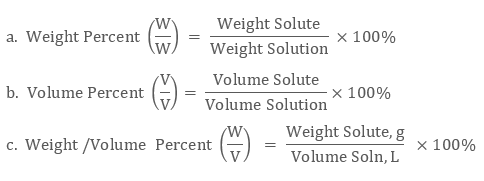concentration of solution formula
What is the concentration of a solution example?
The concentration of the solution tells you how much solute has been dissolved in the solvent.
For example, if you add one teaspoon to two cups of water, the concentration could be reported as 1 t salt per 2 c water.
The vinegar label will report that the solution is 5% by weight acetic acid.What are the 3 types of concentration of solution?
Percent Composition (by mass) Molarity.
Molality.
Mole Fraction.How do you calculate the concentration of a solution?
Step 1: Identify the mass of the solute.
Step 2: Identify the volume of solution.
Step 3: Divide the mass of the solute by the volume of solution to find the concentration of the solution.Molarity (M): Molarity is the notation, where the concentration is expressed in terms of the number of moles of a given solute that is present in 1 litre of the solution.
The formula of molarity is $M=\\dfrac{n}{v}$, where n is the number of moles of the solute and v is the quantity of solution in litres.
|
Beers Law: Determining the Concentration of a Solution
Measure and record the absorbance of the diluted solution. Using your calibration plot (the equation of the line calculate the concentration of Allura Red in |
|
Nsg 231 Calculating IV Solution Concentration
Calculating IV Solution Concentration. The concentration of a solution describes the mass of the solute (amount of drug) divided by the volume of. |
|
AT A GLANCE/ PHARMACY CALCULATIONS INCREASING A
achieve a higher desired concentration.*. Example 1. How much ingredient A should you add to 100mL of a 10% v/v solution to increase it in strength to a 20% |
|
Laboratory Math II: Solutions and Dilutions
The concentration of a solution is how much of the solute is present per unit of volume If molarity or normality the molecular or formula. |
|
Medical Calculations
To calculate the dose in milligrams use the following formula: Example: What is the concentration of a solution if 1g of powder is dissolved in 200. |
|
Laboratory Solution Preparation
PREPARATION OF SIMPLE INORGANIC SALT SOLUTIONS continued on next page. Name / Formula / F.W.. Concentration g/L. Bismuth trichloride. 0.2 M. 63.1 |
|
Concentration of Solutions and Molarity
b) Three moles of sodium chloride dissolved in water produce 6 mol of particles because each formula unit of. NaCl dissociates into two ions. 3 moles Na+ and 3 |
|
UNITS OF CONCENTRATION.pdf
To convert concentrations in mg/L (or ppm in dilute solution) to molarity It has advantages when carrying out titration calculations |
|
1 Calculation of concentration of a solution The study material
Calculation of concentration of a solution The molarity can be calculated either using the formula c = n/V (c = molarity n = substance amount in moles |
|
SOLUTION CONCENTRATIONS
The mass of solution is equal to the mass of solute plus the mass of solvent A solution of 5 0 by mass concentration would contain 5 0 g of solute in 100 0 g of solution (5 0 g of solute and 95 0 g of solvent) Thus, percent by mass gives directly the number of grams of solute in 100 g of solution |
|
Calculations involving concentrations, stoichiometry Metric System
Concentration of a solution • Mass concentration: grams of dissolved substance per liter of solution • Molar concentration: moles of dissolved substance per liter |
|
Volume = Mass / Concentration
Concentration in (w/v) = 100 x (mass solute in g) / (volume solution in mL) Often beginning science students will do calculations writing down only numbers |
|
Beers Law: Determining the Concentration of a Solution
These are your four standard solutions of known molarity 4) Calculations: Determine the actual molarity of your stock solution and all four dilutions based on your |
|
SOLUTIONS, CONCENTRATIONS, STOICHIOMETRY - Cal State LA
The definition is selected so that the formula weight (in amu) and the molar mass (in grams/mol) have the concentration = amount of solute/amount of solution |
|
Chapter 4: Calculations Used in Analytical Chemistry - İYTE
4B-1 Concentration of Solutions The molar concentration c x of a solution of a solute species X is the number of moles of that species that is contained in 1 liter |
|
4 Ways to Express Concentrations in Solutions
Calculations involving Concentrations (Show formula, show work box answers) Percent Composition 1 What is the percent composition of a solution in which |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
The formula below is a quick approach to calculating such dilutions where: V = volume, C = concentration; in whatever units you are working (source solution |
|
Mixing concentrations - Mathcentre
PHARMACY CALCULATIONS MIXING CONCENTRATIONS Calculating the strength of a mixture of two different concentrations Example 1 If you mix 100mL |
|
Solutions - Concentrations
I - Solutions et concentrations 3 1 Exercice : Dilution On désire préparer une solution aqueuse de sulfate de cuivre, de formule CuSO 4 On dispose d'une |