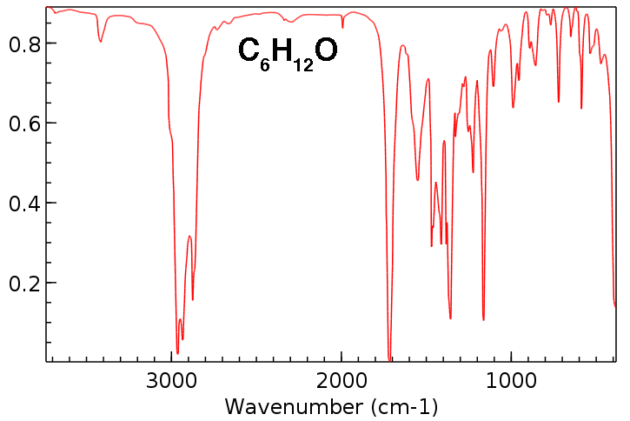
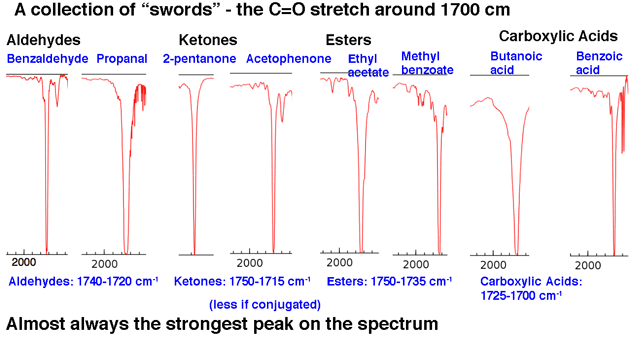
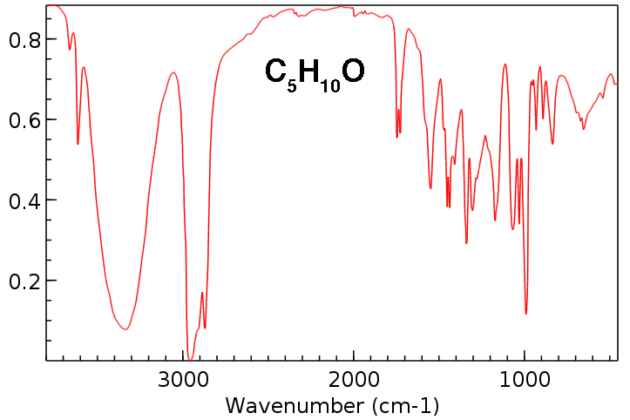
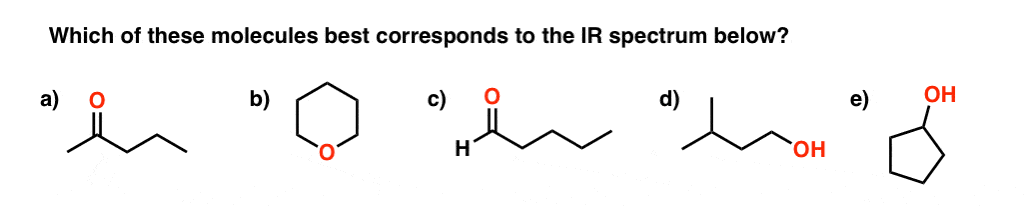
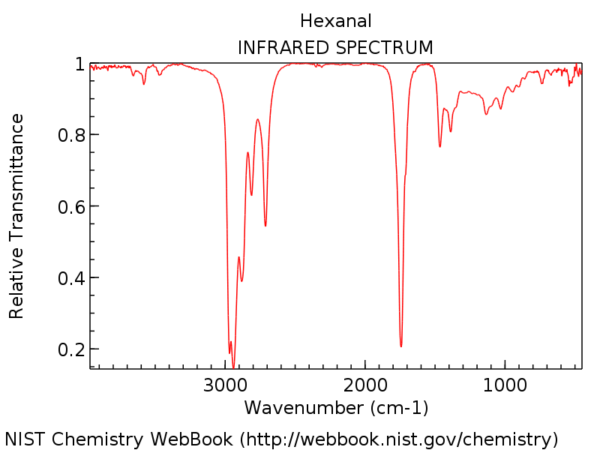
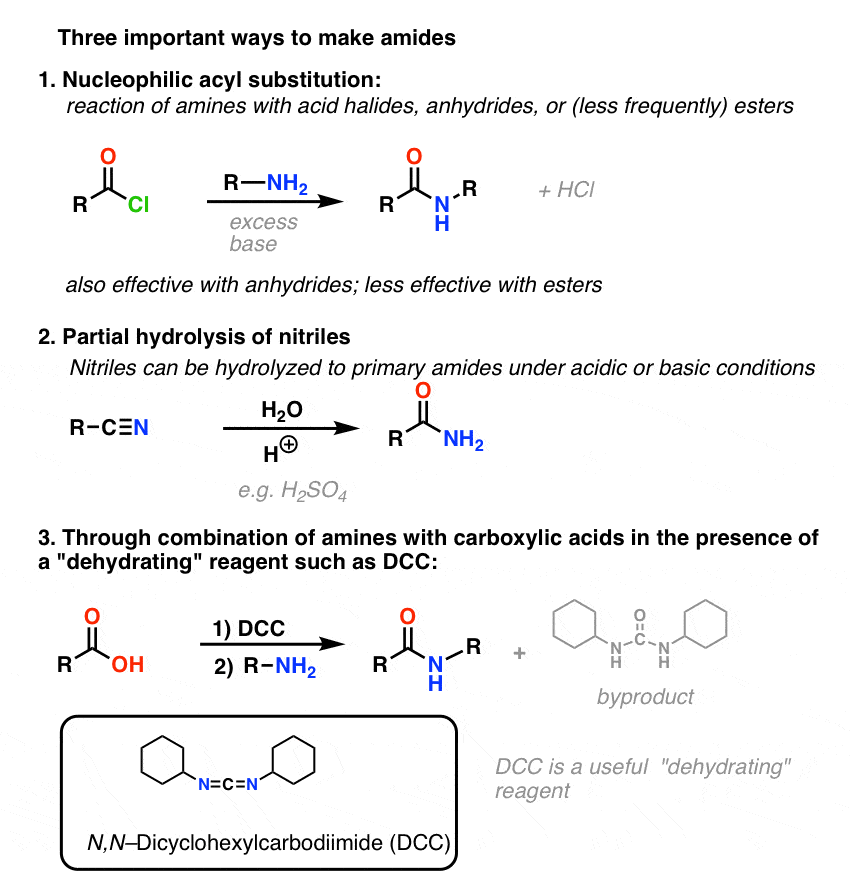
amide functional group ir spectrum
|
EXPT 7 CHARACTERISATION OF FUNCTIONAL GROUPS USING
You can interpret IR spectra by recognising characteristic shapes and patterns within the spectrum and by applying the information obtained from extensive |
|
CH2SWK 44/6416 IR Spectroscopy 2013Feb5 1 1 Theory and
5 fév 2013 · IR most useful for identification of functional groups (See Appendix B epage 131) 4 2 Peaks are not solely due to one functional group |
|
Characteristic vibrational frequencies of functio
The infrared spectrum of an organic compound contains a large number of bands due to the stretching and bending vibrational modes of each bond Thus it is quite |
|
IR Absorption Frequenciespdf
Typical IR Absorption Frequencies For Common Functional Groups Bond Functional Group Frequency in cm-1 (Intensity*) C-H Alkane 3000-2850 (s) -CH3 1450 |
What is the spectrum of amide in IR?
The absorption spectrum bands of amide infrared radiation (IR), amide I (1600–1800 cm–1), amide II (1470–1570 cm–1), amide III (1250–1350 cm–1), and amide A (3300–3500 cm–1), are peaks of infrared characterization of amide.
What functional group is 3000 cm-1?
IR SPECTRUM OF ALKANES
Of these the most useful are the C-H bands, which appear around 3000 cm-1.
Since most organic molecules have such bonds, most organic molecules will display those bands in their spectrum.First of all the IR (infrared) frequency light is passed through the compound and accordingly the amount and the frequency of the light absorbed relates and identifies the functional groups and the structure of the compound and which ultimately helps us to identify the compound.
What is the stretching frequency of amides in IR?
The NH2 stretching frequencies of amides in chloroform show a restricted range. ν̄NHasym varies from 3510 cm−1 to 3532 cm−1. ν̄NHsym varies from 3389 cm−1 to 3424 cm−1.
The higher frequency of NH2 stretching frequencies of amides over amines is attributed to an increased NH2 angle.
|
INFRARED SPECTROSCOPY (IR)
The amide functional group combines the features of amines and ketones because it has both the N-H bond and the C=O bond. Therefore amides show a very strong |
|
Infrared Tables (short summary of common absorption frequencies
disappear i.e. a symmetrically substituted alkyne!). Infrared spectra are generally informative about what functional groups are present |
|
Infrared Spectroscopy
15-May-2013 Different types of bonds and thus different functional groups |
|
IR - spectroscopy
Amide I band ? C=O stretching Amide II band ? N-H bending in plane. Why the wavenumber of I amide band is usualy below 1700 cm-1 ? 18. Page 19. Amides bond |
|
How to Read and Interpret FTIR Spectroscope of Organic Material
FTIR infrared spectrum |
|
CHEMISTRY PAPER No. 12: ORGANIC SPECTROSCOPY Module
Module 9: Characteristic vibrational frequencies of functional groups carbonyl bond. The C=O stretching band in IR spectra of amide is called amide I band |
|
FTIR Analysis of Protein Structure
Before describing the structural information that FTIR spectroscopy provides a with the carboxylic acid group on another amino acid to form an amide ... |
|
The Amide I Spectrum of Proteins—Optimization of Transition Dipole
10-Feb-2020 Dipole Coupling Parameters Using Density Functional Theory. Calculations ... proteins and to simulate the infrared spectrum in the amide I. |
|
Elucidation of Functional Groups on Gram-Positive and Gram
cell walls (i.e. hydroxyl |
|
Lab 14: Qualitative Organic Analysis
one major functional group (alcohol ketone |
|
Infrared Tables (short summary of common absorption frequencies
Amides Anhydrides Acid Chlorides saturated = 1725 conjugated = 1690 aromatic = does not have C=O band IR Spectrum aldehydes C O aldehyde C-H |
|
Ir lecture part 2
by finding peaks at 1600 and 1500 cm-1 and C-H out-of-plane bending to give either an alcohol, N-H containing amine or amide, or carboxylic acid For -NH2 |
|
Infrared Spectroscopy - IFSC/USP
15 mai 2013 · The complexity of this spectrum is typical of most infrared spectra, and Infrared absorption data for some functional groups not listed in the preceding table are aldehyde, ester, or carboxylic acid; furthermore it can tell you |
|
IR 2 - IR - spectroscopy
hydrogen bonds Spectrum of carboxylic acid in gas phase – only monomeric molecules 15 δ N-H bending in plane Why the wavenumber of I amide band is usualy below 1700 cm-1 ? 18 ω CH aromatic (out of plane) 711cm-1 645 20 |
|
IR SPECTROSCOPY - UPV
same IR spectrum (except enantiomers) • Simple ➢sp3 C-H, just below 3000 cm-1 (to the right) ➢sp2 C-H, just The C=O group of an amide absorbs at an |
|
IR Spectroscopy Electromagnetic radiation has dual wave/particle
for infrared spectroscopy of organic compounds is about 2500 The carbonyl group in IR spectroscopy ester, amide, anhydride, acid chloride, and thioester |
|
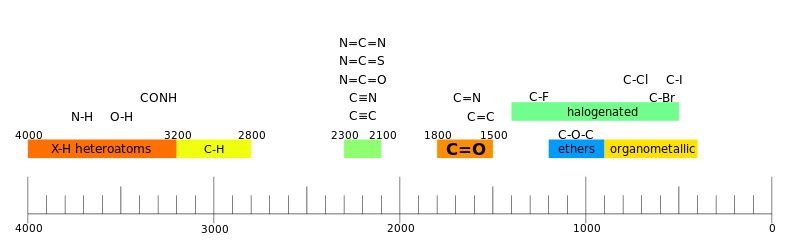
IR absorptions
C-H O-H N-H Triple Bond Region (2000-2500) C C C N Double Bond Region Amide N-H C=O Carbonyl Group C=O C–O Carboxylic Acids O-H C=O |