addition polymerization products
What is the difference between addition polymers and condensation polymers?
Addition polymers are formed of chains of carbon atoms with strong C-H bonds and are resistant to biodegradation. Condensation polymers are formed by the elimination of water or other small molecules from two or more reactant monomers.
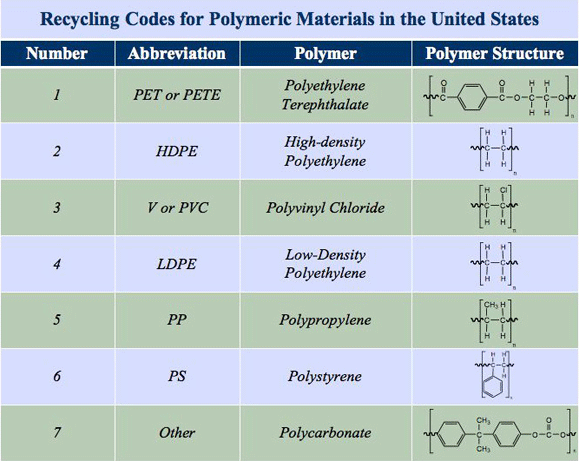
Which addition polymer is formed by polymerization of ethene and vinyl chloride?
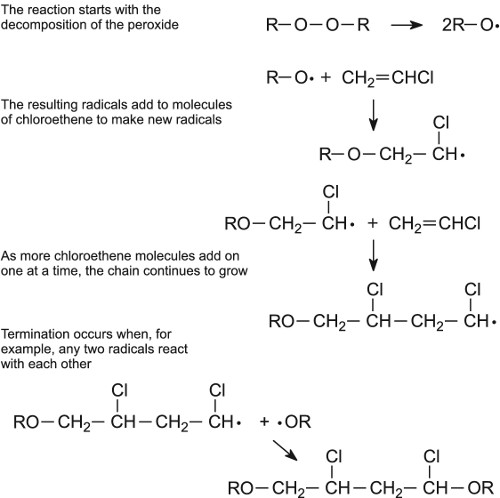
Important addition polymers include PE and PVC which are formed by polymerization of ethene (CH 2 CH 2) and vinyl chloride (chloroethene; CH 2 CHCl), respectively. Addition polymers are formed of chains of carbon atoms with strong C-H bonds and are resistant to biodegradation.

GCSE Chemistry

Addition Polymers Crash Course: HDPE LDPE PVC Polystyrene and PTFE // HSC Chemistry

Addition polymerizationpolymer introduction to polymer chemistry class 12
|
Untitled
MAKING SYNTHETIC POLYMERS. 1. Addition Polymerization. Addition polymerization is a reaction in which identical alkene/alkyne monomers are joined through |
|
Untitled
(b) addition polymerization. (c) oxidation condensation polymerization. 3. In a condensation polymerization the two products formed are a polymer and a |
|
Untitled
MAKING SYNTHETIC POLYMERS. * 1. Addition Polymerization. Addition polymerization is a reaction in which identical alkene/alkyne monomers are joined through |
|
Acrylamide Polymerization — A Practical Approach
Polyacrylamide gels are formed by copolymerization of acrylamide and bis-acrylamide (“bis” N |
|
Addition Polymerization
A chain reaction ensues as the new free radical repeats step 7 with the third alkene in the chain. This chain reaction may con- tinue as long as there are |
|
Addition Polymerization of Anhydrosugar Derivatives. I. A
of values is reported. Comparison of the Periodate Oxidation Products from 3'-. Amino-3 '-deoxycytidine 3 '-Amino-3 '-deoxyadenosine and. |
| Organic Chemistry: Polymerization Reactions |
|
Name_____________________
Which is an example of an addition reaction? A) an addition reaction. B) an esterification reaction. C) a polymerization reaction. D) a substitution reaction. 5 |
|
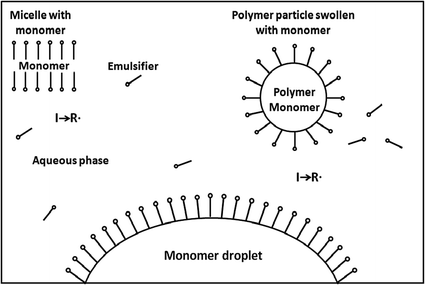
Continuous Addition Polymerization
Jul 6 2005 This term indicates that the converted monomers must continuously be replaced by adding a feed solution during the polymerization reaction. |
|
2004 Chem 2OB3 Lab Manual – Experiment 5. Synthetic Polymers.
PART A: Addition Polymers. Chain growth or addition polymers are formed by a reaction in which monomer units add to another with no other by-products. The |
|
Acrylamide Polymerization — A Practical Approach
Polyacrylamide gels are formed by copolymerization of acrylamide and bis-acrylamide (“bis” N |
|
Addition Polymerization of Anhydrosugar Derivatives. I. A
of values is reported. Comparison of the Periodate Oxidation Products from 3'-. Amino-3 '-deoxycytidine 3 '-Amino-3 '-deoxyadenosine and. |
|
Polymers
product radical thus formed reacts with another radical to form the polymerised product. The addition polymers formed by the polymerisation of a single. |
|
Addition polymerization of norbornene catalyzed by palladium(2+)
Macromolecules 1992 25 |
|
Products intended for the construction maintenance and repair of
Of other addition polymerization products. 3917. Tubes pipes and hoses |
|
Organocatalytic Conjugate-Addition Polymerization of Linear and
18 Nov 2013 Formation of single-addition product enamine I (the deoxy-Breslow intermediate) from the reaction of MMA + IMes and its X-ray crystal. |
|
Organocatalytic Conjugate-Addition Polymerization of Linear and
18 Nov 2013 Formation of single-addition product enamine I (the deoxy-Breslow intermediate) from the reaction of MMA + IMes and its X-ray crystal. |
|
NZQA - NCEA Level 2 Chemistry (91165) 2017
Addition reactions involve two (or more in the case of the polymers) molecules combining to make one molecule. An addition reaction occurs when double bonds |
|
Palladium-Catalyzed Living/Controlled Vinyl Addition
14 Oct 2021 addition polymerization of 3-methyl-3-carboxymethyl CPEs catalyzed by ... and a polymer product was obtained in 52% yield with a Mn of. |
|
Visible Light Photocatalytic Thiol–Ene Reaction: An Elegant
27 Jan 2015 Finally this highly efficient thiol?ene reaction was employed for the synthesis of polymers by step-growth addition polymerization. ? ... |
|
Addition Polymerization - Flinn Scientific
Polymers • Addition reactions • Chain reaction • Free-radical initiators Materials Student volunteers, 7 or more Safety Precautions This activity is considered |
|
ADDITION POLYMERS - PDST
Addition polymers are formed when individual molecules, known as monomers, join to form long chains These chains are called polymers Monomer Polymer Common Name Uses Ethene Poly(ethene) Polythene Low-density: plastic bags, squeezy bottles High-density: buckets, lunch boxes |
|
Polymerization PDF
Products that are commonly referred to as “plastics” by the general public are known In addition to synthetic polymers, such as, polyvinyl chloride, polystyrene, |
|
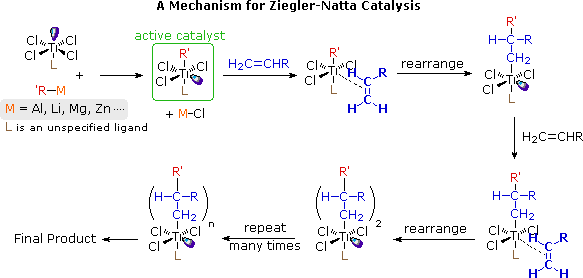
POLYMER CHEMISTRY Lecture/Lession Plan -2
Addition polymerization proceed through two types of mechanism 1) Chain initiation: This is the first step of polymerization reaction where initiator activate the |
|
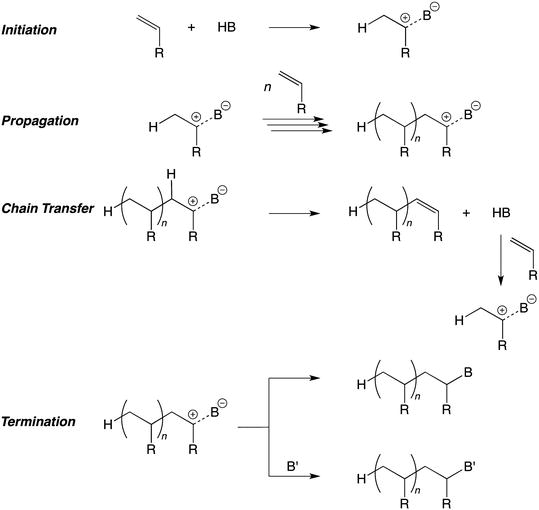
Cationic Addition Polymerization (Fundamental)
acid, yields viscous products that have turned out polymers [–(CH2–CHR)n–] The initial reaction is an electrophilic addition of HB (a proton) onto the double |
|
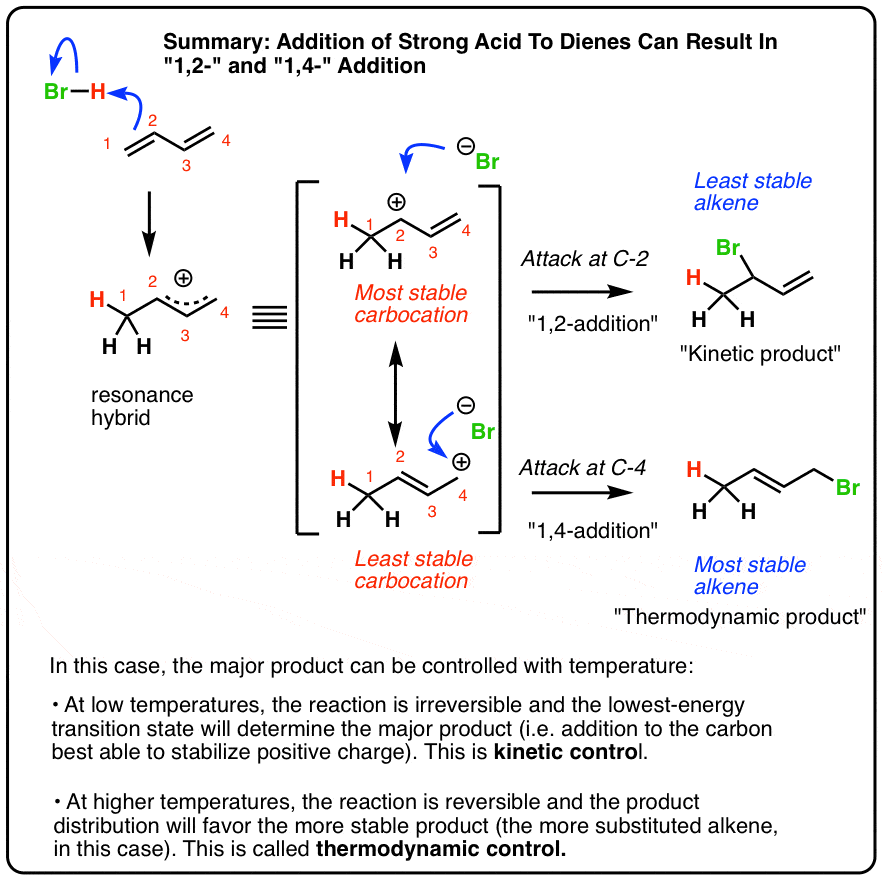
310 POLYMERIZATION Polymerization is the formation of extremely
We can diagrammatically represent this polymerization reaction with a monomer, butadiene This type of reaction is called a 1,2 addition because the first and |
|
310-polymerspdf
O Addition polymerization is a reaction in which identical alkene/alkyne monomers are joined through multiple addition reactions to form a polymer O EXAMPLE |













.jpg)