addition polymerization pdf
|
ADDITION POLYMERS
ADDITION POLYMERS Many reactions of organic compounds fall into four classes Substitution: an atom or group of atoms replaces another e g CH4 + Cl2 ⇒ CH3Cl + HCl Addition: two molecules react to form one e g C2H4 + Br2 ⇒ C2H4Br2 Elimination: one molecule reacts to form more than one e g C2H5OH ⇒ C2H4 + H2O |
|
Addition Polymers
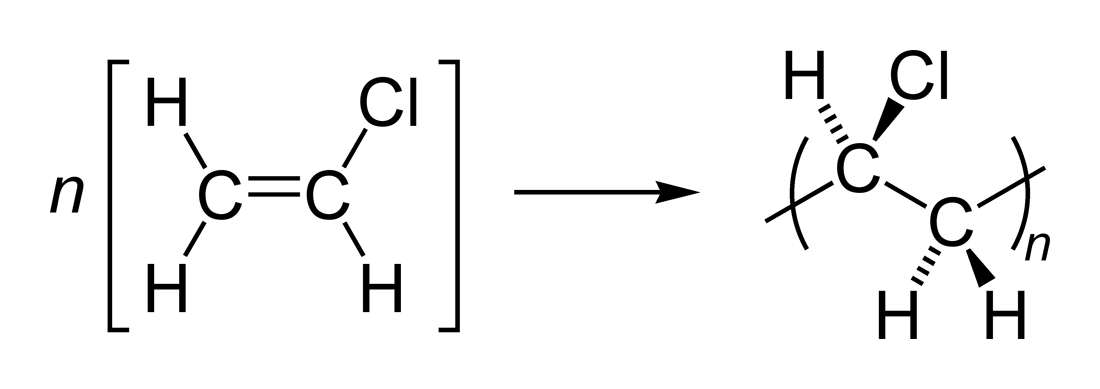
monomers to form a polymer is called polymerization Most polymerization reactions require a catalyst An addition polymer forms when unsaturated monomers react to form a polymer Ethene undergoes addition polymerization The ethene mol-ecules bond to one another to form the long-chain polymer polyethylene as described in the equation below |
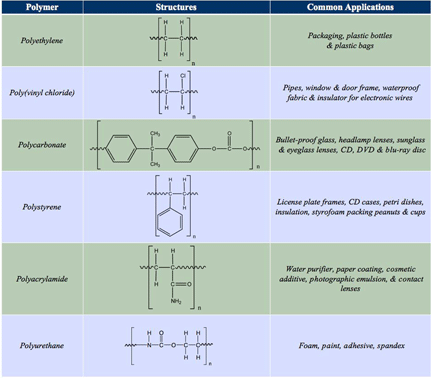
What are the uses and applications of Common polymers?
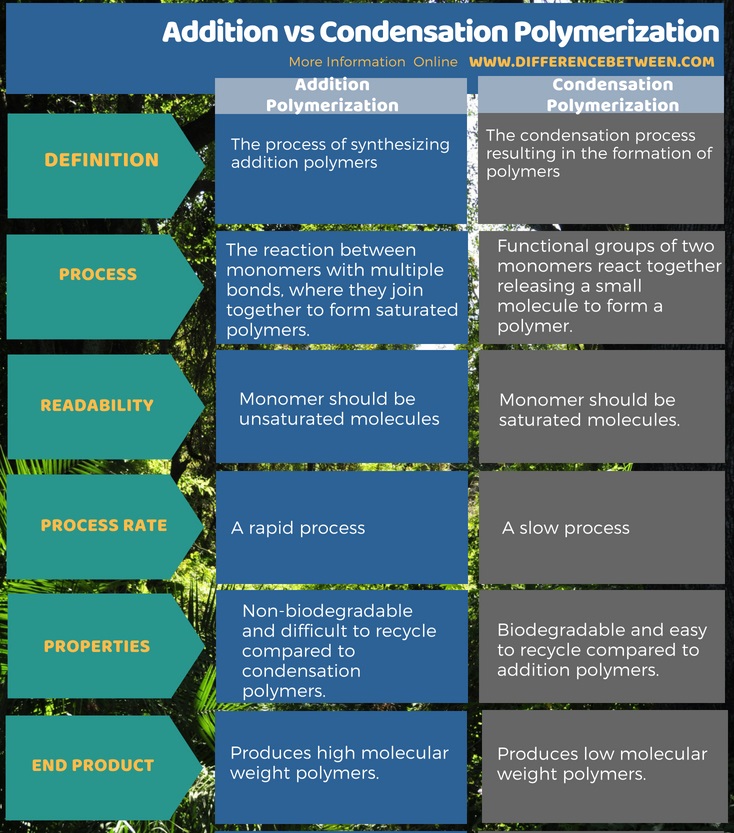
Know the uses/applications of common polymers. Addition polymerization and condensation polymerization are two modes of polymerization reactions in the formation of polymers. In addition polymerization, the monomer molecules bond to each other without the loss of any other atoms.

GCSE Chemistry

s=

Addition Polymerization
|
Polymerization by Addition Addition polymerization can be divided
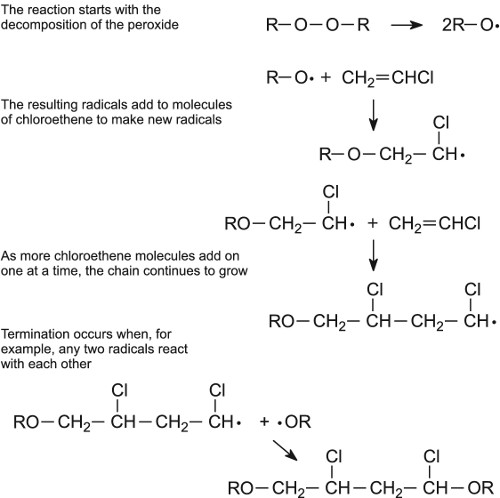
There are many types of initiators which used in addition polymerization for starting this reaction by its decomposition to free radical and active center as |
|
Continuous Addition Polymerization
Jul 6 2005 3.3.3. Addition of One Mixed Monomer Feed solution. 52. 3.3.3.1. At Constant Specific Rate of Copolymerization. |
|
Chapter 6 - POLYMER CHEMISTRY Lecture/Lession Plan -2
Examples: polyethylene polyvinylchloride |
|
Chapter Two Polymerization Mechanisms
Dec 23 2018 Polymers are classified as addition or condensation polymers depending on the type of polymerization reaction involved in their synthesis. |
|
Addition Polymerization of Anhydrosugar Derivatives. I. A
29. 1928). tion based on the dehydration of glucose to high molecular weight polymers in the presence of acids has been investigated |
|
Acrylamide Polymerization — A Practical Approach
Polyacrylamide gels are formed by copolymerization of acrylamide and bis-acrylamide (“bis” N |
|
Addition Polymerization
Use this interactive classroom activity to teach students about the steps involved in the preparation of addition polymers via free-radical chain reactions. |
|
Polymerization PDF
The small molecular unit is called a monomer. In addition to synthetic polymers such as |
|
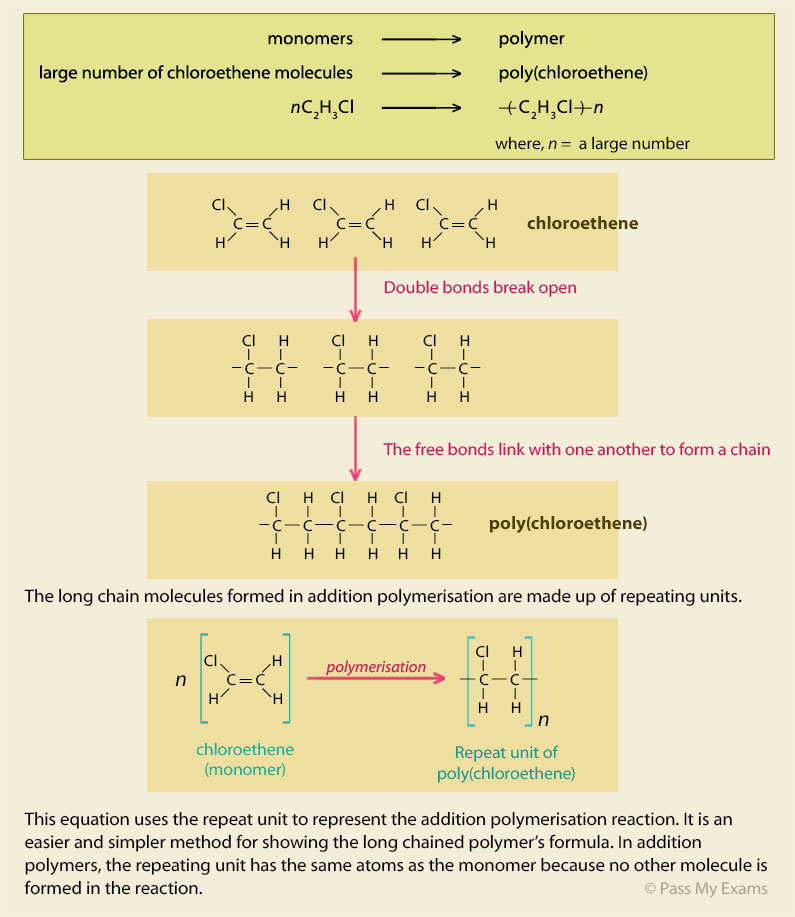
ADDITION POLYMERS
The reaction is known as an addition reaction because the monomers join together to form one big molecule the polymer |
|
Worksheet: Addition Polymerization
Q2: Which of the following is the best description of an addition polymer? A A polymer that can be easily added to other polymers. B A polymer made by linking |
|
Chapter 6 - POLYMER CHEMISTRY Lecture/Lession Plan -2
Examples: polyethylene polyvinylchloride |
|
Chapter 3. Chain-growth (addition) polymerization 3.1. Free-radical
Association step: addition of a single monomer molecule to the initiating radical. A free-radical polymerization has 3 main steps: •Initiation of the active |
|
ADDITION POLYMERS
The reaction is known as an addition reaction because the monomers join together to form one big molecule the polymer |
|
Chapter Two Polymerization Mechanisms
23 déc. 2018 Polymers are classified as addition or condensation polymers ... scheme is based on the polymerization mechanism involved in polymer ... |
|
Polymers
15.2 Write the names of monomers of the following polymers: 15.3 Classify the following as addition and condensation polymers: Terylene Bakelite |
|
Polymerization by Addition Addition polymerization can be divided
Addition polymerization can be divided into two main types: 1- Polymerization with chain growth by free radicals. 2- Ionic polymerization : Anionic |
|
Continuous Addition Polymerization
6 juil. 2005 Reaction Mechanism of Free Radical Polymerization. 8. 2.2. Reaction Kinetics of Free Radical Polymerization. 9. 2.3. Continuous Addition ... |
|
Chapter 4. Ionic and coordination (addition) polymerization 4.1
Ionic and coordination (addition) polymerization. 4.1. Similarities and contrast on ionic polymerization. 4.2. Cationic polymerization. |
|
Polymerization PDF
In addition to synthetic polymers such as |
|
Introducing Polymers Synthetic Addition Polymers
Synthetic Addition Polymers. • An addition polymer is a very long organic molecule formed as the result of addition reactions between monomers with. |
|
Polymerization by Addition Addition polymerization can be divided
Addition polymerization can be divided into two main types: 1- Polymerization with chain growth by free radicals 2- Ionic polymerization : Anionic polymerization |
|
Polymerization PDF
which are formed by addition reactions that link together monomers containing multiple bonds These polymers usually form via a free-radical mechanism in |
|
POLYMER CHEMISTRY Lecture/Lession Plan -2
6 1 1 Mechanism of addition polymerization Addition polymerization proceed through two types of mechanism A) Free radical mechanism B) Ionic mechanism |
|
ADDITION POLYMERS - PDST
These chains are called polymers The reaction is known as an addition reaction because the monomers join together to form one big molecule, the polymer, |
|
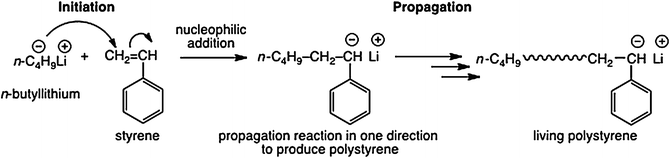
Addition Polymerization - Flinn Scientific
Select six or more student volunteers who will act as alkene monomers— molecules with at least one carbon-carbon double bond The simplest alkene monomer |
|
R71 Polymerization
Chains (i e , addition) polymerization requires an initiator (I) and proceeds by adding one This assumption is very reasonable for this initiation mechanism |
|
(addition) polymerization 41 Similarities and contrast on ionic
Ionic and coordination (addition) Similarities and contrasts in ionic polymerization The exact mechanism of coordination polymerization is still unclear |
|
Cationic Addition Polymerization (Fundamental)
The addition definitely follows a chain-growth mechanism, and such “anionic addition polymerization,” involving nucleophilic initiators and electron-deficient |












![Gel Formation in Addition Polymerization 1 - [PDF Document] Gel Formation in Addition Polymerization 1 - [PDF Document]](http://pediaa.com/wp-content/uploads/2017/06/Difference-Between-Chain-Growth-and-Step-Growth-Polymerization-Comparison-Summary.png)