colligative properties of solutions depend on only
How does Raoult's law affect ligative properties?
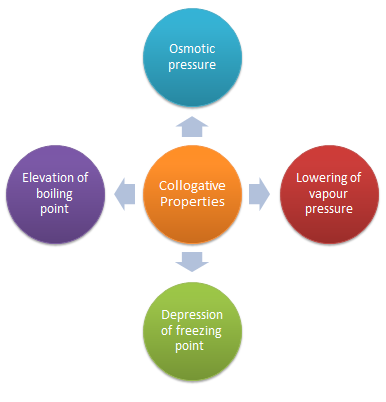
Colligative properties depend only on the number of dissolved particles (that is, the concentration), not their identity. Raoult’s law is concerned with the vapour pressure depression of solutions. The boiling points of solutions are always higher, and the freezing points of solutions are always lower, than those of the pure solvent.

Colligative Properties

13.3 Colligative Properties General Chemistry

11.3 Colligative Properties High School Chemistry
|
Chapter 13: Physical Properties of Solutions
Electrolyte Solutions. Colligative properties depend only on the number of dissolved particles not on the type of particle. Therefore |
|
CHAPTER 13
BIG IDEA. Ionic compounds dissociate in aqueous solutions. Colligative properties of a solution depend only on the number of solute particles present. ONLINE |
|
Molar Mass by Freezing Point Depression
Several important properties of solutions depend on the number of solute particles in the solution and not on These properties are called COLLIGATIVE ... |
|
CHEMISTRY 142 – Example Problems - Solutions and Colligative
Since all colligative properties depend only the number of solute particles the extent of dissociation will affect the magnitude of the freezing point |
|
General Chemistry
that depend only on the number of solute particles in solution and not on the nature of the solute particles. o The colligative properties are. ➢ vapor- |
|
Untitled
Colligative properties are properties of a solution that depend only on the number not the identity |
|
Chapter 11
– Solutions – ions or molecules (small particles). – Colloids – larger particles Colligative properties: depend only on the number of solute particles in ... |
|
Chapter 5: The Thermodynamic Description of Mixtures
Colligative properties. Colligative Property: A solution property that depends only on the number of solute molecules or ions present and not on any specific |
|
Ion Speciation
Colligative properties of solutions depend on number of ions in solution So Kc not really a constant |
|
Chapter 13 Properties of Solutions
Solutions. Colligative Properties. • Changes in colligative properties depend only on the number of solute particles present not on the identity of the |
|
CHAPTER 13
Ionic compounds dissociate in aqueous solutions. Colligative properties of a solution depend only on the number of solute particles present. ONLINE LABS. |
|
Chapter 13: Physical Properties of Solutions
Colligative Properties: Properties that depend only on the number of solute particles in solution and not the nature of the solute. |
|
Molar Mass by Freezing Point Depression
Several important properties of solutions depend on the number of solute The colligative properties are: vapor pressure lowering boiling point ... |
|
General Chemistry
Colligative properties (or collective properties) are properties that depend only on the number of solute particles in solution. |
|
SOLUTIONS Y
of solutions which are dependent only on the number of solute particles. (you will learn explain the significance of colligative properties;. |
|
SECTION 16.1 PROPERTIES OF SOLUTIONS (pages 471–477)
17 Feb 2012 SECTION 16.3 COLLIGATIVE PROPERTIES OF. SOLUTIONS (pages 487–490) ... Properties of a solution that depend only on the number of particles. |
|
Experiment on Colligative properties Colligative properties are the
d) osmotic pressure. These properties depend only on the TOTAL. CONCENTRATION OF ALL THE SOLUTE. PARTICLES IN THE SOLUTION and completely. |
|
Chapter 13 Properties of Solutions
In a solution the solute is dispersed uniformly throughout the solvent. Changes in colligative properties depend only on the number of solute. |
|
Solutions and Their Physical Properties
dissolved solute(s) and a dilute solution has only a small quantity. colligative properties depend only on the number of solute particles in solution. |
|
I. Multiple Choice Questions (Type-I) 1. Which of the following units
NCERT Exemplar Solutions of Class 12 Chemistry Chapter. 2 Solutions. Colligative properties depend on ______. ... (iii) (b) and (c) only. (iv) (c) only. |
|
Colligative Properties - Patna Science College
that partial vapour pressure of a solvent in a solution is equal to the vapour properties of dilute solutions which depend only on number particles of solute |
|
Colligative Properties, Solutions & Distillation Boiling point elevation
depend only on what the solvent is and the the solute concentration ◇ The origin of this behavior is basically the same as the origin of osmotic pressure Entropy |
|
Chapter 7 Mixtures Colligative properties Debye-Hückel
Colligative properties are the properties of dilute solutions that depend only on the number of solute particles present They include: 1 The elevation of boiling |
|
Chemistry Colligative Properties Of Solutions Section - Scrumptious
1 Lowering of Vapour Pressure In a pure solvent, the entire surface is occupied by the molecules of the solvent Colligative properties depend only on the |
|
Solutions - NCERT
Their utility or importance in life depends on their In this Unit we shall consider only binary solutions (i e , colligative property as it depends on the number |
|
CHAPTER 13
BIG IDEA Ionic compounds dissociate in aqueous solutions Colligative properties of a solution depend only on the number of solute particles present ONLINE |
|
Chapter 13 Properties of Solutions - Madison County Schools
Solutions The intermolecular forces between solute and solvent particles must be Changes in colligative properties depend only on the number of solute |
|
Chapter 13: Physical Properties of Solutions
Colligative Properties terminology: Some of the solute will fall out of solution ( as a solid) if the Colligative properties depend only on the number of dissolved |