colligative properties of solutions depend on
Do all colligative properties have a common feature?
This indicates that all colligative properties have a common feature, namely that they are related only to the number of solute molecules relative to the number of solvent molecules and not to the nature of the solute. For a given solute-solvent mass ratio, all colligative properties are inversely proportional to solute molar mass.
Why is Raoult's law a colligative property?
Raoult's law suggests that the difference between the vapor pressure of the pure solvent and the solution increases as the mole fraction of the solvent decreases. The change in the vapor pressure that occurs when a solute is added to a solvent is therefore a colligative property.

Molality and Colligative Properties

Colligative Properties Explained

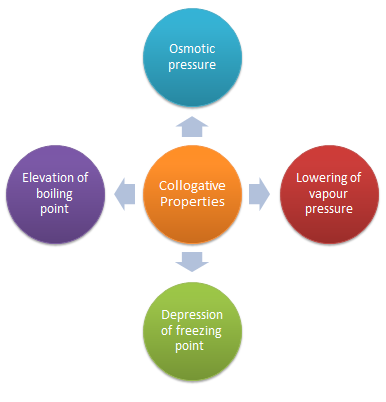
Colligative Properties
|
З А Н Я Т И Е N 9
- a 1 M solution of NaCl or a 1 M solution of CaCl2? Solution: Colligative properties depend on the number of dissolved particles so the solution with the |
|
Physical pharmacy I
Colligative Properties of Solutions. Lowering of Colligative properties of solution include: 1 ... ❖ Colligative properties almost do not depend on the actual. |
|
Colligative properties of solutions
1 февр. 2021 г. always less than that of the pure solvent and depends on the concentration of the solute. The relationship between solution vapor pressure and. |
|
(a) Mole Fraction (b) Isotonic Solution (c) Van
What is meant by colligative property? Ans Those properties of solution which depend upon the number of particles of solute and solvent but not on the |
|
7 SOLUTIONS
The colligative properties of the solutions depend only upon the number of solute particles present in the solution and not on their nature But sometimes while |
|
Molar Mass by Freezing Point Depression
solutions depend on the number of solute particles in the solution and not on the ... These properties are called COLLIGATIVE PROPERTIES because they all depend ... |
|
Physical Properties of Solutions
solution. Colligative Properties of Solutions. Some important properties of solutions depend on the number of solute particles in solution whether these |
|
Chapter 13 Properties of Solutions
Since the colligative properties of electrolytes depend on the number of particles dissolved solutions of electrolytes (which dissociate in solution) should |
|
Colligative Properties: Freezing Point Determination
22 июн. 2020 г. Colligative properties of solutions depend on the quantity of solute dissolved in the solvent rather than the identity of the solute. The ... |
|
Colligative properties of biological liquids
Colligative properties are properties of solutions that depend on the number of molecules in a given volume of solvent and not on the properties (e.g. size |
|
SOLUTIONS
Ans: The colligative properties of solutions depend on the number of solute particles present in solution. Various relations derived for colligative |
|
General Chemistry
Colligative properties (or collective properties) are properties that depend only on the number of solute particles in solution. |
|
Colligative properties of solutions
Colligative properties depend only on applied to the solution to stop osmosis ... ideal gas and a solute in a dilute solution. |
|
Colligative Properties: Freezing Point Determination
22 juin 2020 Colligative properties of solutions depend on the quantity of solute dissolved in the solvent rather than the identity of the solute. The ... |
|
Ion Speciation
29 janv. 2015 Colligative properties of solutions depend on number of ions in solution ... ?can be calculated by equations depend on ionic strength e.g.. |
|
Molar Mass by Freezing Point Depression
Several important properties of solutions depend on the number of solute The colligative properties are: vapor pressure lowering boiling point ... |
|
7 SOLUTIONS
7.7 COLLIGATIVE PROPERTIES. Do you know that there are certain properties of dilute solutions which depend only on the number of particles of solute and not |
|
SOLUTIONS Y
The colligative properties of the solutions depend only upon the number of solute particles present in the solution and not on their nature But sometimes while |
|
Chapter 13: Physical Properties of Solutions
Colligative Properties: Properties that depend only on the number of solute particles in solution and not the nature of the solute. |
|
Colligative Properties
Properties of solutions that depend on the number of molecules present and not on colligative properties to measure the molecular weight of polymers. |
|
SOLUTIONS AND THEIR COLLIGATIVE PROPERTIES 1
Henry's Law - the moderate solubility of gases in a liquid Pgas = kXgas where k is Henry's Law constant for a particular gas and is dependent on temperature and |
|
Solutions and Colligative Properties
Solutions and Colligative Properties There are several solution properties that depend on the relative numbers of solute and solvent particles Several of these |
|
Colligative Properties - Patna Science College
properties of dilute solutions which depend only on number particles of solute present in the solution and not on their identity are called colligative properties |
|
Solutions - NCERT
Their utility or importance in life depends on their pressure and colligative properties Almost all processes in body occur in some kind of liquid solutions |
|
Colligative Properties
Properties of solutions that depend on the number of molecules present and not on the colligative properties to measure the molecular weight of polymers |
|
Colligative properties of biological liquids
Colligative properties are properties of solutions that depend on the number of molecules in a given volume of solvent and not on the properties (e g size or |
|
Colligative Properties of Foods - Encyclopedia of Life Support Systems
Keywords: Colligative properties, freezing-point depression, boiling point, osmotic are interdependent, and since ideal solutions depend on the number of |
|
Chapter 7 Mixtures Colligative properties Debye-Hückel
Colligative properties are the properties of dilute solutions that depend only on the number of solute particles present They include: 1 The elevation of boiling |














![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](https://doubtnut-static.s.llnwi.net/static/q-thumbnail/16290971.png)












