addition polymerization equation
How do you define addition polymerization?
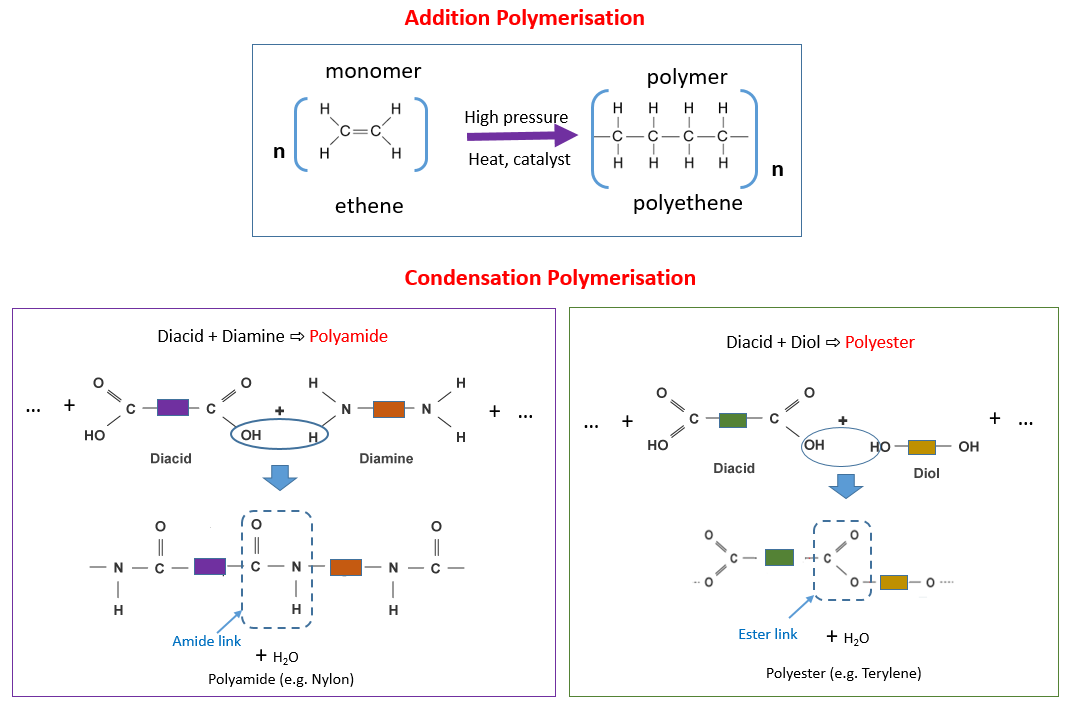
Define addition polymerization. Draw the structure of a polymer from its monomer. Know the uses/applications of common polymers. Addition polymerization and condensation polymerization are two modes of polymerization reactions in the formation of polymers.
How are polymers synthesized?
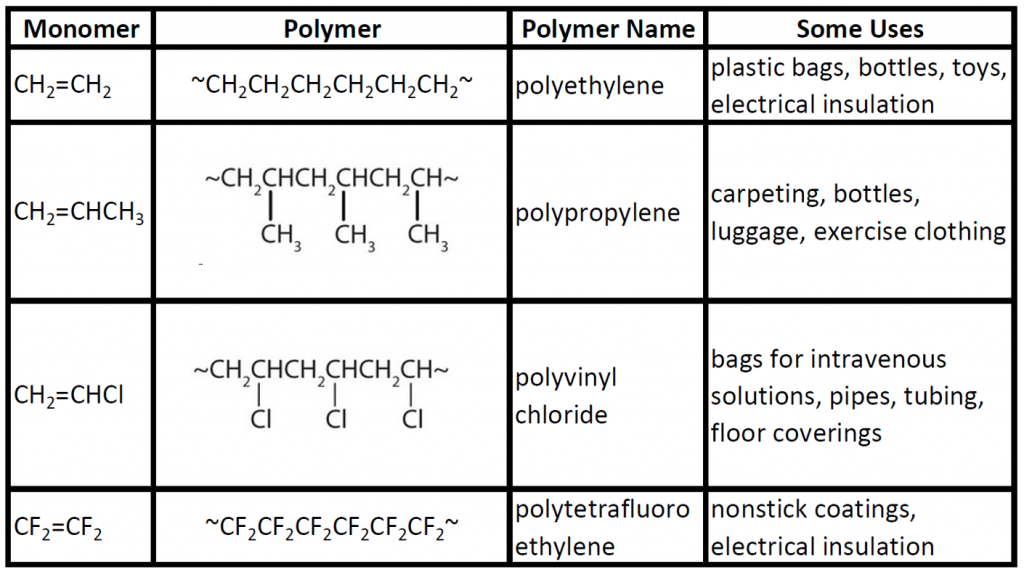
Some of these polymers are synthesized by methods involving initiators other than free radicals - carboanions, carbocations, or coordination compounds, for instance. Vinyl polymers are addition polymers, which have the same atoms as the monomer in their repeat units.
What are the two types of polymerization reactions?
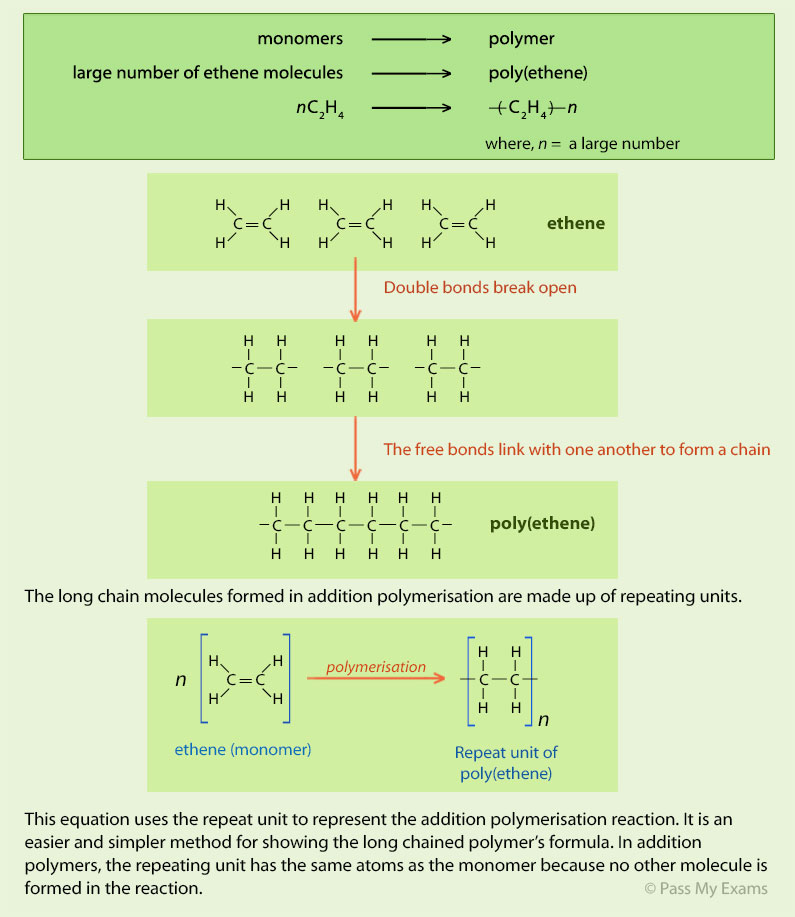
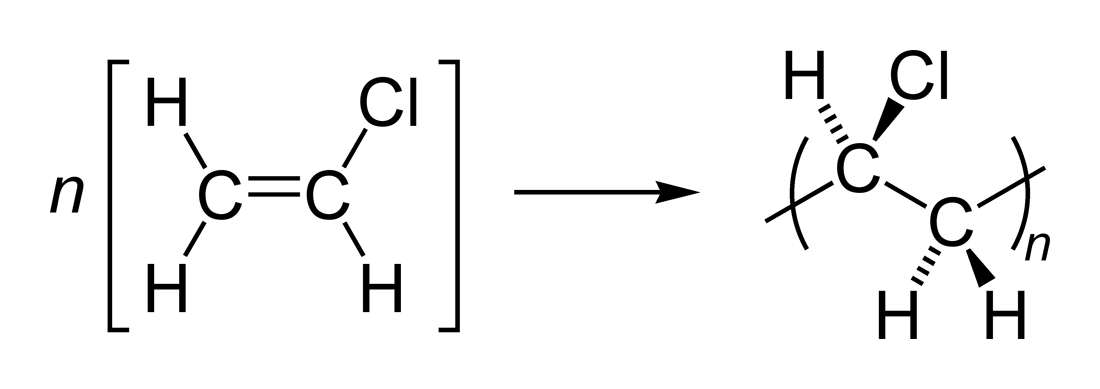
There are two general types of polymerization reactions: addition polymerization and condensation polymerization. In addition polymerization, the monomers add to one another in such a way that the polymer contains all the atoms of the starting monomers. Ethylene molecules are joined together in long chains.

GCSE Chemistry

Simple explanation of addition polymerisation using polythene (polyethene) GCSE

Addition polymerizationpolymer introduction to polymer chemistry class 12
|
Continuous Addition Polymerization
Jul 6 2005 Applicability of Homopolymerization Continuous. Addition Rate Equations to Copolymerizations. 50. 3.3.3. Addition of One Mixed Monomer Feed ... |
|
1 050929 Quiz 1 Introduction to Polymers 1) In class we discussed a
e) What is the value of <R. 2. > (in terms of N and l) using this equation (1)? 1) Vinyl polymers are generally made by addition polymerization. |
|
Alkenes
In addition to the single covalent sigma- bonds which hold the carbon atoms xiii) Write a balanced equation for the addition polymerisation used to form ... |
|
Name__________________________Review Organic Reactions
A) an addition reaction. B) an esterification reaction. C) a polymerization reaction. D) a substitution reaction. 5. Given the balanced equation |
|
Chap 15 Solns
(b) We are now asked to compute the number-average molecular weight. Since the degree of polymerization is 26000 |
|
Chemrevise
The simplest addition polymer is made from ethene and is called poly(ethene). Modern naming of polymers Polymerisation equations are written in the form. |
|
Pearson Edexcel International GCSE in Chemistry (9-1)
(iv) Equation 2 shows an example of an addition reaction. Polymers can be classified as addition polymers or condensation polymers. |
|
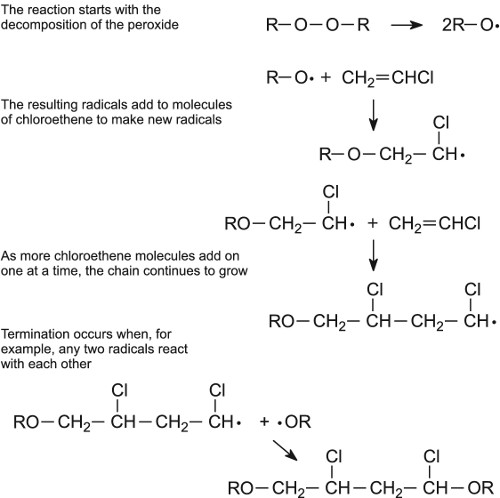
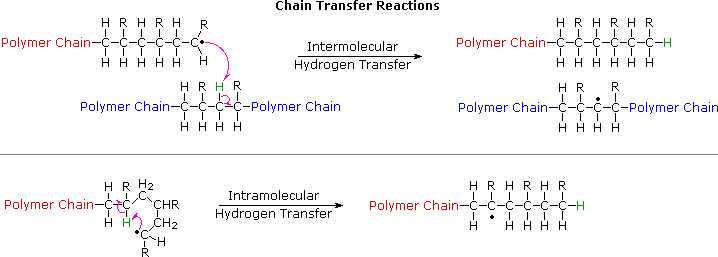
Chapter 3. Chain-growth (addition) polymerization 3.1. Free-radical
Association step: addition of a single monomer molecule to the initiating radical. A free-radical polymerization has 3 main steps: •Initiation of the active |
|
Sec. R7.1 Polymerization
For a batch reactor the mole balance on P1 and using Equation (RE7.1-2) to elim- Chains (i.e. addition) polymerization requires an initiator (I) and ... |
|
MODULE-1
? Natural and synthetic polymers. ? Addition and condensation polymers. ? Homochain and Heterochain polymer. ? Organic and inorganic polymers and element- |
|
Chapter 11 Polymerization - ScienceDirectcom
Gel point The Carothers equation can also be used to estimate the conversion needed to reach the so-called gel point in condensation polymerization involving |
|
R71 Polymerization
For a batch reactor the mole balance on P1 and using Equation (RE7 1-2) to elim Chains (i e , addition) polymerization requires an initiator (I) and proceeds by |
|
MODULE-1 - VSSUT
They are often formed by addition polymerization leading to long linear chain In this equation it was seen that the glass transition temperature e of a copolymer |
|
1 050929 Quiz 1 Introduction to Polymers 1) In class we discussed a
b) Give the 4 monomers used for the addition polymerization and their iii) Using the Carothers equation what would be the critical extent of reaction based on |
|
Living Anionic Addition Polymerization
It is noteworthy that in the case of radical anion initiators, such as alkali-metal naphthalenide, f value in the above equation is 2 In this case, the mole of resulting |
|
Polymerization - Wright State University
Chain-growth polymerization or addition polymerization Step-growth Divide the first equation by the second equation, we obtain the Mayo-Lewis equation:: |
|
Addition Polymerisation
C:\Users\Paul McCormack\Desktop\Addition Polymers and Polyester Student Write an equation to show the polymerisation of Terylene and poly(lactic acid) |