colligative property of water containing rock salt
Is osmotic pressure a colligative property?
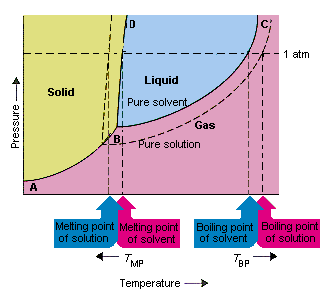
This equation suggests that osmotic pressure is another example of a colligative property, because this pressure depends on the ratio of the number of solute particles to the volume of the solution n / V not the identity of the solute particles. It also reminds us of the magnitude of osmotic pressure.
Why is Raoult's law a colligative property?
Raoult's law suggests that the difference between the vapor pressure of the pure solvent and the solution increases as the mole fraction of the solvent decreases. The change in the vapor pressure that occurs when a solute is added to a solvent is therefore a colligative property.
|
Ice Cream - University of San Diego Home Pages
with salts (NaCl or CaCl2) or brine solutions made of these salt to reduce ice antifreeze Most common antifreeze solutions contain How does understanding colligative properties help us with making ice cream? When you Since ice water only reaches a minimum temperature of 0°C, we take advantage of colligative |
|
Title: Colligative Properties Lab - ICE CREAM Purpose: To
Purpose: To investigate the colligative property of freezing point depression Rinse the quart bag quickly under cold water to remove any excess rock salt 10 |
|
Colligative Properties 2018notebook
11 mar 2018 · Why do you put rock salt down on the roads when its icy? Mar 114:18 PM DEMO ‐ Boiling Point of Water Mar 114:18 PM |
|
Freezing Point Depression
The freezing point depression of a solution is a colligative property of the solution which is rock salt Procedure 1 Place 10 mL of distilled water in an 8 inch test tube (Each solution contains 1 00 g of solute dissolved in 10 0 g of water ) 5 |
|
Molar Mass by Freezing Point Depression
The colligative properties are: vapor pressure lowering, boiling point elevation, water 0 0 1 86 Notice that the freezing point of a substance or a mixture is the temperature at Add approximately ten grams of rock salt and more 6) Immerse the large tube containing the freezing point apparatus into the rock -ice bath |
|
Freezing Point - Learning Outcomes Introduction
Examples of colligative properties include: 1) boiling point elevation 2) When solutes such as salt or glucose are added to water, the freezing point of water * note: As this experiment relies on having an accurate amount of solute dissolved in Prepare a cold water bath by combining water, rock salt, and ice in the |
|
163 Colligative Properties of Solutions
tion to prior knowledge by having stu- solute particles, and not upon their identity is called a colligative property rock salt (NaCl) to a slurry of ice-water |
|
Colligative properties of solutions lab report - Squarespace
These colligative properties include vapour pressure setting, boiling point Rock salt (NaCl), calcium chloride (CaCl2) or a mixture of the two is used to melt ice contains 5 0 g methanol, CH3OH, in water at 37 °C? If a solution is placed in |
|
Chapter 15: Solutions
such as ocean water can contain more than one solute Some combinations of Colligative properties include vapor pressure lowering, boiling point elevation |
|
You will have to write a lab report for each laboratory you do
constant is a property of the solvent, not the solute MATERIALS Erlenmeyer flask ✓ Water ✓ Rock Salt ✓ Ice ✓ Unknown solid METHOD Part One: The |



















/glass-saucepan-on-a-gas-burner-with-boiling-water-dor961844-58fced4e3df78ca159b1f51c.jpg)