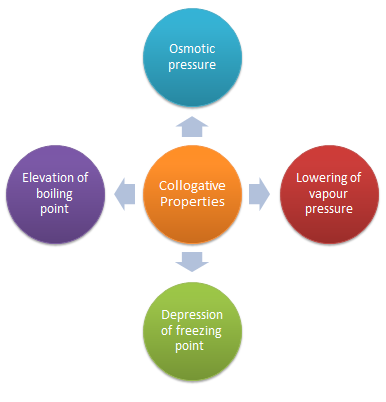
colligative properties of solutions definition
What are anomalous colligative properties?
Anomalous colligative properties are colligative properties that deviate from the ideal colligative behavior. It is quantified by the introduction of the Van't Hoff factor. The boiling points of solutions are higher than that of the pure solvent. This effect is directly proportional to the molality of the solute.
What are colligative properties in chemistry?
In chemistry, colligative properties are those properties of solutions that depend on the ratio of the number of solute particles to the number of solvent particles in a solution, and not on the nature of the chemical species present.

Colligative Properties Explained

Colligative Properties

13.3 Colligative Properties General Chemistry
|
Colligative properties of solutions
Definition 2: Osmotic pressure is the pressure of the solute molecules if they were in gaseous state at the same temperature and in the same volume. van 't |
|
Solutions
studying the properties of the solutions like vapour pressure and colligative (vi) Molarity: Molarity (M) is defined as number of moles of solute. |
|
Solutions
studying the properties of the solutions like vapour pressure and colligative (vi) Molarity: Molarity (M) is defined as number of moles of solute. |
|
Solutions and Colligative Properties
Solvation: It is defined as the process of interaction of solvent molecules with solute particles to form aggregates. When water is used as solvent it is |
|
SOLUTIONS & COLLIGATIVE PROPERTIES
This is because volume depends on temperature and the mass does not. 2. VAPOUR PRESSURE. 2.1 Definition. Vapour pressure of a liquid/solution is the pressure |
|
Colligative Properties Of Solutions Include All The Following Except
Mosby's Dictionary of Medicine Nursing & Health Professions Mosby |
|
THERMODYNAMICS OF SOLUTIONS Solutions quantification
Its definition and relation with other variables |
|
A Lecture on Colligative Properties in an Undergraduate Curriculum
colligative properties using many examples of how to calcu- late the adjustment of tonicity of pharmaceutical solutions through colligative properties. |
|
Colligative Properties Of Solutions Include All The Following Except
Approximately 11000 new and revised definitions reflect the latest developments in health care |
|
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES. 1. Definitions: (a). Solvent and solute. (b). Solubility and miscibility. (c). Dilute and concentrated solutions. |
|
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1 Definitions: (a) Solvent and solute (b) Solubility and miscibility (c) Dilute and concentrated solutions |
|
Colligative Properties
Historically, colligative properties have been one means for determining In an ideal solution, the vapor pressure of the solvent, PA, is reduced from the vapor |
|
Solutions and Colligative Properties
sugar(s)+ H2O( l) →sugar solution Solubility- It is defined as the maximum amount of solute which dissolves completely in given amount of solvent at a constant |
|
Lecture 4: Colligative Properties
By definition a colligative property is a solution property (a property of mixtures) for which it is the amount of solute dissolved in the solvent matters but the kind of |
|
Colligative properties of biological liquids
Colligative properties are properties of solutions that depend on the number of molecules in a given volume of solvent and not on the properties (e g size or mass) of the molecules |
|
Chemistry Colligative Properties Of Solutions Section - Scrumptious
By definition, one of the properties of a solution is a colligative property if it depends only on the ratio of the number of particles of solute and solvent in the solution, |












![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](https://s3.studylib.net/store/data/007302781_1-f23981f39523c797163da6b1a9e96840.png)

