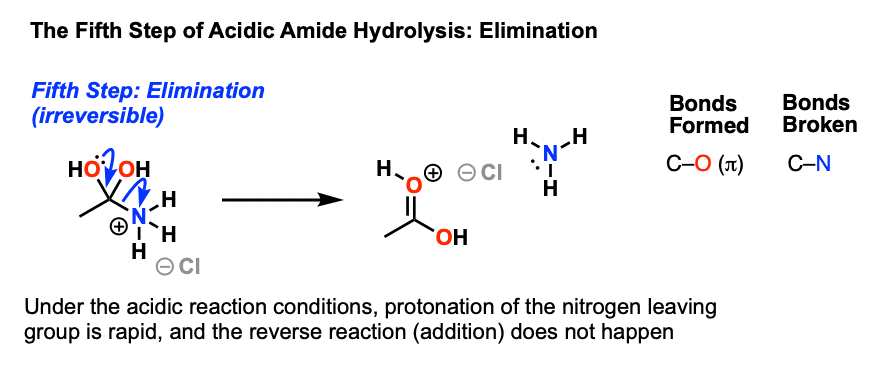
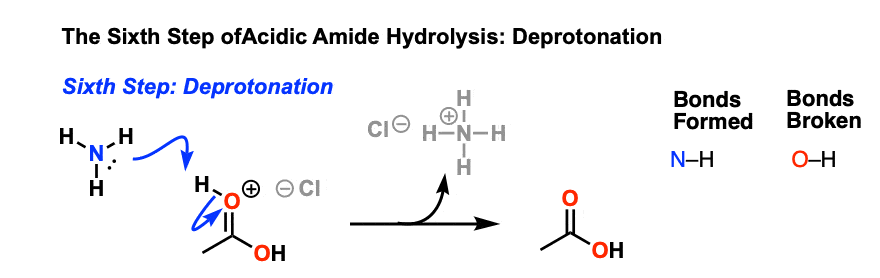
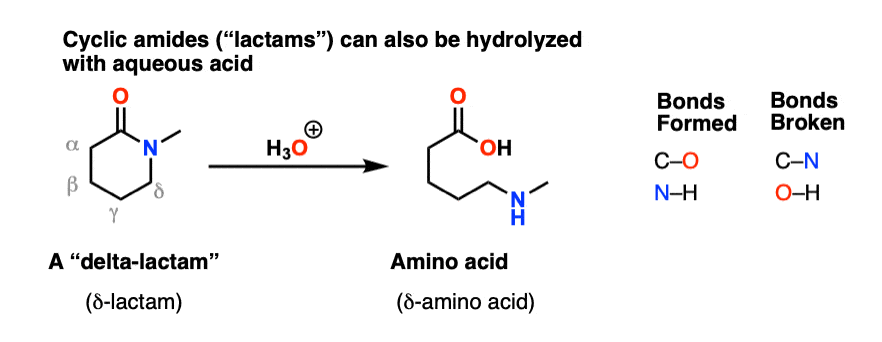
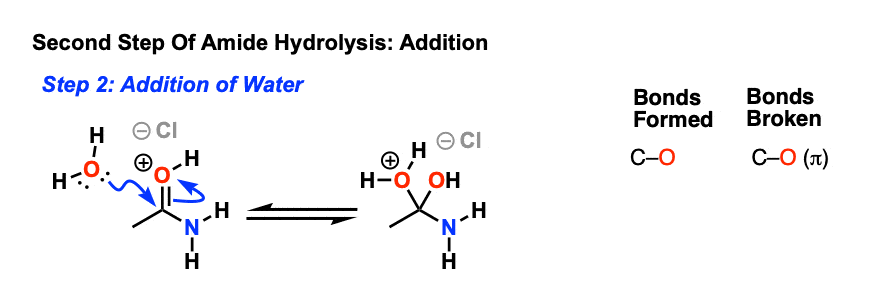
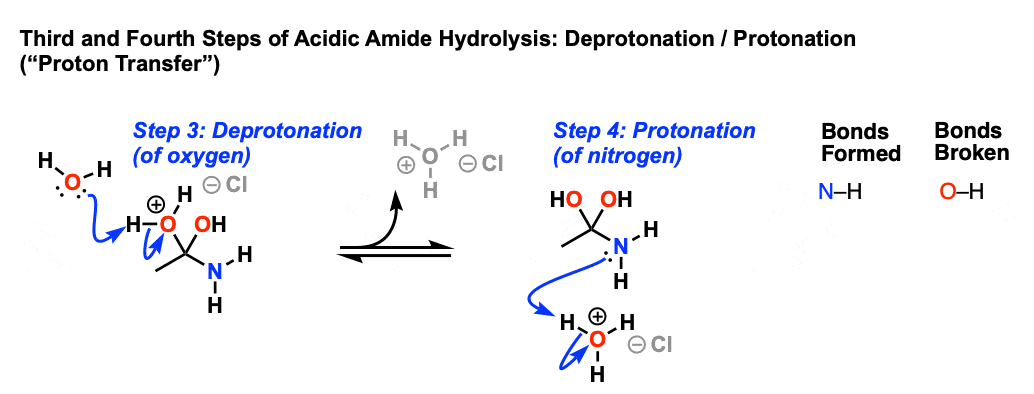
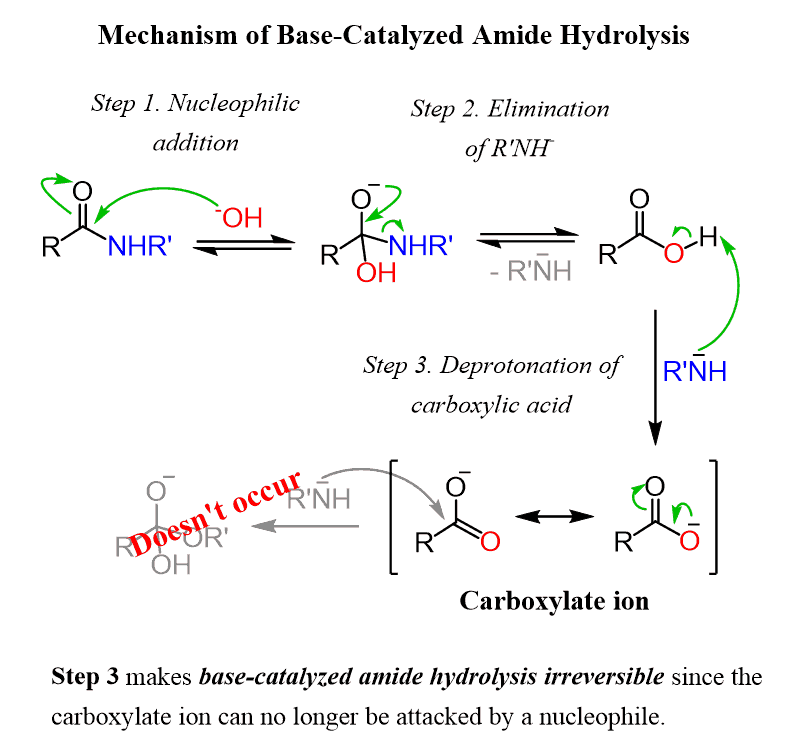
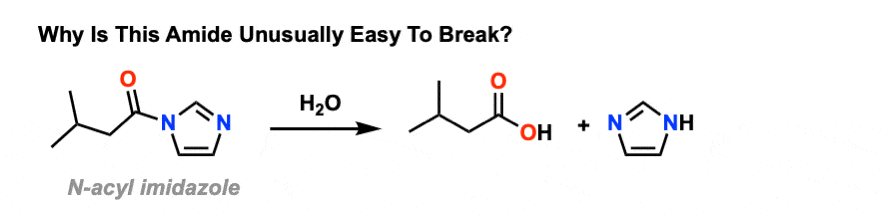
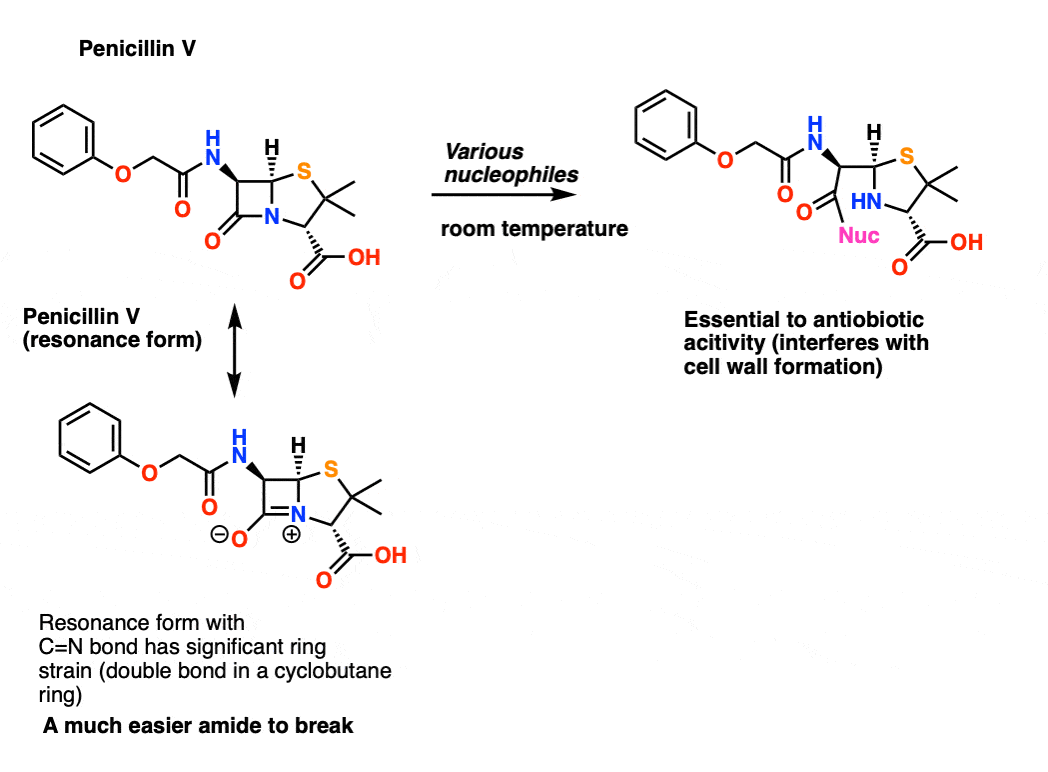
amide hydrolysis under acidic conditions
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
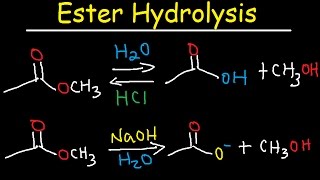
Simple alkyl esters such as methyl and ethyl esters are hydrolysed under acidic conditions in a reaction mechanism that is described as an AAC2 reaction: the |
|
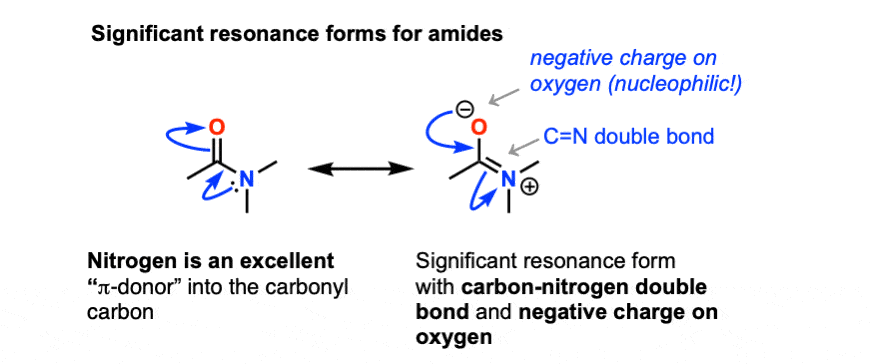
Acidic and Basic Amide Hydrolysis //
characterises the response of the reaction rate to changing acid conditions and its values are approximately the same for a series of compounds and may be |
|
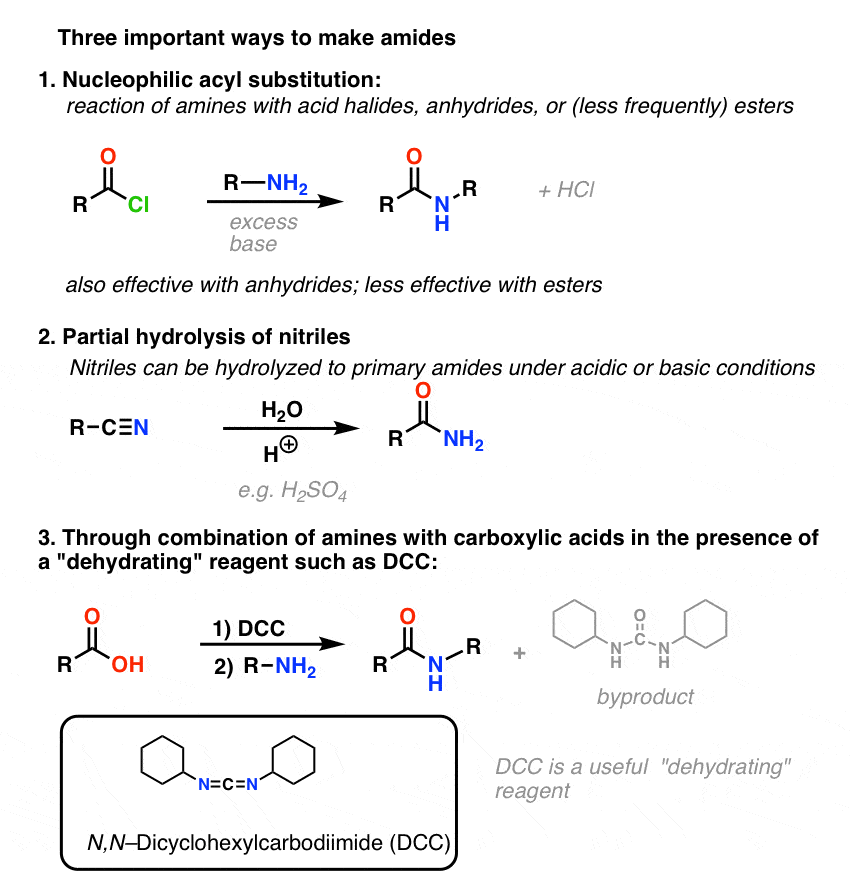
Chapter 6 Amines and Amides
Unlike amines amides are not basic • Amide hydrolysis can take place under acidic or Hydrolysis of amides under acidic and basic conditions amide |
Dilute acid hydrolysis occurs at concentration of 0.3%–2.5% (wt./wt.) at high temperature around 120°C–210°C, while concentrated acid hydrolysis happens with concentration of 41%–86% and low temperature of around 25°C–70°C [81].
What are the conditions for amide bond hydrolysis?
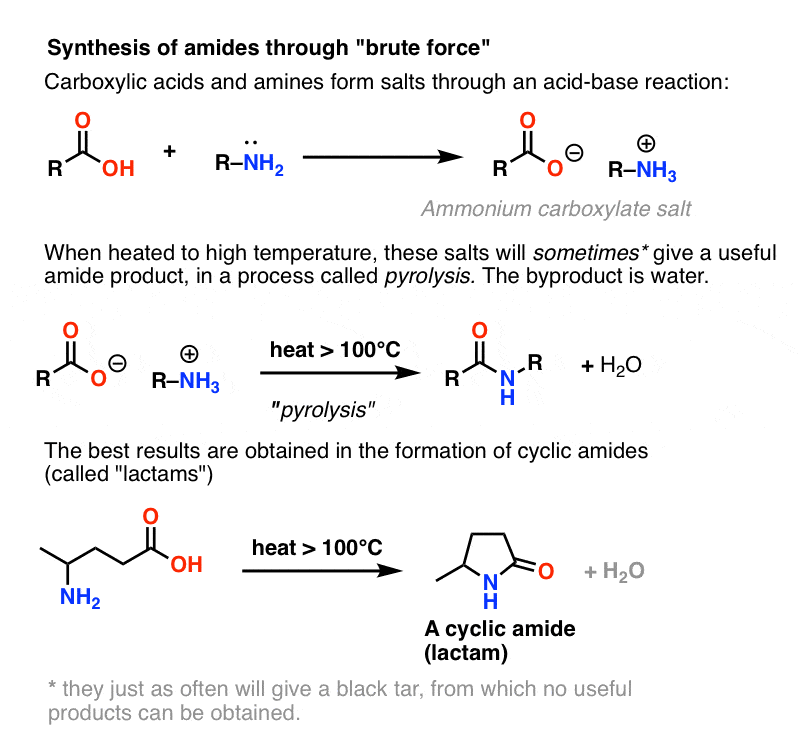
Amides can be hydrolyzed into a carboxylic acid and ammonia or an amine by heating in an acidic or basic aqueous solution.
In both cases, acid-base reactions occurring with the products make the overall reaction irreversible.
How does amide hydrolysis under acidic conditions?
Hydrolysis of Amide
In an acidic medium, amide interacts with the water molecule to give a carboxylic acid and the salt of ammonia or amine salt.
In a basic medium, amide interacts with the water molecule to give a carboxylic acid and the salt of ammonia or amine salt.
|
Mild hydrolysis or alcoholysis of amides. Ti(IV) catalyzed conversion
Reaction of primary amides (e.g. l a or 6-13) or 0-methylhydroxamates (lb and lc) Les conditions d'hydrolyse dCcrites ne compromettent pas I'intCgritC. |
|
A mild alkaline hydrolysis of N- and NN-substituted amides and
In general nitriles and amides are exceptionally stable to acid and basic hydrolysis and classically they are hydrolyzed under vigorous reaction conditions |
|
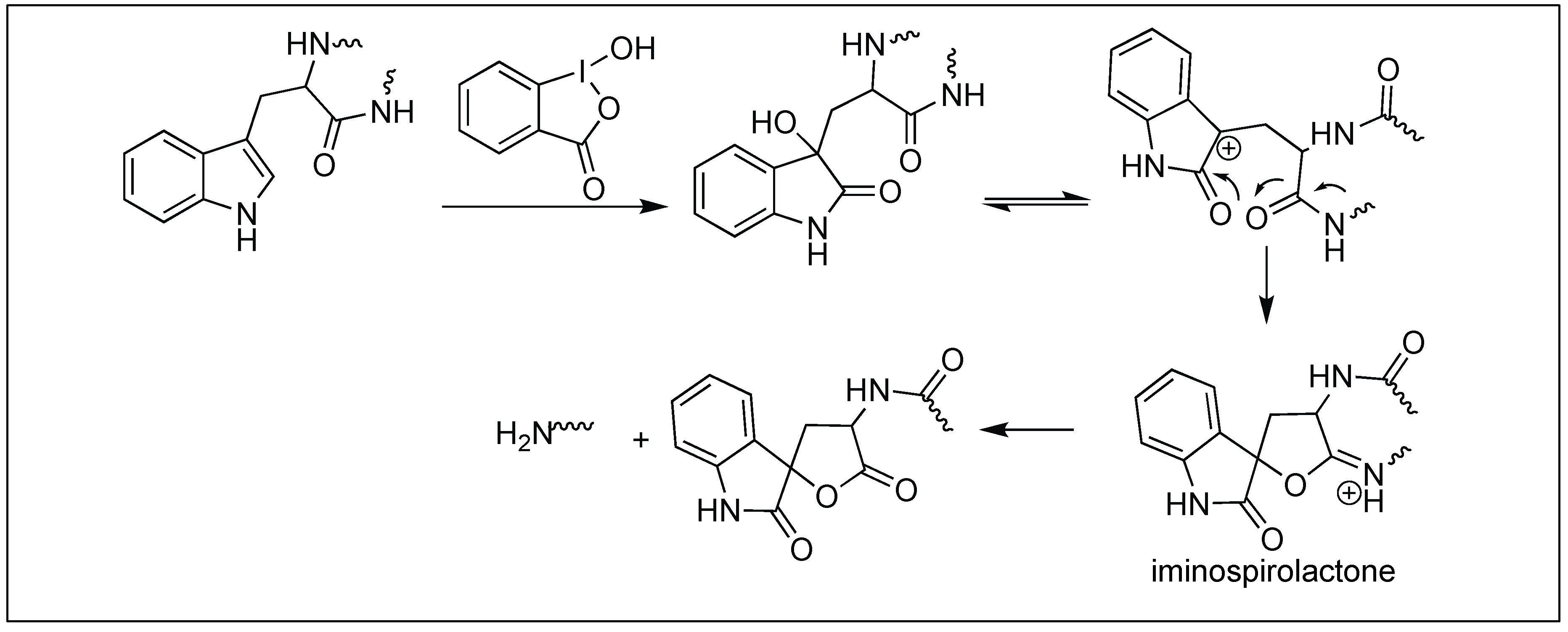
The Hydrolysis of Vitamin B12. Studies with Model Amides
these and other model amides in aqueous hydrochloric acid-dioxane at 50° are recorded. Aquocobalamin has been hydrolyzed under identical conditions; |
|
Theoretical Studies of Hydrolysis and Stability of Polyacrylamide
The hydrolysis reaction of the amide group (AM) to form acrylic acid (AA) is amide hydrolysis under different conditions have been extensively studied ... |
|
Catalysis of Amide Hydrolysis and Formation under Neutral
by H20 and aniline or morpholine under conditions comparable to those used for the catalyzed hydrolysis of the amides. Experimental Section. General. |
|
Study on Hydrolysis Properties and Mechanism of Poly(3
10. jul. 2022 product under acidic conditions [8] and the carboxylate was preferred under ... when the amide was hydrolyzed under alkaline conditions. |
|
Study on Hydrolysis Properties and Mechanism of Poly(3
10. jul. 2022 product under acidic conditions [8] and the carboxylate was preferred under ... when the amide was hydrolyzed under alkaline conditions. |
|
THE KINETICS OF HYDROLYSIS OF THE AMIDE GROUP IN
mechanism by which free aspartic acid is liberated from proteins under mild conditions of acid hydrolysis.8-11. Earlier workers have provided fragmentary |
|
Models of zinc-containing proteases. Catalysis of cobalt(III
Abstract: The rate of intramolecular amide hydrolysis in substitutionally inert Co(III) corresponding trans isomer under acidic conditions (D). The. |
|
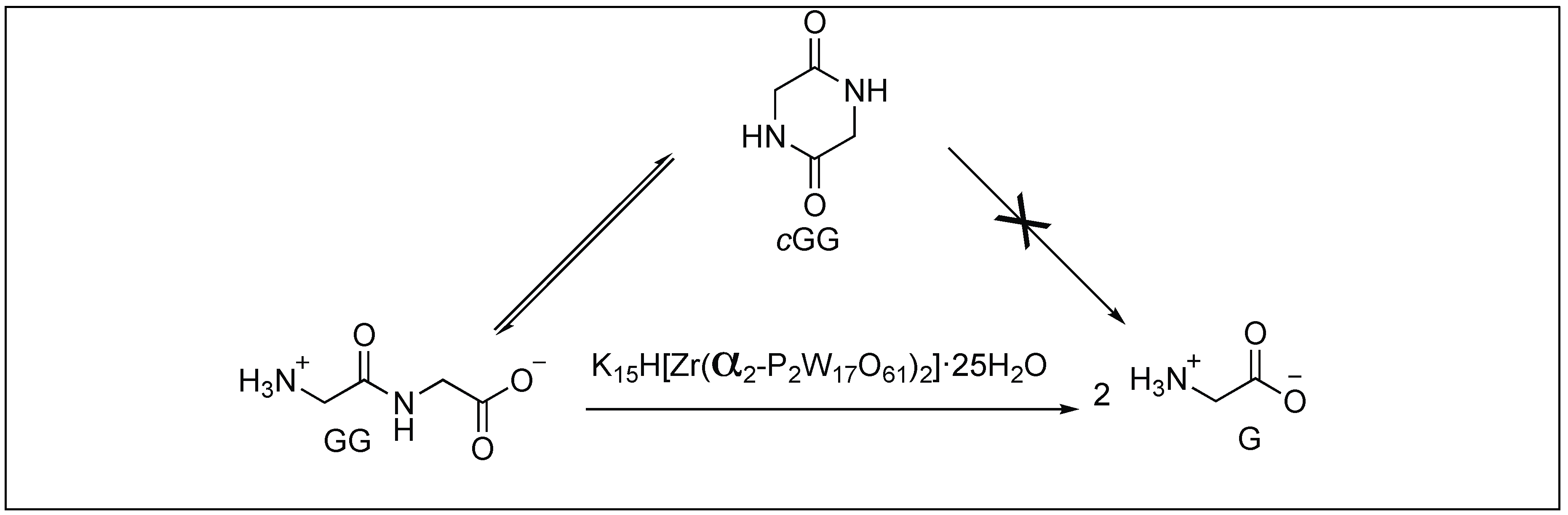
On the hydrolysis mechanisms of amides and peptides
15 mai 2018 · different amide bonds within the small fragment dipeptide Table 3 3 Dashed lines indicate slopes of –1 (low pH), +1/2 (PAGVH, moderate pH) |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions; • account for the irreversibility of the hydrolysis reaction under basic |
|
HYDROLYSIS
general, hydrolysis products predominant under neutral conditions, whereas SN2 reaction, where the phosphate ester anion is acting as the leaving group |
|
THE HYDROLYSIS OF AMIDES IN THE ANIMAL - ScienceDirect
and confirmed by Werner,G that under certain conditions the creased; the velocity of amide hydrolysis by acid, on the other hand, is roughly proportional to the |