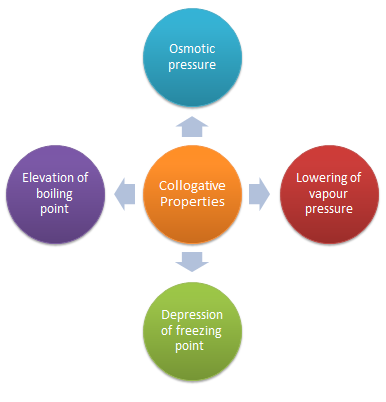
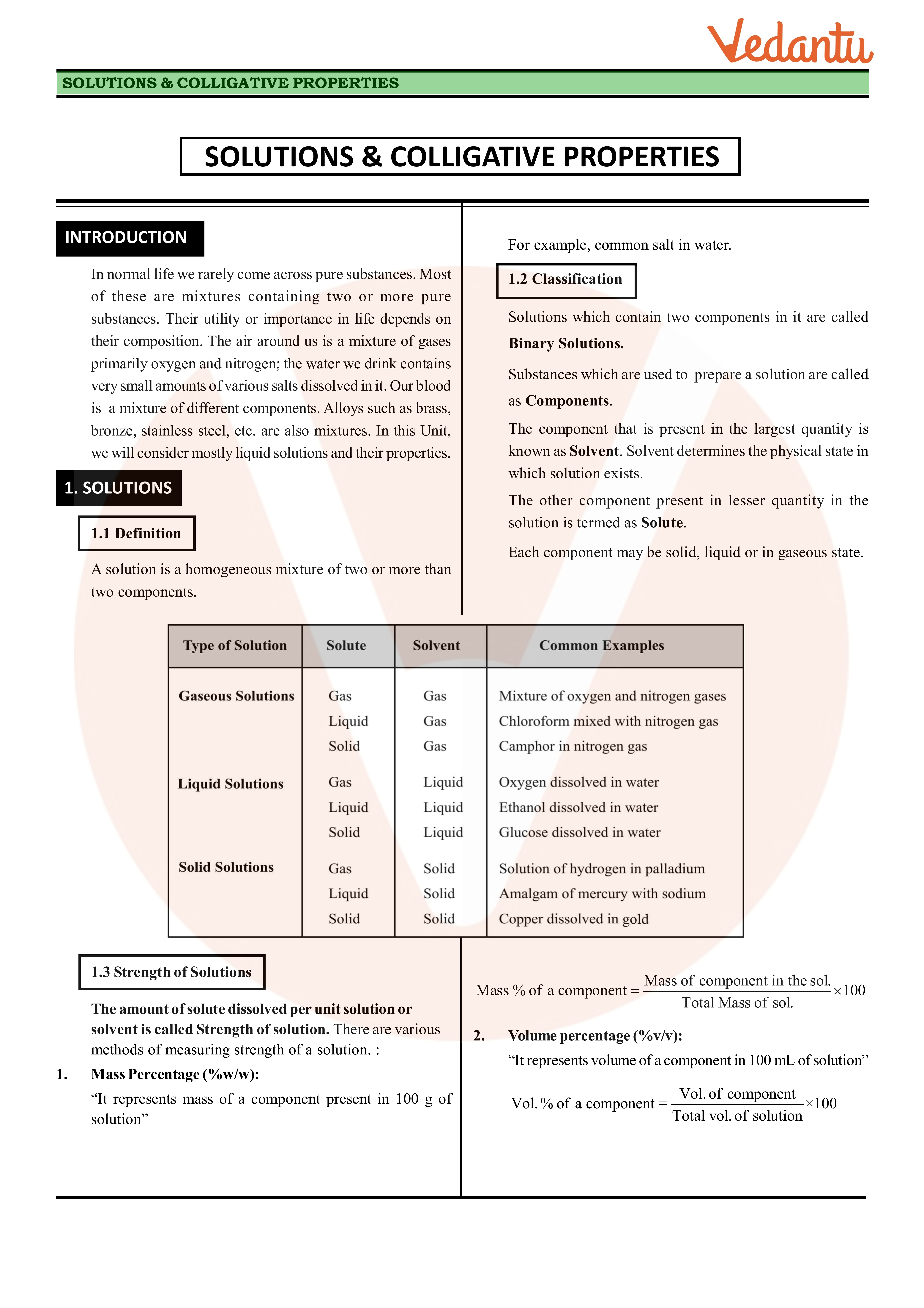
colligative properties definition chemistry
|
Colligative Properties of Foods - Encyclopedia of Life Support Systems
colligative properties (i e , freezing point depression, elevation of boiling point, and A combination of Equations (2) and (3) to define the chemical potential of |
|
Colligative Properties
Historically, colligative properties have been one means A well known result from introductory chemistry is that the boiling point elevation is propor- tional to |
|
Colligative Properties Homework Answer - Uninove
5) The phenomenon of lowering of vapour pressure is defined as: Solutions colligative properties - Chemistry test Colligative Properties Homework Answer This |
|
Experiment 1: Colligative Properties - ULM
Background: Colligative properties are properties of a solvent, such as freezing point depression Now substituting Eq 3 into the unit definition of molality yields |
|
Colligative properties - General Chemistry, Boston University
Increased vapor entropy corresponds to lower vapor pressure, that is, fewer particles in a given volume and so a greater number of arrangements W Examples: |
|
Phase Equilibria and Colligative Properties
The chemical potential of a system is zero at equilibrium To maintain used to develop a broad range of equations, the equations for colligative properties The Gibbs free 9 2 An osmotic pressure defined by the height difference of liquid |
|
AP* Chemistry PROPERTIES OF SOLUTIONS
Colligative Properties— properties that depend on the number of dissolved particles; A solution does not have a sharply defined freezing point, a solvent does |