inductive effect chemistry definition
What is the inductive effect of a sigma atom?
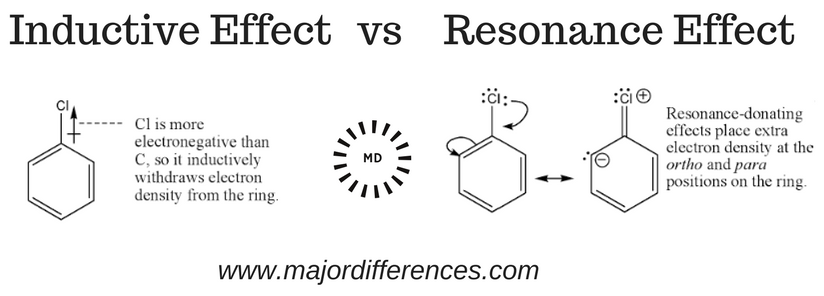
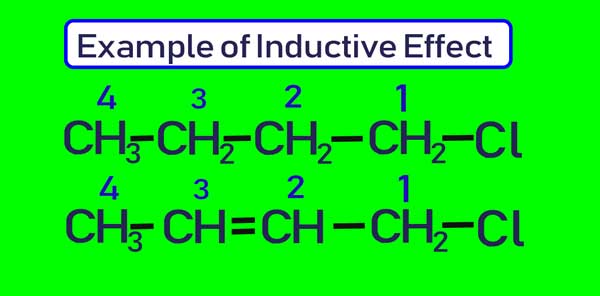
The effect of the sigma electron displacement towards the more electronegative atom by which one end becomes positively charged and the other end negatively charged is known as the inductive effect. The - I effect is a permanent effect & generally represented by an arrow on the bond. [citation needed]
What is the difference between inductive effect and resonance?
Rather, the electrons are distributed evenly across the atoms, forming intermediate bonds between single and double bonds. The inductive effect and resonance both relate to the distribution of electrons in a chemical bond, but are two different and distinct effects.
What is the difference between electron displacement and inductive effect?
An electron displacement effect is an effect that electrons displace from their position. Inductive effect is a partial shifting/displacement of σ electrons towards a more electronegative atom of σ bond. This means σ (or single bond electrons) shifts towards a more electronegative atom. Because of this, partial charges develop.
What is inductive effect?
Inductive effect is a partial shifting/displacement of σ electrons towards a more electronegative atom of σ bond. This means σ (or single bond electrons) shifts towards a more electronegative atom. Because of this, partial charges develop. We can see a σ-bond between C and X, both sharing one electron.

The Inductive Effect Electronic Effects Chemistry Khan Academy

Inductive Effect Organic Chemistry

Inductive Effect
|
RESONANCE AND INDUCTION TUTORIAL Jack DeRuiter The
Induction or the inductive effect of an atom or functional group is a Resonance may be defined as bonding or sharing of electrons between more than two. |
|
Inductive effect is defined as - permanent displacement of shared
CONCEPTS IN ORGANIC CHEMISTRY. • Inductive Effect Positive Inductive effect : (+I effect Electron ... Electromeric effect is defined as the complete. |
|
Inductive and resonance effects based on coreâ•electron binding
Department of Chemistry State University of Campinas |
|
Inductive Effects
The terms "resonance" and "induction" refer to the electronic effects that atoms or functional groups may have within a compound. These effects are defined |
|
Chemical shifts
inductive effects present in a molecule reduce the electron density in the hydrogen 1s orbital dishielding. (shift to higher frequencies) is expected. |
|
ORGANIC CHEMISTRY – SOME BASIC PRINCIPLES AND
separate branch of chemistry called organic chemistry. The functional group may be defined as an ... ?-bond is referred to as the inductive effect. |
|
Fundamentals of Organic Chemistry CHAPTER 1. INTRODUCTION
2 mai 2020 Know dipole moment & inductive effect in chemical compounds. ? Classify the organic compounds according to functional groups. ? Define the ... |
|
The inductive effect: theory and quantitative assessment
Institute of Organic Chemistry and Biochemistry Academy of Sciences of the Czech ABSTRACT: The concept of the inductive effect is defined in terms of ... |
|
A Quantum Theory Atoms in Molecules Study about the Inductive
9 avr. 2020 The inductive effect concept is fundamental in organic chemistry and has been used to rationalize trends observed due to the presence of ... |
|
The Inductive Effect and Chemical Reactivity. I. General Theory of
The Inductive Effect and Chemical Reactivity. 2263. The corrected mean heat of combustion of tita- nium nitride is 2337.36 cal. per gram for the sample. |
|
Inductive Effect
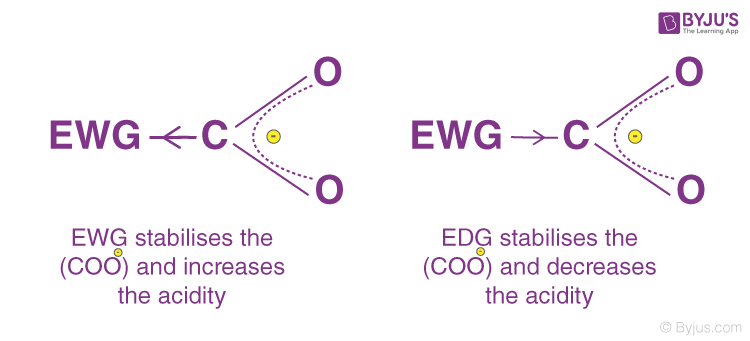
Following are some examples of groups in decreasing order of their -I effect Page 5 2 Positive inductive effect (+I effect) • When a chemical |
|
Inductive Effects
These effects are defined below and are dependent on the valence, bonding Induction or the inductive effect of an atom or functional group is a function of that groups 1) electronegativity, 2) derived from amide chemistry Typically amides |
|
Inductive Effect - Womens College Samastipur
Inductive Effect Definition : The partial displacement of sigma electron pairs towards more The physical and chemical properties of compounds is controlled Positive Inductive Effect (+I effect) : Electron donating substituents tend to |
|
INDUCTIVE EFFECT - satyapsingh
It influences the chemical and physical properties of compounds ILLUSTRATION OF For example, in cases of halogens, the negative inductive effect is more |
|
PDF :8 - BSc Chemistry
Module No and Title Module 1: Revisit to Electronic Effects: Inductive and For example, C - X (X = halogen) bond is polar with C having a δ+ charge and the |
|
Research Article Topological Model on the Inductive Effect - CORE
19 fév 2014 · effects in chemistry [1] According to the International Union of Pure and Applied Chemistry (IUPAC) the inductive effect is defined as |
|
Organic Chemistry - MES Asmabi College
Electron Displacement Effects: Inductive effect: Definition - Characteristics - +I and -I groups Applications: Explanation of substituent effect on the acidity of |
|
Chapter 1: Introduction - University of Arizona
define explicit features as ―fully visible elements of a given chemical positive inductive effect decreases acid strength because electron density is transmitted |