbae378273/1613663930?v\u003d1)