composition of seawater pdf
|
Chemistry of Sea water
composition of sea water Dittmar (1884) made careful determinations on 77 water samples representative of all oceans which had been collected on the voyage around the world of H M S Challenger He determined the halides sulphate magnesium calcium and potassium On composite samples |
|
Lecture 12
The composition of average seawater and river water in mmol kg-1 Element Na Mg Ca K Sr Cl SO4 HCO3 Br Seawater (mmol kg-1) 468 0 |
|
COMPOSITION OF SEAWATER
1 COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances Salinity is a measure of the amount of dissolved salts in seawater measured in parts per thousand (ppt) To change the salinity of an ocean three different things can occur: evaporation precipitation or freezing |
|
Lecture 4: Major Ions Conservative Elements and Dissolved
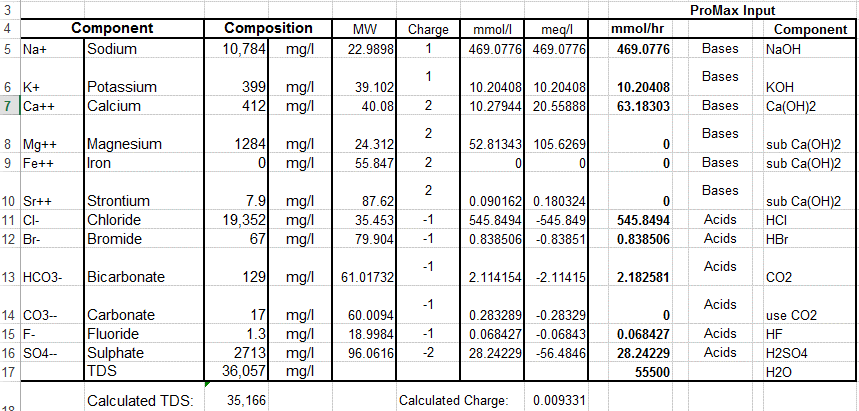
11 major ions all other elements in seawater are present in concentrations less than 1ppm (1mg/kg seawater) at S = 35 000 (from Pilson) Ion Formula g/Kg mmol/Kg Sodium Na+ 10 781 468 96 Magnesium Mg2+ 1 284 |
|
Seawater: Composition and Properties 3
Seawater contains an abundance of organic and inorganic constituents These are present in dissolved suspended very finely distributed or colloidal form The dissolved constituents of seawater can be assigned to the following main categories: Inorganic main constituents that form the salt content of seawater with its cationic and anionic compone |
What are the saline components of seawater?
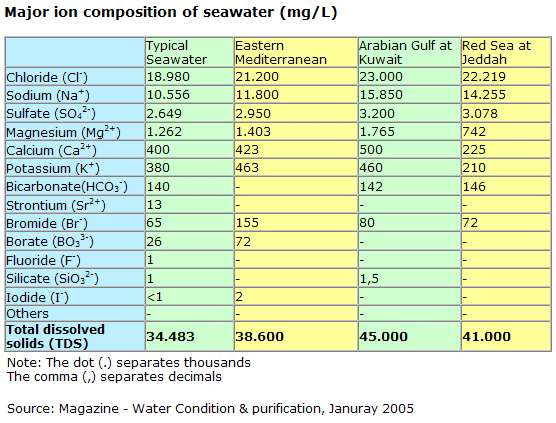
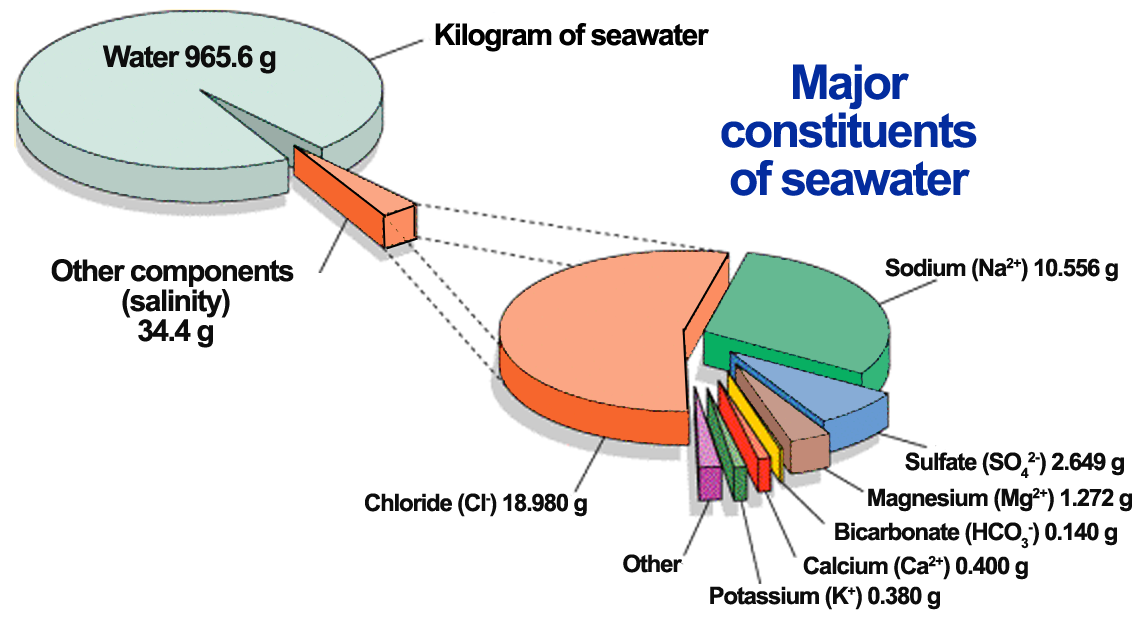
These saline components account for over 99% of the total salt content of seawater (see Fig. 3.1). The remaining around 1% of salt content consists mainly of the cations strontium and barium and the anions bicarbonate, carbonate, bromide, borate, uoride, and nitrate.
What elements are found in seawater?
All naturally occurring elements on Earth have been found in seawater, both in high concentrations, such as in the main constituents of seawater salt content, and also in very low and tiny concentrations in trace substances in seawater. The salt content in the Earth’s seas and oceans differs very widely (see Table 3.1).
How much salt is in seawater?
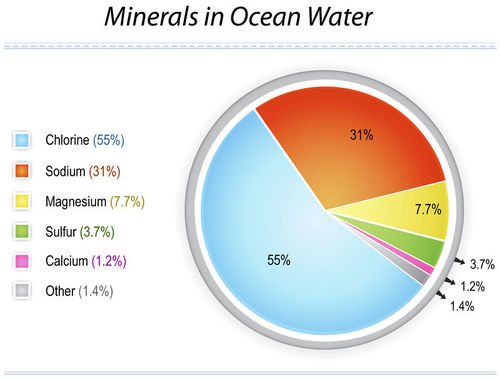
42 g/kg in the Red Sea and sometimes even up to 49 g/kg in the Arabian Gulf. The salt contained in seawater consists mainly of six saline components, namely, the cations sodium, magnesium, calcium, and potassium and the anions chloride and sulphate. These saline components account for over 99% of the total salt content of seawater (see Fig. 3.1).
3.1.1 Composition
Seawater contains an abundance of organic and inorganic constituents. These are present in dissolved, suspended, very finely distributed, or colloidal form. The dissolved constituents of seawater can be assigned to the following main categories: Inorganic main constituents that form the salt content of seawater with its cationic and anionic compone
Chlorinity and Salinity
The salt content (S) of seawater is de ned as the quantity of dissolved inorganic fi substance per kilogram of seawater, i.e. the mass proportion of dissolved inorganic substance. The salt content includes not only ionogenic but also neutral undissoci-ated and weakly dissociated inorganic components. However, it does not include dissolved organic s
3 :2 ð Þ
Chlorinity is measured by Knudsen-Mohr titration of a seawater sample with silver nitrate solution. This determines all the halogenides present in the sample, i.e. not only chloride but also the concentrations of bromide and iodide (of which there is a negligibly small concentration in seawater): Agþ seawater AgCl AgBr AgJ þ ð Þ þ S ð Þ þ s s ð Þ
(K or K0
sp sp ) characterizes the solubility limit of a low-solubility compound in a solution. With knowledge of the activity coef ficients of the components involved in the reaction and with knowledge of the value of the stoichiometric solubility product (K sp), the thermodynamic solubility product (K0 sp) can be calculated as link.springer.com
ADH
ffiffiffiffiffi Im bi BDH ai p ffiffiffiffiffi Im þ þ link.springer.com
Φ0 a,a0
E-parameter equations for single anion activity coefcient ( γX): fi Nc link.springer.com
|
Chemical composition of seawater; Salinity and the major constituents
Note: practical salinity is unit-less and is not a SI quantity! Salinity Measurement – The Past. Standard Mean Ocean Water (SMOW): S ≈ 35 |
|
Rohling EJ (2013) Oxygen Isotope Composition of Seawater
2 lip 2013 Rohling E.J. (2013) Oxygen Isotope Composition of Seawater. In: Elias S.A. (ed.) The Encyclopedia of Quaternary Science vol. 2 |
|
Seawater composition
Although officially |
|
Chemical composition of seawater; Major constituents
24 sty 2013 Note: practical salinity is unit-less and is not a SI quantity! Salinity Measurement – The Past. Standard Mean Ocean Water (SMOW): S ≈ 35 |
|
Optical variability of seawater in relation to particle concentration
25 sie 2010 composition and size distribution in the nearshore marine environment at Imperial. Beach |
|
Chemical composition of seawater; Salinity and the major constituents
Molar units. 1. Molarity = moles per litre of solution = M. • Commonly used terms include: mM = millimolar = millimoles per litre = 10-3 moles per litre. |
|
Neoproterozoic variations in the C-isotopic composition of seawater
The recent proliferation of stratigraphic studies of 13C variation in carbonates and organic C in later Neopro- terozoic and basal Cambrian successions |
|
Variation in the strontium isotopic composition of seawater (8 Ma to
Measurements of 87Sr/86Sr on samples of planktonic foraminifers were used to reconstruct changes in the Sr isotopic composition of seawater for the past 8 |
|
The Chemical Composition of Seawater
The Chemical Composition of. Seawater. Winn Johnson. 25 August 2015. Regional Maritime University. Page 2. Seawater Chemistry Supports: 35 grams of salt / ... |
|
%/8%r composition of seawater during the Phanerozoic
Ab&&--*7Sr/s6Sr measurements of 108 sedimentary carbonate rocks have been used to trace variations in the strontium isotopic composition of seawater during |
|
Chemical composition of seawater; Salinity and the major constituents
Salt dissolved in ocean water alters the properties of water. Freezing point of seawater is ~ -1.8?C. Density continually increases to freezing point |
|
Chemical composition of seawater; Major constituents
Chemical composition of seawater;. Major constituents. OCN 623 – Chemical Oceanography. 1/24/2013. Reading. Libes Chapters 2 - 4. |
|
Seawater composition
the thermodynamic properties of seawater are directly influenced by the mass of dissolved constituents (i.e. Absolute Salinity) whereas Practical Salinity |
|
Chemical composition of seawater; Salinity and the major constituents
Salt dissolved in ocean water alters the properties of water. Freezing point of seawater is ~ -1.8?C. Density continually increases to freezing point |
|
The Properties of Seawater
Berg Random Walks in Biology |
|
1 Chpt 4: Major Ions of Seawater James Murray (10/01/04) Univ
Oct 1 2004 At a salinity of S = 35.000 seawater has the composition given in Table 4-1 (from Pilson |
|
Rohling EJ (2013) Oxygen Isotope Composition of Seawater
Both text and figures draw heavily on the water-based oxygen isotope part of a previously published essay on stable oxygen and carbon isotopes in Foraminifera ( |
|
2017 MIT Seawater Properties Table r2
Zubair "Thermophysical properties of seawater: A review of existing correlations and data |
|
The International Association for the Properties of Water and Steam
Sept 7 2008 Thermodynamic Properties of Seawater ... chemical composition of sea salt is very similar in all oceans |
|
Boron and magnesium isotopic composition of seawater
Aug 18 2010 environmental variables and processes. The isotopic composition of seawater for both systems plays a central role in these studies and is an ... |
|
Chemistry of Sea water - ResearchGate
In table 33 the composition is shown by referring the substances to a standard concentration, Cl = 19 00 '/00, and by means of the ratios I between the different |
|
CHEMICAL COMPOSITION OF SEAWATER AND THE VARIATION
the ionic composition of seawater is calculated The Wattenberg equation for the estima- tion of calcium in seawater is corrected according to the recent values of |
|
Chemical composition of seawater; Major constituents - SOEST Hawaii
Chemical composition of seawater; Major constituents OCN 623 – Chemical Oceanography 1/24/2013 Reading Libes, Chapters 2 - 4 Can skim section 4 3 |
|
The Chemical Composition of Seawater - WordPresscom
The Chemical Composition of Seawater Winn Johnson 25 August 2015 Seawater Chemistry Supports: • ½ O 2 F- 0 07 35 grams of salt / liter of seawater |
|
On the Composition of Sea-Water in the Different Parts of the - JStor
occurs in the organic substances and in the ammonia which are dissolved in sea- water 3 Chlorine -Next to the elements of water chlorine is the element which |
|
General Chemistry of Seawater - Encyclopedia of Life Support
And now, the oceans cover 70 8 per cent of the surface of the Earth with the seawater composition remain unchanged for thousands of years prior to the present |
|
The Properties of Seawater - Jones & Bartlett Learning
However, despite its simple chemical composition, water is a complex substance with truly remarkable physical properties For example, the melting and boiling |
|
1 Chpt 4: Major Ions of Seawater James Murray (10/01/04) Univ
1 oct 2004 · Major ions are defined as those elements whose seawater concentration is has the composition given in Table 4-1 (from Pilson, 1998) |
|
Seawater composition
Although, officially, chlorinity superseded salinity to describe the salt content of seawater, the relationship obtained by Knudsen was still being (erroneously) used |
|
INTRODUCTION TO CO2 CHEMISTRY IN SEA WATER
Will this be the same all over the oceans? Why? • How should I modify the CO2 composition of a sample of sea water to reach a desired target composition? |