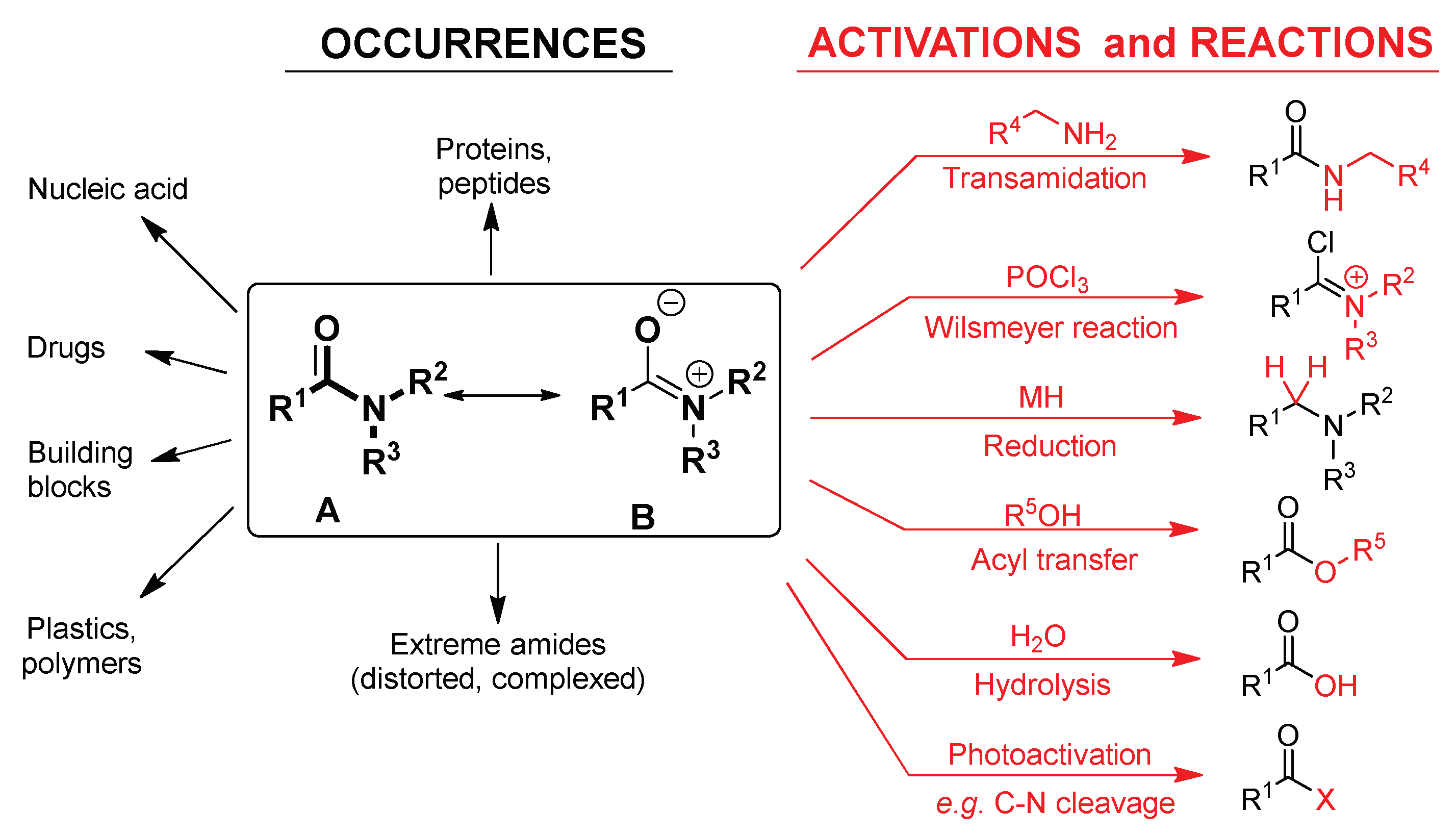
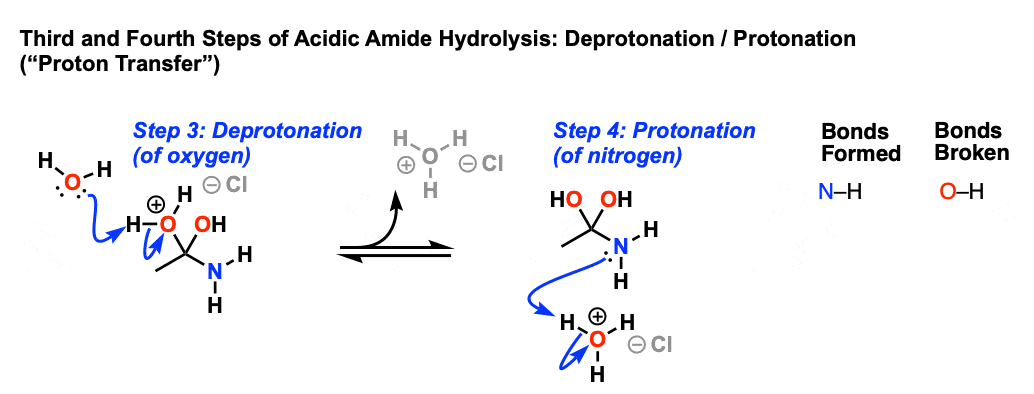
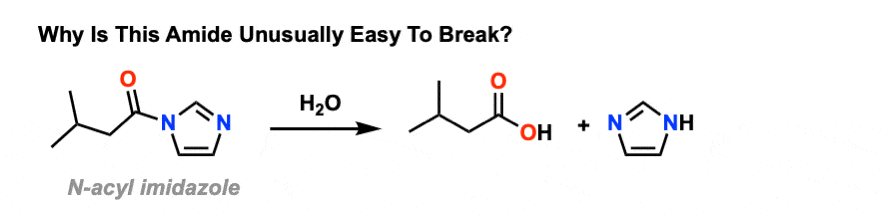
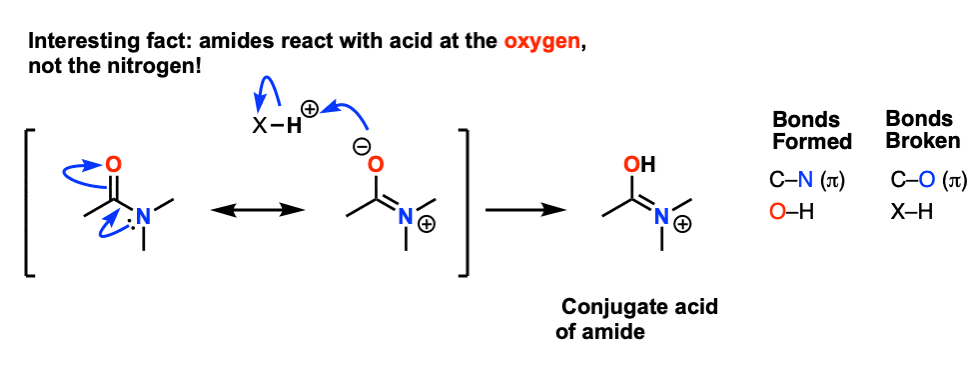
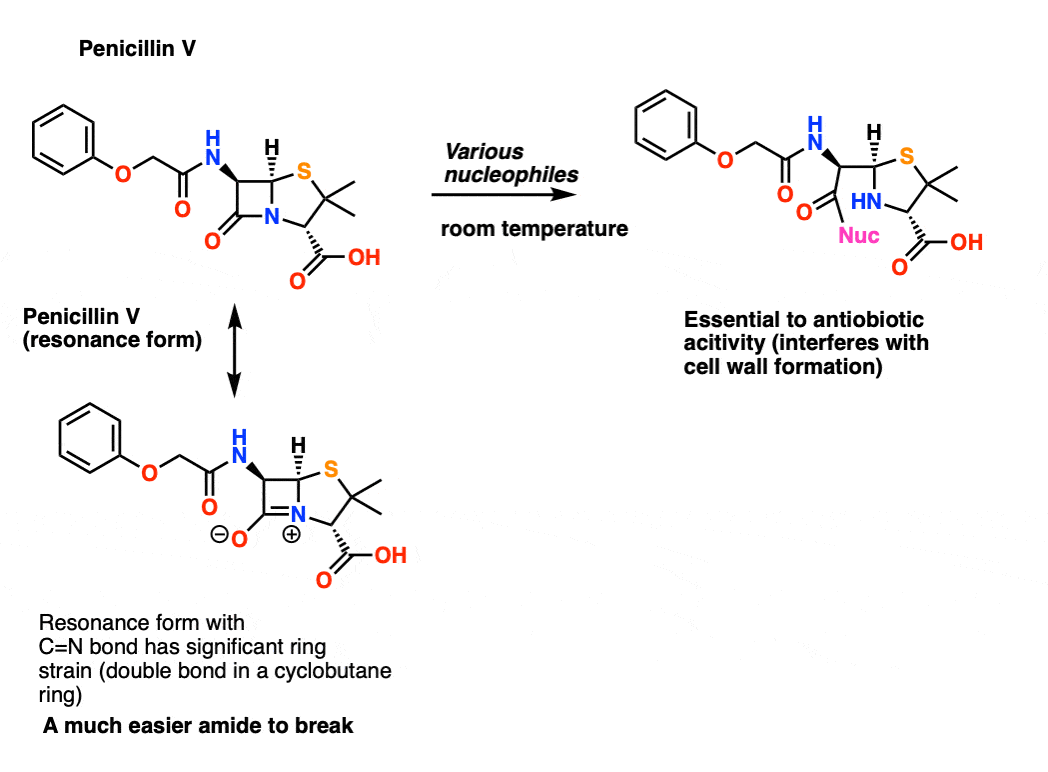
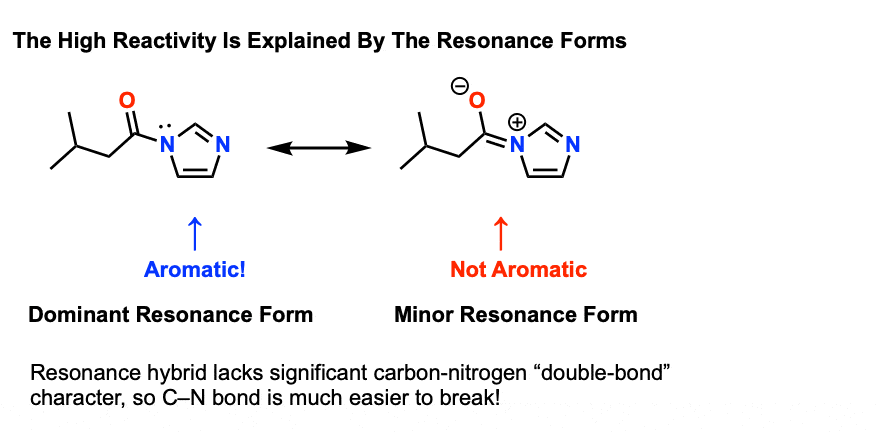
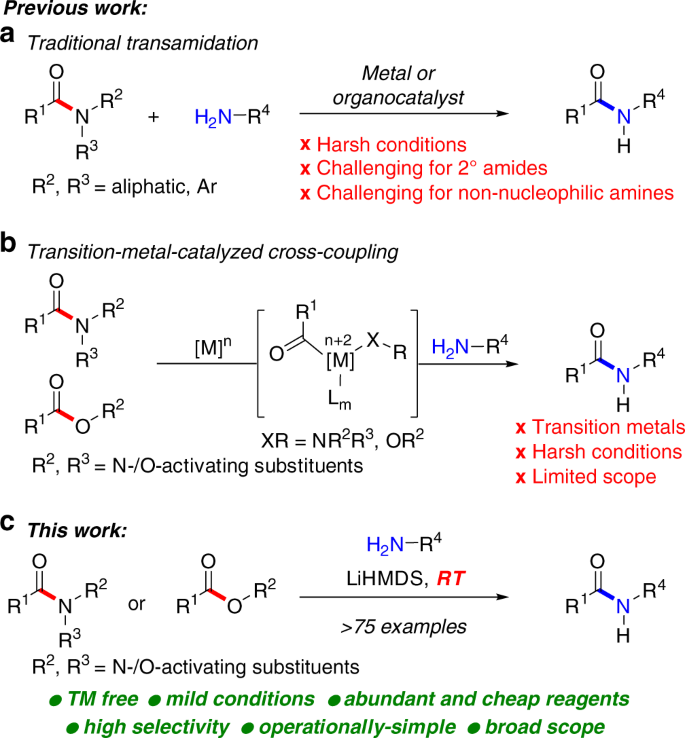
amide hydrolysis under basic conditions
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
However the rate of ester hydrolysis can be substantially increased by carrying out the reaction under acidic or basic conditions Acid-Catalysed Hydrolysis of |
|
Acidic and Basic Amide Hydrolysis //
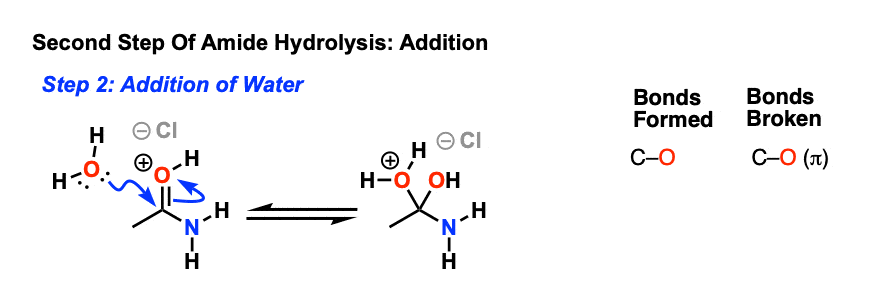
A Substituent Effects -The mechanism generally accepted (Scheme 1) for the hydrolysis of amides in dilute acid solutions involves attack by a water molecule |
|
HYDROLYSIS
Hydrolysis is an important degradation reaction in surface ground fog and porewaters and can be a dominant pathway in biological systems as well In general |
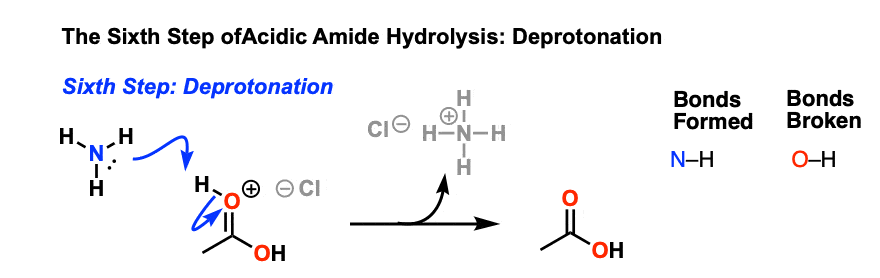
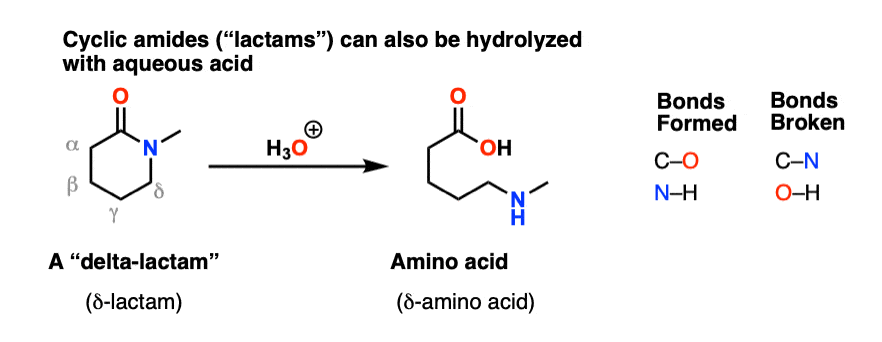
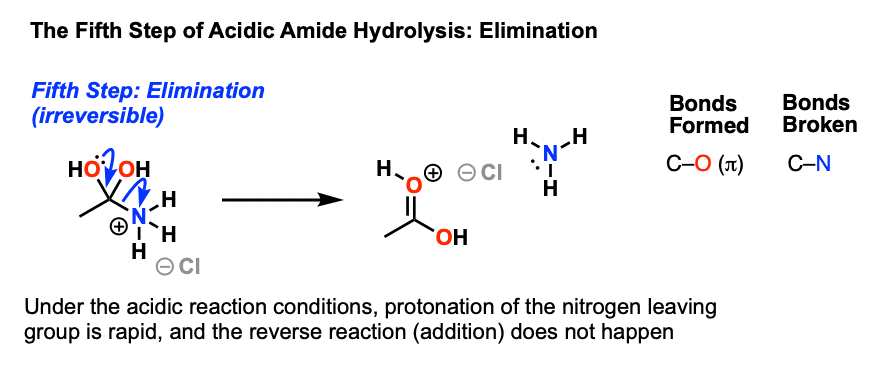
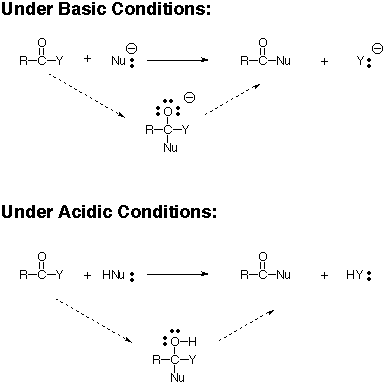
Amides can be hydrolyzed into a carboxylic acid and ammonia or an amine by heating in an acidic or basic aqueous solution.
In both cases, acid-base reactions occurring with the products make the overall reaction irreversible.
What is the mechanism of amide hydrolysis in basic conditions?
In a basic medium, amide interacts with the water molecule to give a carboxylic acid and the salt of ammonia or amine salt.
Mechanism: In base catalysed amide hydrolysis, the amide is heated with boiling aqueous NaOH or KOH.
The nucleophilic hydroxide ion adds to the carbonyl carbon to form a tetrahedral intermediate.
What are the basic conditions for amide?
Amides undergo many chemical reactions, although they are less reactive than esters.
Amides hydrolyse in hot alkali as well as in strong acidic conditions.
Acidic conditions yield the carboxylic acid and the ammonium ion while basic hydrolysis yield the carboxylate ion and ammonia.
|
A mild alkaline hydrolysis of N- and NN-substituted amides and
In general nitriles and amides are exceptionally stable to acid and basic hydrolysis and classically they are hydrolyzed under vigorous reaction conditions |
|
On the hydrolysis mechanisms of amides and peptides
Apr 20 2018 H2O ? RCOOH + R?NH2) at moderate pH conditions at ... enly claiming that |
|
Theoretical Study of Base-Catalyzed Amide Hydrolysis: Gas-and
doubly negatively charged intermediates which expel the amine in its deprotonated form.710 |
|
Base-promoted hydrolysis of amides at ambient temperatures
ed with the same enzyme system under the same conditions as expected |
|
Models of zinc-containing proteases. Catalysis of cobalt(III
Abstract: The rate of intramolecular amide hydrolysis in substitutionally inert Co(III) complexes has acidic neutral |
|
Theoretical Studies of Hydrolysis and Stability of Polyacrylamide
Both acid and base can catalyze the hydrolysis reaction by lowering amide hydrolysis under different conditions have been extensively studied from both. |
|
Study on Hydrolysis Properties and Mechanism of Poly(3
Jul 10 2022 had better hydrolysis stability under the same conditions. ... when the amide was hydrolyzed under alkaline conditions. |
|
Study on Hydrolysis Properties and Mechanism of Poly(3
Jul 10 2022 had better hydrolysis stability under the same conditions. ... when the amide was hydrolyzed under alkaline conditions. |
|
Reactions of the carboxamide group
B. Alkylation under Basic Conditions Reactions of the carbosamide group ... czrbonyl reactivity and the nucleophilic properties of the amide group. |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of hydrolysis of amides under acidic and basic reaction conditions We have seen that carboxylic acid derivatives usually react through nucleophilic addition- elimination mechanisms The parent carboxylic acid can be obtained from all its derivatives by this mechanism through a hydrolysis process |
|
On the hydrolysis mechanisms of amides and peptides
15 mai 2018 · H2O → RCOOH + R′NH2) at moderate pH conditions at which kineticists apply a rate posed that the slope of +1/2 may be real, and that the under- enly claiming that, for amide hydrolysis in dilute alkaline con- ditions (pH |
|
HYDROLYSIS
general, hydrolysis products predominant under neutral conditions, whereas SN2 reaction, where the phosphate ester anion is acting as the leaving group |
|
Carboxylic Compounds, Nitriles, and Their Interconversion
a normal carboxylic acid ester B under mild conditions (mechanism: Figure 9 13) The base-mediated partial hydrolysis of a nitrile → a primary amide is |