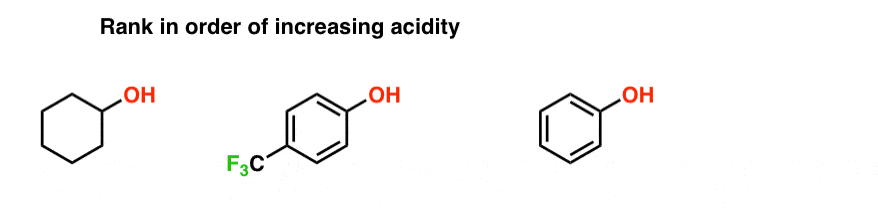
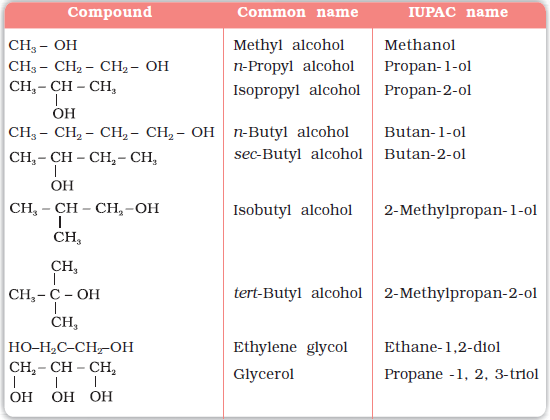
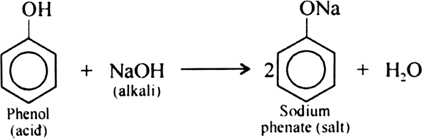compare acidity of alcohols and phenols
Is p hydroxybenzaldehyde more acidic than phenol?
Thus, p -hydroxybenzaldehyde is more acidic (p Ka = 7.9) than phenol (p Ka = 9.89). The following data for isomeric four-carbon alcohols show that there is a decrease in boiling point with increasing substitution of the OH-bearing carbon. How might you account for this trend?
Which compound is more acidic methanol or ethanol?
Compounds with a smaller Ka and larger p Ka are less acidic, whereas compounds with a larger Ka and smaller p Ka are more acidic. As shown in Table 17.1, simple alcohols like methanol and ethanol are about as acidic as water, but the more highly substituted tert -butyl alcohol is somewhat weaker.
Is phenol a weak acid?
Compounds like alcohols and phenol which contain an -OH group attached to a hydrocarbon are very weak acids. Alcohols are so weakly acidic that, for normal lab purposes, their acidity can be virtually ignored. However, phenol is sufficiently acidic for it to have recognizably acidic properties - even if it is still a very weak acid.
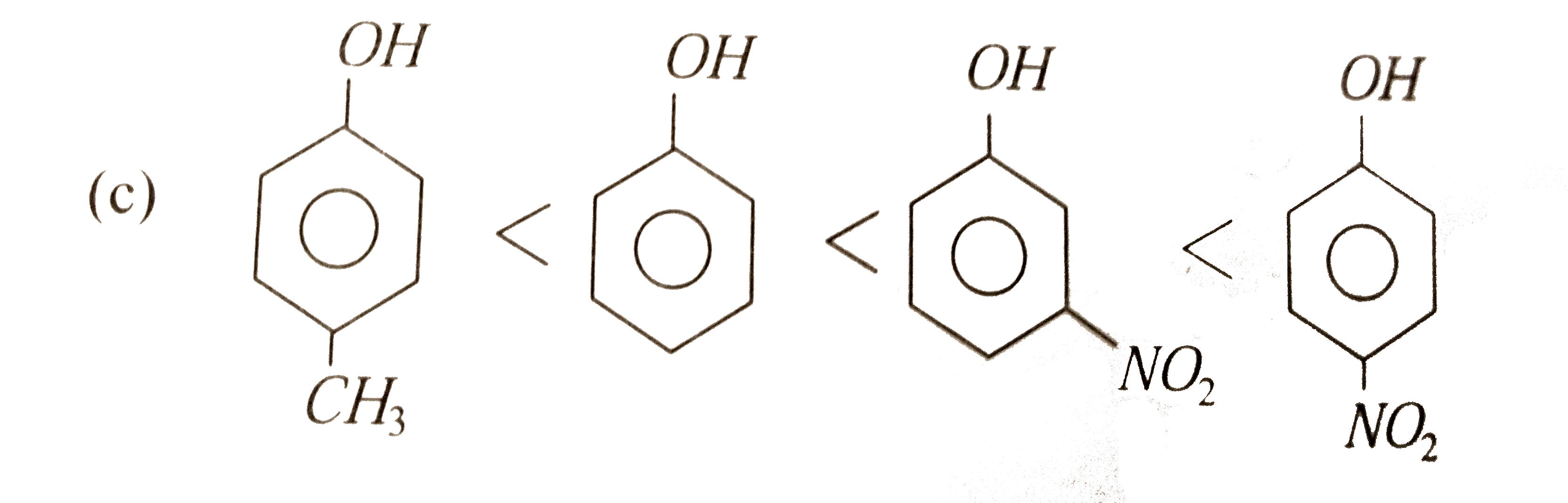
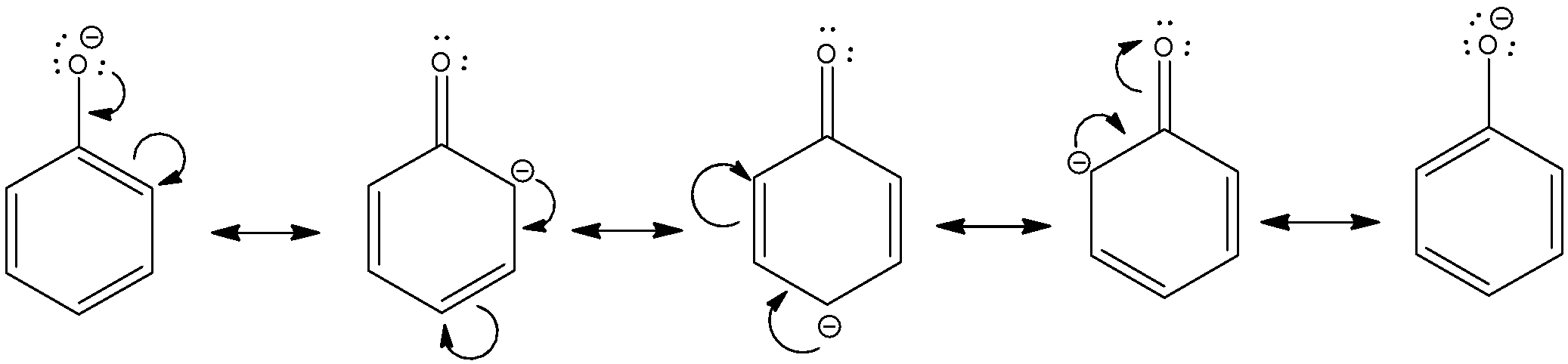
Why is phenol more acidic than phenols with electron donating substituents?
Phenols with an electron-withdrawing substituent are more acidic because these substituents delocalize the negative charge; phenols with an electron-donating substituent are less acidic because these substituents concentrate the charge.
|
Chapter 17: Alcohols and Phenols
17.3: Properties of alcohols and phenols: acidity and basicity: Like water alcohols are weak Brønsted bases and weak. Brønsted acids. |
|
Alcohols Phenols and Ethers
From the above data you will note that phenol is million times more acidic than ethanol. Arrange the following compounds in increasing order of their acid |
|
Alcohols and Phenols (pp. 571-574)
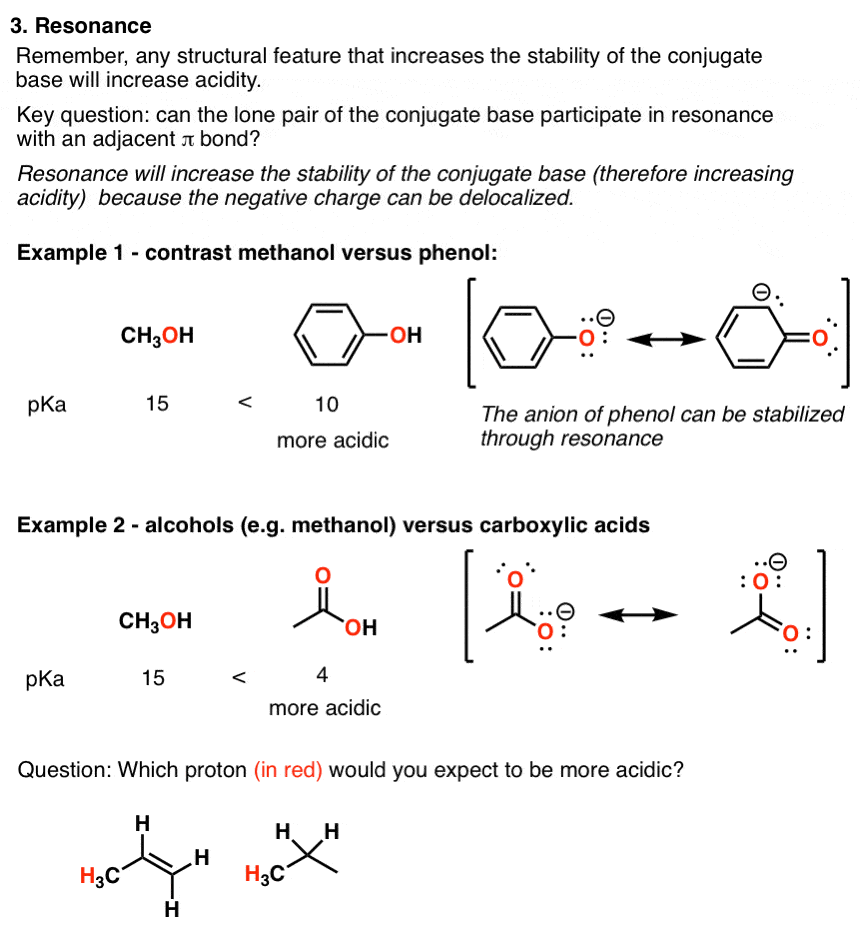
explore three factors for comparing the acidity of alcohols. 1. Resonance. As a striking example compare the pKa values of cyclohexanol and phenol:. |
|
Acidity of Alcohols and Phenols
Acidity of Alcohols and Phenols. 1. Page 2. INTRODUCTION. •Alcohols are the organic compounds that contain alkane hydrocarbon chain in. |
|
6-Alcohols-and-Phenols.pdf
Some alcohols react with ZnCl2 in an acidic aqueous solution to give an alkyl choride. You will compare the solubility of phenol in water in. |
|
Chapter 5 Carboxylic Acids and Esters
Carboxylic acid. Alcohols. Aldehydes/Ketones. Ethers. Alkanes. Comparing Physical Properties carboxylic acid with an alcohol or phenol (plus an. |
|
120 Chapter 24: Phenols. Alcohols contain an OH group bonded to
24.5: Substituent Effects on the Acidity of Phenols. Electron-donating substituents make a phenol less acidic by destabilizing the phenoxide ion (resonance |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
phenols are more acidic than alcohols. The higher acidity of carboxylic acids as compared to phenols can be understood similarly. The conjugate. |
|
Chapter 17: Alcohols and Phenols
76 Chapter 17: Alcohols and Phenols phenol (aromatic alcohol) pKa~ 10 alcohol pKa~ 16-18 O C H C O C C H enol keto chemistry dominated by the keto |
|
120 Chapter 24: Phenols Alcohols contain an OH group bonded to
24 5: Substituent Effects on the Acidity of Phenols Electron-donating substituents make a phenol less acidic by destabilizing the phenoxide ion (resonance effect) |
|
Alcohols, Phenols and Ethers - NCERT
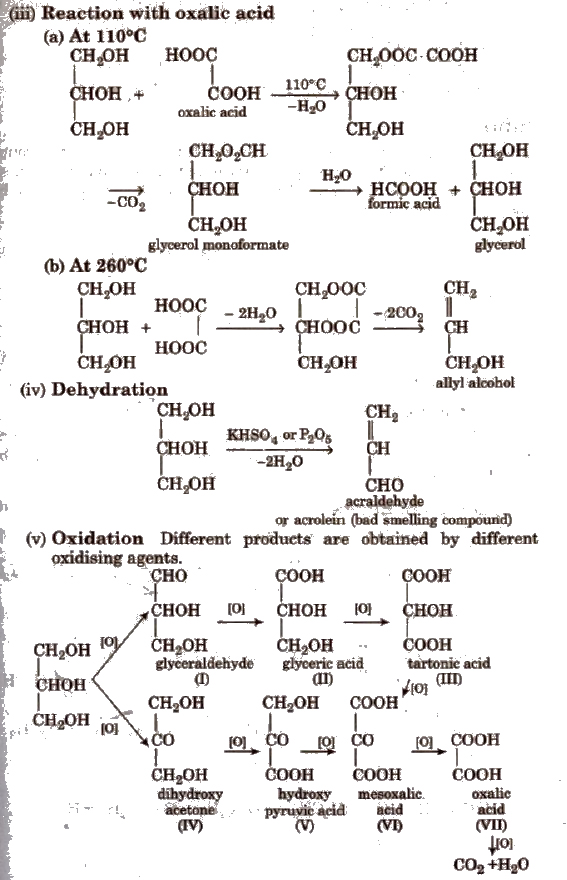
Acidic dehydration of alcohols, to give an alkene is also associated with substitution reaction to give an ether The method is suitable for the preparation of ethers |
|
Chapter 3 Alcohols, Phenols, and Ethers - Angelo State University
Chapter Objectives: • Learn to recognize the alcohol, phenol, and ether functional groups • Learn the IUPAC system for naming alcohols, phenols, and ethers |
|
6-Alcohols and Phenols
Some alcohols react with ZnCl2 in an acidic aqueous solution to give an alkyl choride You will compare the solubility of phenol in water, in the weak base |
|
Acidity of phenols, effect of substituents on ac - Yengage
Phenols are less acidic than Carboxylic acids (pKa ≈ 5) COMPARISON OF ACIDITY OF PHENOLS AND ALCOHOLS • Phenol exists as resonance hybrid of |
|
ALCOHOLS, PHENOLS AND ETHERS - NIOS
Name simple alcohols according to IUPAC system of nomenclature; ○ List general Explain the greater acidity of phenols as compared to alcohols; ○ Discuss Compare the boiling points of ethyl alcohol and dimethyl ether Which one |
|
Carboxylic Acid Structure and Chemistry
amines, alcohols, phenols, aldehydes, ketones, esters, amides and isosteric " relative acidity" of carboxylic acids it may be beneficial to compare these |
|
Organic Chemistry II
9 août 2019 · Alcohols, phenols and ethers may be viewed as organic derivatives of water A comparison of boiling points among isomeric alcohols; The boiling o The above reactions show that alcohols and phenols are acidic in nature |
|
Unit 11 Alcohols, Phenols and Ethers One mark questions - PUE
5 Give two reactions that show acidic nature of phenol Ans: Reaction (1) C6H5OH + Na → C6H5ONa + H2 ( |