colligative properties examples in chemistry
What are colligative properties?
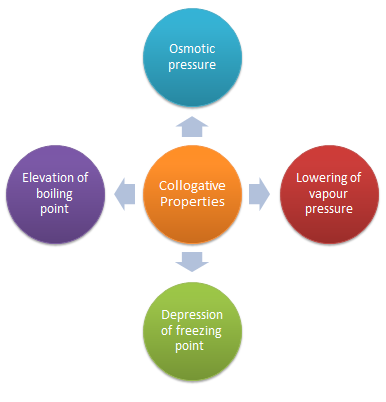
These colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure. This small set of properties is of central importance to many natural phenomena and technological applications, as will be described in this module.
What are the ligative properties of a solvent?
Colligative properties include vapor pressure, boiling point, freezing point, and osmotic pressure. The addition of a nonvolatile solute (one without a measurable vapor pressure) decreases the vapor pressure of the solvent.
When should the equations for colligative properties be applied?
In practice, this means the equations for colligative properties should only be applied to dilute real solutions when a nonvolatile solute is dissolved in a volatile liquid solvent. For any given solute to solvent mass ratio, any colligative property is inversely proportional to the molar mass of the solute.
Are vapor pressure and osmotic pressure colligative properties?
As we will see, the vapor pressure and osmotic pressure of solutions are also colligative properties. When we determine the number of particles in a solution, it is important to remember that not all solutions with the same molarity contain the same concentration of solute particles.
How Colligative Properties Work
Dissolving a solute in a solvent introduces extra particles between solvent molecules. This reduces the concentration of the solvent per unit of volume, essentially diluting the solvent. The effect depends on how many extra particles there are, not their chemical identity. For example, dissolving sodium chloride (NaCl) yields two particles (one sod
The 4 Colligative Properties
Freezing Point Depression Freezing points of solutions are lower than freezing points of pure solvents. The depression of the freezing point is directly proportional to solute molality. Dissolving sugar, salt, alcohol, or any chemical in water lowers the freezing point of the water. Examples of freezing point depression include sprinkling salt on ice to melt it and chilling vodka in a freezer without freezing it. The effect works in other solvents besides water, but the amount of the temperature change varies by solve
Ostwalt and The History of Colligative Properties
Chemist and philosopher Friedrich Wilhelm Ostwald introduced the concept of colligative properties in 1891. The word “colligative” comes from the Latin word colligatus(“bound together”), referring to the way solvent properties are bound to solute concentration in a solution. Ostwald actually proposed three categories of solute properties: 1. Collig
References
Laidler, K.J.; Meiser, J.L. (1982). Physical Chemistry. Benjamin/Cummings. ISBN 978-0618123414.McQuarrie, Donald; et al. (2011). General Chemistry. University Science Books. ISBN 978-1-89138-960-3.Tro, Nivaldo J. (2018). Chemistry: Structure and Properties(2nd ed.). Pearson Education. ISBN 978-0-134-52822-9. sciencenotes.org

Colligative Properties

13.3 Colligative Properties General Chemistry

Colligative Properties Explained
|
Solutions
pressure and colligative properties. For example qualitatively we can say that the solution ... contain 20 g of ethylene glycol and 80 g of water. |
|
Read Free Chemistry Colligative Properties Practice Questions And
You could speedily download this Chemistry Colligative Properties Practice your score · Tons of sample problems and drills with detailed explanations. |
|
Experiment 1: Colligative Properties
In this example the dip is followed by a short plateau in the temperature. This plateau is at the freezing point of the pure solvent as shown in Figure 1. When |
|
Bookmark File PDF Section 163 Colligative Properties Of Solutions
(Free Sample) GO TO Objective NEET Chemistry Guide with DPP & CPP Sheets 9th Edition Disha Experts 2021-10-05 The thoroughly revised & updated 9th Edition |
|
Chemistry Colligative Properties Of Solutions Section Review
Chemistry interview questions and answers PDF download with free sample book covers beginner's questions textbook's study notes to practice worksheets. |
|
CHEMISTRY 142 – Example Problems Solutions and Colligative
CHEMISTRY 142 – Example Problems. Example Problems Solns and Colligatives 2013.doc. Solutions and Colligative Properties. |
|
Download File PDF Section 163 Colligative Properties Of Solutions
MATLAB the book provides examples of its application in both fundamental and developing areas of chemistry |
|
Untitled
CHEMISTRY 142 - Example Problems. = Solutions and Colligative Properties. To be taken up in class or solutions will property of the solvent and a direct. |
|
Colligative Properties Of Solutions Include All The Following Except
studies are all illustrated by many worked examples. measures and activities colligative properties |
|
Bookmark File PDF Section 163 Colligative Properties Of Solutions
(Free Sample) GO TO Objective NEET Chemistry Guide with DPP & CPP Sheets 9th. Edition Disha Experts 2021-10-05 The thoroughly revised & updated 9th Edition of |
|
Colligative Properties of Foods - Encyclopedia of Life Support Systems
Department of Chemistry and Biology and Department of Chemical and Food Keywords: Colligative properties, freezing-point depression, boiling point, osmotic pressure If the solution is described by Raoult's law, for example, the solute is |
|
Experiment 1: Colligative Properties - ULM
To construct a cooling curve one would warm their sample, pure solvent or solution, to well above its melting point, then allow it to cool As the sample cools the |
|
Colligative Properties - Patna Science College
eg When acetone and chloroform are mixed together a hydrogen bond is formed between them which increases inter molecular attraction between them and hence decreases the vapour pressure The dilute solutions show more or less ideal behavior and obey Raoult's law |
|
Gen Chem--Chapter 14 lecture notes - Cal State LA
3 jui 2013 · Colligative Properties Vapor pressure lowering Example: What is the vapor pressure of a saturated NaCl solution at 25 oC? Po = 23 76 Torr |
|
Example Problems on Solutions and Colligative Properties
CHEMISTRY 142 – Example Problems Example Problems Solns and Colligatives 2013 doc Solutions and Colligative Properties To be taken up in class or |
|
Colligative properties of biological liquids
Colligative properties include: lowering of vapor pressure; elevation vapor pressure of each chemical component and the mole substances (for example, red |
|
LECTURE NOTES - VSSUT
K L Kapoor, A Textbook of Physical Chemistry, 2nd Edition, Macmillan, 2011 3 H Kuhn Examples of solutions showing negative deviation: Colligative properties are the properties of only dilute solution which are supposed to behave |
|
Chapter 7 Mixtures Colligative properties Debye - TCD Chemistry
Chemical Thermodynamics : Ge Duesberg Chapter 7 Colligative properties are the properties of dilute solutions that Examples of electrolytes NaCl |
|
Chapter 13 Properties of Solutions Classification of Matter
Use colligative properties of solutions to calculate molar masses of solutes Example At 20 °C the vapor pressure of water is 17 5 torr If we add enough |
|
Colligative Properties Homework Answer - Uninove
Colligative properties are not dependent on the chemical nature of the solution's components Thus Colligative Properties - Definition, Types, Examples |
















![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](https://www.vedantu.com/content-images/revision-notes/cbse-class-12-chemistry-notes-chapter-2-solutions/1.webp)
























/glass-saucepan-on-a-gas-burner-with-boiling-water-dor961844-58fced4e3df78ca159b1f51c.jpg)




