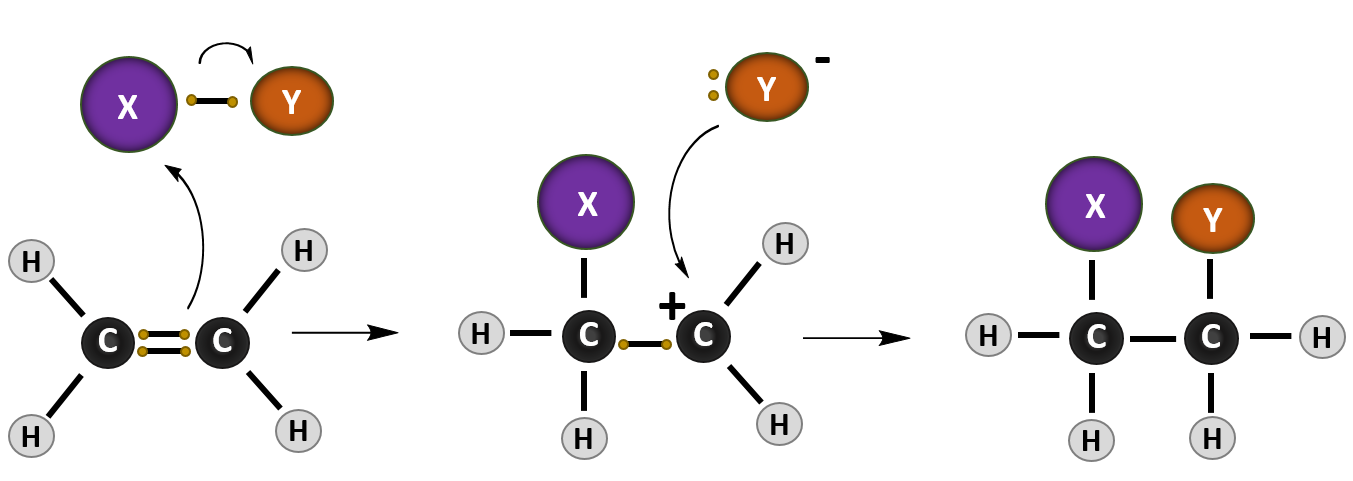
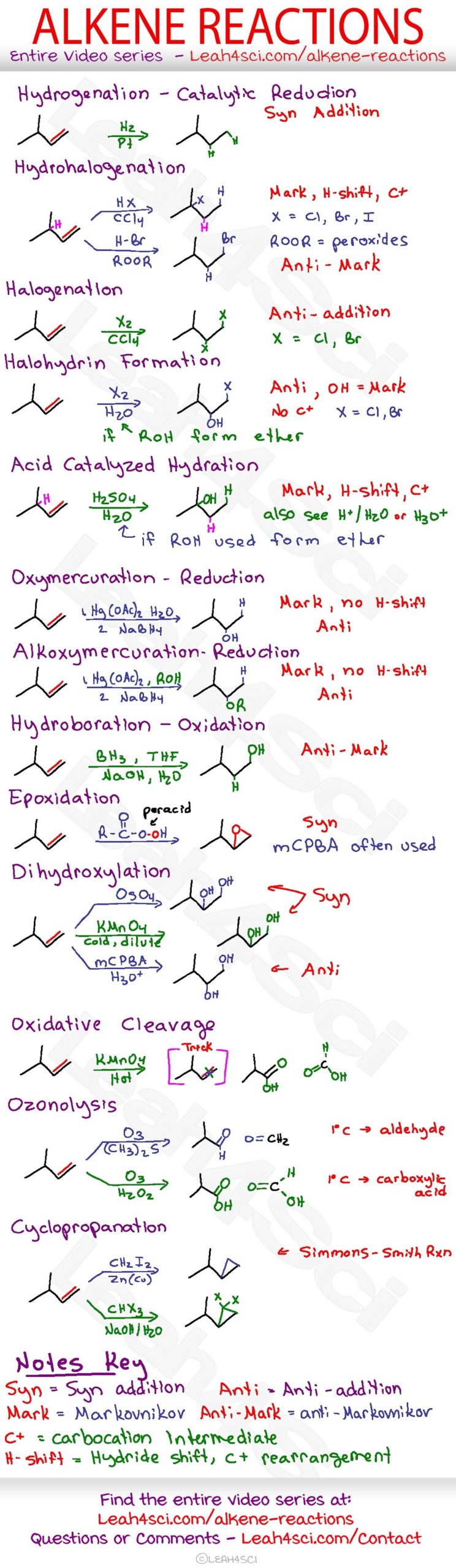
addition of hcl to alkene
What type of reaction is alkene?
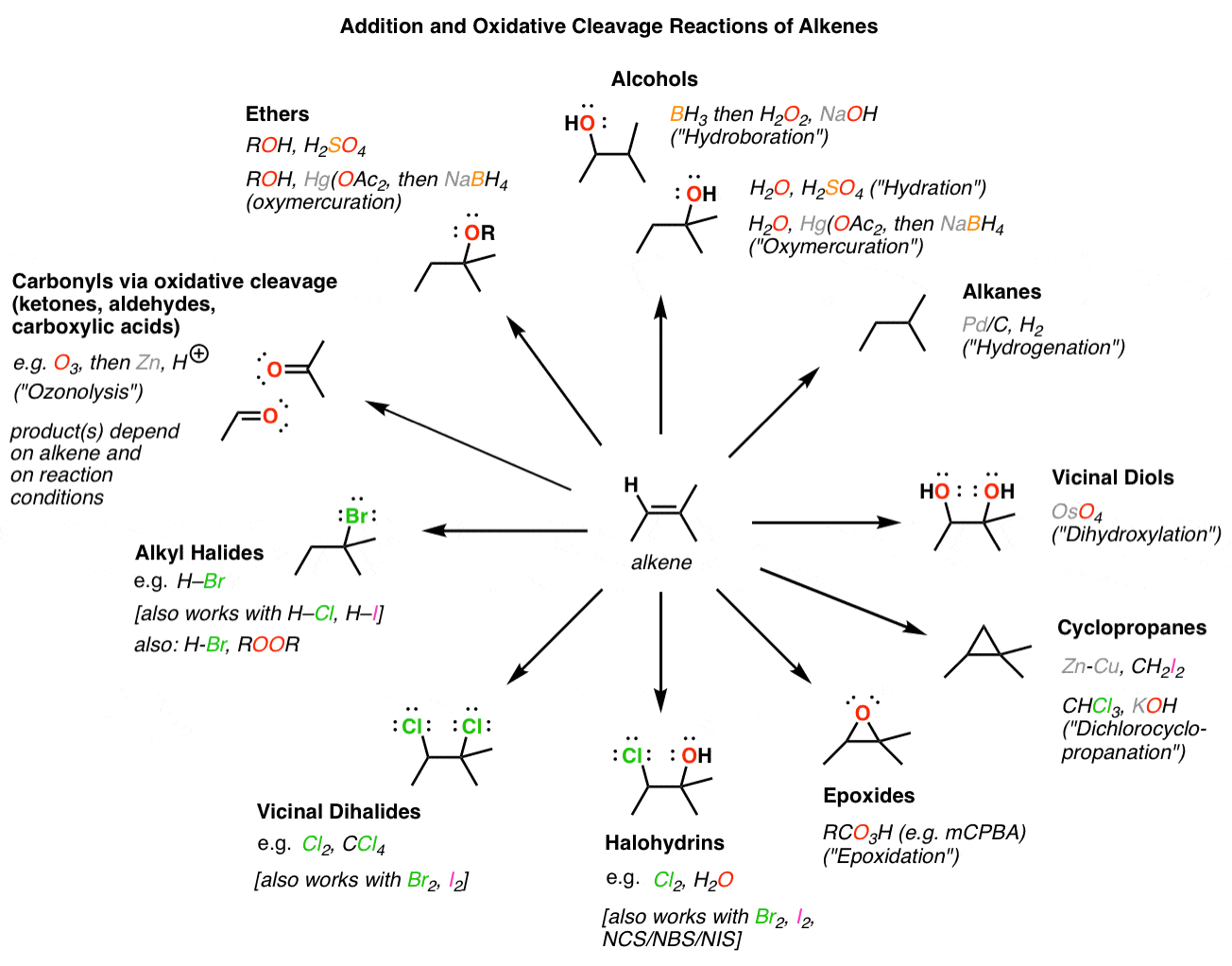
The most common type of reaction for alkene is the addition reaction to a C=C double bond. In addition reaction, a small molecule is added to multiple bonds, and one π bond is converted to two σ bonds (unsaturation degree decreases) as a result of the addition. An addition reaction is the opposite process of elimination.
What happens when halogen is added to an alkene?
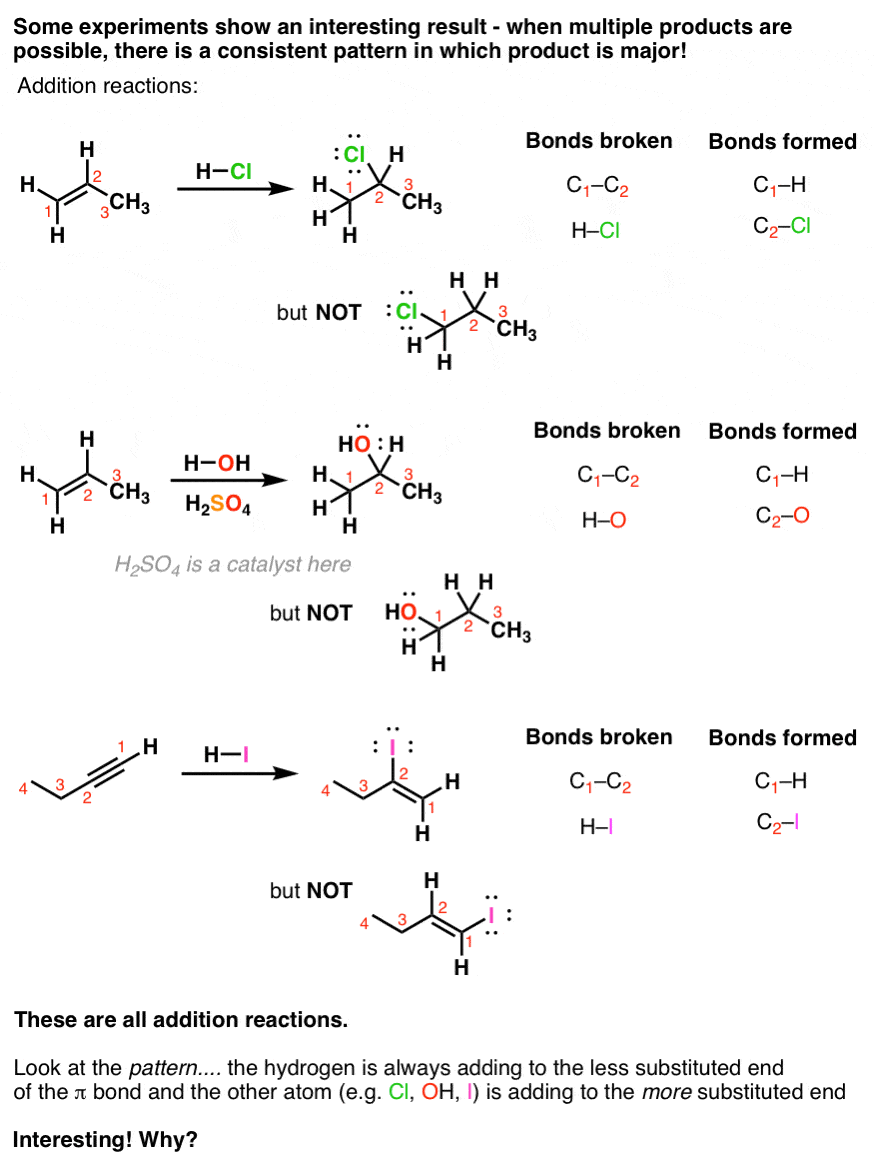
The reaction tends to occur such that the halogen ends up attached to the carbon of the alkene attached to the fewest hydrogen atoms, a phenomenon known as Markovnikov’s Rule. Addition of H-X to alkenes occurs through a protonation of the alkene to give a carbocation intermediate, followed by addition of the halide to the carbocation.
What happens when HCL is added to ethene?
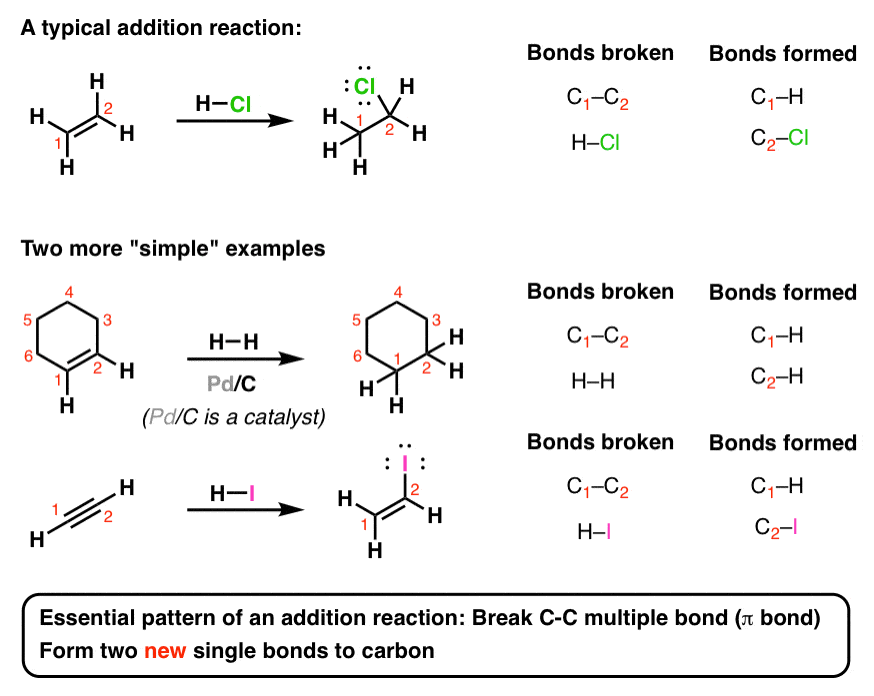
All alkenes undergo addition reactions with the hydrogen halides. A hydrogen atom joins to one of the carbon atoms originally in the double bond, and a halogen atom to the other. For example, with ethene and hydrogen chloride, you get chloroethane: Figure 7.8.1 7.8. 1 Electrophilic addition of HCl to ethene.
What happens when hydrohalic acids are added to alkenes?
When hydrohalic acids (HCl, HBr, HI) are added to alkenes, addition reactions can occur, resulting in formation of a C-H and C-halogen bond and breakage of a C-C pi bond. The reaction tends to occur such that the halogen ends up attached to the carbon of the alkene attached to the fewest hydrogen atoms, a phenomenon known as Markovnikov’s Rule.

Addition of HCl to an Alkene

Addition of HCl to an alkene with a carbocation rearrangement

8.2 Hydrohalogenation of Alkenes Organic Chemistry
|
Mechanisms – Answers a. Addition of HCl or HBr to an alkene
Addition of HCl or HBr to an alkene. Answer: Step 1. The nucleophilic pi bond attacks the proton of H-Cl forming a carbocation on the Markovnikov end (most |
|
The Use of silica and alumina to catalyze the addition of hydrogen
Sept 1 1999 addition of hydrogen chloride to alkenes. Ying Liu ... Hydrogen chloride does not add to alkenes readily unless the alkene is either highly ... |
|
Reactions of Alkenes and Alkynes
Chemists account for the addition of HX to an alkene by a two-step mechanism which we illustrate by the reaction of 2-butene with hydrogen chloride to give 2- |
|
ORGANIC CHEMISTRY I – PRACTICE EXERCISE Alkene reactions
D) addition of HCl. E) A B |
|
Chemistry 210 - Chapter 6 - quiz 2
Which alkene below is thermodynamically the most stable? A. 1-hexene. B. trans-3-hexene Addition of HCl to 3-methyl-1-pentene gives two products. |
|
Free Radical Addition and Transfer Reactions of Hydrogen Chloride
An ab- normal addition of hydrogen chloride to propylene has also been observed but indications are that such additions to most. I-alkenes will be difficult. |
|
Free Radical Addition and Transfer Reactions of Hydrogen Chloride
normal addition of hydrogen chloride to alkenes had failed.1 This result was accounted for on the basis that the energy requirement for step (2) of the. |
|
CHEM 203 Topics Discussed on Sept. 18 Carbocations carbonium
Sept 18 2005 Principle organic reactions |
|
4.7 addition of hydrogen halides to alkenes
addition of HCl because additions of HBr and HI are faster. Hydrogen halide addition to alkenes is a highly regioselective reaction because. |
|
Gas phase addition of hydrogen chloride to propylene
molecular addition of hydrogen chloride to alkenes has too high an activation energyto be observed at room tempera- ture in any reasonable time. |
|
Mechanisms – Answers a Addition of HCl or HBr to an alkene Answer
Addition of HCl or HBr to an alkene Answer: Step 1 The nucleophilic pi bond attacks the proton of H-Cl, forming a carbocation on the Markovnikov end (most |
|
ADDITION Many reagents add to the alkene bond The - SIUE
+ k[alkene][XY] 2 C=C X-Y X-Y Additions of HCl, HBr The ionic addition of HX produces the more stable carbocation which then reacts with the bromide ion |
|
Reactions of Alkenes
Markovnikov's rule: In the addition of HX or H2O to an alkene, H adds to the carbon of the double bond having the greater In addition of HCl to an alkene |
|
Reactions of Alkenes
The alkene abstracts a proton from the HBr, and a carbocation and bromide ion are (HCl and HI perform analogous electrophilic addition reactions) |
|
Reactions of Alkenes and Alkynes
The most characteristic reaction of alkenes is addition to the carbon–carbon The addition of HCl to propene gives 2-chloropropane (isopropyl chloride); |
|
Addition of Hydrogen Halides To Alkenes - Master Organic Chemistry
Acids (HX) include HCl, HBr, and HI • First step is protonation of alkene to give a carbocation, with the pi bond acting as a base • Rearrangements may occur to |
|
Alkenes - Addition Reactions - BITS Pilani
3 Addition of hydrogen halides — C = C — + HX → — C — C — H X a) HX = HI, HBr, HCl b) Markovnikov orientation CH 3 CH=CH 2 + HI → CH |
|
64 Electrophilic Addition of Hydrogen Halides to Alkenes
6 4 Electrophilic Addition of Hydrogen Halides to Alkenes 0°C CH 3 Cl CH 3 (100 ) Markovnikov's Rule Example 3 Example 3 HCl |
|
46 ADDITION REACTIONS OF ALKENES 47 ADDITION OF
In an addition reaction, the carbon–carbon p bond of the alkene and the XLY bond of the reagent addition of HCl because additions of HBr and HI are faster |




![Alkenes PDF - [PDF Document] Alkenes PDF - [PDF Document]](https://reader012.staticloud.net/reader012/html5/20190321/5a71265b7f8b9ab1538c8fb5/bg1.png)


![Alkenes PDF - [PDF Document] Alkenes PDF - [PDF Document]](https://demo.fdocuments.in/img/742x1000/reader018/reader/2020011115/577cd6d11a28ab9e789d5462/r-1.jpg?t\u003d1610367734)
![Alkenes PDF - [PDF Document] Alkenes PDF - [PDF Document]](https://0.academia-photos.com/attachment_thumbnails/55680846/mini_magick20190114-13705-dvf8y0.png?1547462022)