concept of free energy pdf
|
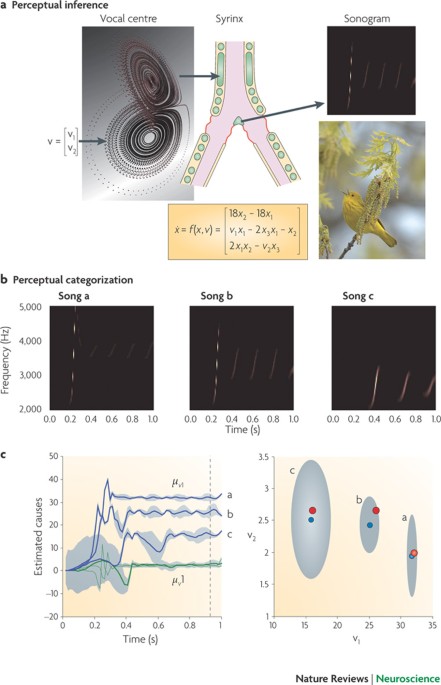
The free-energy principle: a rough guide to the brain?
The free-energy principle is an attempt to explain the structure and function of the brain starting from the very fact that we exist: this fact places constraints on our interactions with the world which have been studied for years in evolutionary biology and systems theory |
|
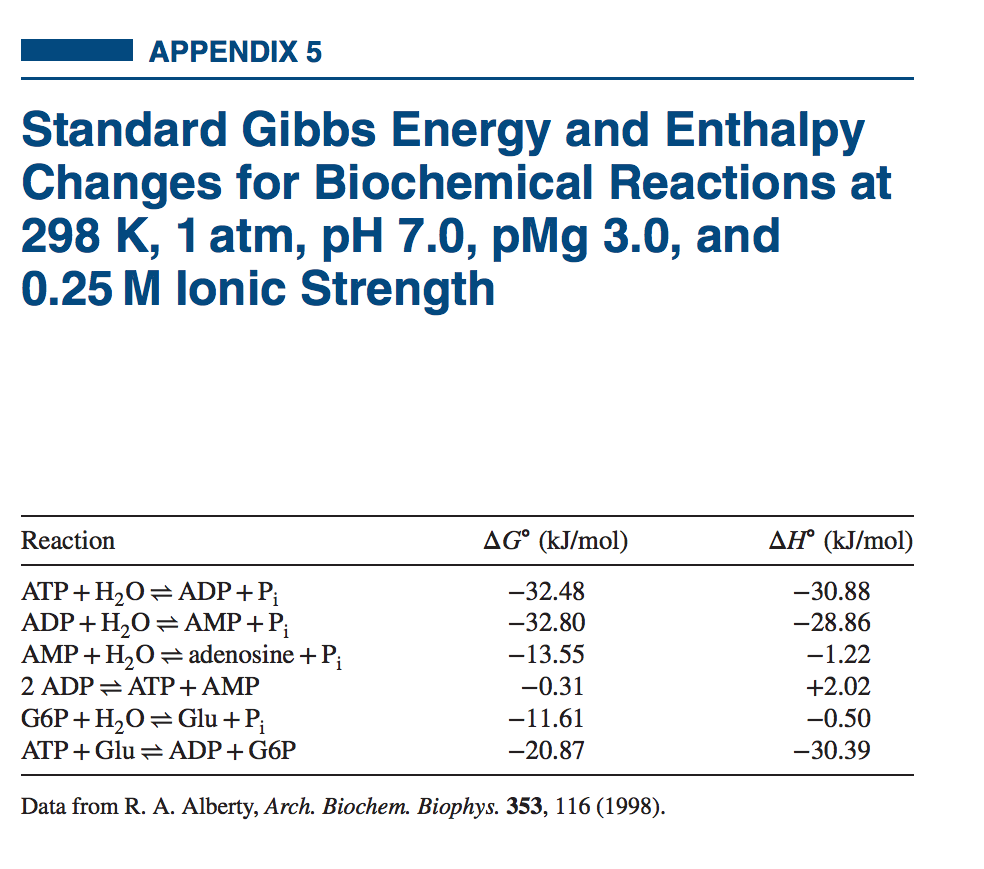
PRINCIPLES OF CHEMICAL THERMODYNAMICS
1 When a reaction proceeds spontaneously with the release of free energy ∆G has a negative value and the reaction is said to be exergonic A reaction with a negative ∆G is called thermodynamically stable 2 In endergonic reactions the system gains free energy and ∆Gis positive |
|
Chapter 18 Entropy Free Energy and Equilibrium
The term “free” meanes that Gibbs free energy is an amount of energy of a molecular system which can be used for work (exchanged from a heat to mechanical work or in an opposite direction) The rest of the energy which can not be convered into work is related to a molecular dissorder (entropy) G = H - TS If a reaction is spontaneous DS |
|
Free Energy Exergy and Energy: The Exergetic Content of Energy
One of its important results was the concept of free energies which provided the general idea of available energies with specific examples — especially the Gibbs free energy of electrochemical energy!electrical work Further development of available energy has evolved into the theory of exergy |
|
Lecture 8: Free energy
Lecture 8: Free energy Matthew Schwartz Statistical Mechanics Spring 2019 Lecture 8: Free energy Introduction Using our various ensembles we were led to a rather simple di erential relation between energy and other state variables Eq (55) of Lecture 7: dE = TdS ¡ PdV + dN (1) |
Why is free energy called free energy?
Then the free energy change (at constant T) is: where the inequality becomes an equality if and only if the expansion is done reversibly. So we see that when work is done at constant temperature, the free energy of the system is depleted to do the work. This is why free energy is called free: it is the energy available to do work.
Is there a grand free energy?
There is one more free energy that is used sometimes, called the grand free energy . We're not going to use it until Lecture 11, because it's harder to interpret physically, but I include the releant formulas here since they are closely related to the other free energies from this lecture.
How do you calculate free energy in a mechanical system?
Mechanical systems all xed degrees of freedom, so S = 0 and F = E. Free energy is a generalization of energy whose importance is revealed by working at nite T. Suppose we have some system in thermal contact with a heat bath, and we want the system to do work by pushing a piston (which in turn can lift a weight, or whatever).
How do you calculate Gibbs free energy?
So the total Gibbs free energy on the mixed side, generalizing Eq. (63), is w(T ; P) Gw = is the chemical potential of the pure solvent. Nw Equilibrium requires the chemical potential of the solvent to be the same on both sides of the barrier. So Ns w(T ; Ppure) = mixed(T = Pmixed Ppure. In ¡ This is known as van 't Ho 's formula.

Gibbs free energy introduction Biomolecules MCAT Khan Academy

Bioenergetics biochemistry concept of free energy Exergonic and endergonic redox potential

The Laws of Thermodynamics Entropy and Gibbs Free Energy
|
Lecture 8: Free energy
the second term is work from expanding volume |
|
The free-energy principle: a unified brain theory? (PDF)
13 janv. 2010 Entropy is therefore a measure of uncertainty. The free-energy principle: a unified brain theory? Karl Friston. Abstract |
|
Free Energy: Alternative Designs for Awareness and Choice
As design examples the Free Energy concepts act to stimulate reflection and reaction – rather than product designs |
|
Chapter 14 (and 15.4): Entropy and Free Energy
Nature tends to move towards states corresponding to an increase in entropy. Quantitative definition of entropy (Boltzmann). S = k lnW o k = Boltzmann constant |
|
Sans titre
27 oct. 2011 In thermodynamics the Gibbs free energy (IUPAC recommended name: Gibbs energy ... where potential energy is defined as capacity to do work |
|
Variational Characterization of Free Energy: Theory and Algorithms
20 nov. 2017 We revisit the well-known Jarzynski equality for nonequilibrium free energy sampling within the framework of importance sampling and Girsanov ... |
| Rényi Entropy and Free Energy |
|
The free-energy principle: a rough guide to the brain? [pdf]
Free-energy: an information theory measure that bounds (is greater than) the surprise on sampling some data given a generative model. Generalised coordinates: |
|
Theory of nonequilibrium free energy transduction by molecular
19 sept. 2019 understanding free energy transduction in molecular machines. ... and thermodynamic concepts that figure prominently in such models. |
|
Free Energy - Biblioteca Cejamericas
Free Energy: Alternative Designs for Awareness and Choice 'Free Energy' consists of conceptual design examples – two of which are described in this paper |
|
Lecture 8: Free energy
3 Helmholtz Free Energy We de ne the Helmholtz free energy as F E ¡TS (22) Free energy is a concept particularly useful at constant temperature We can |
|
The Free Energy Generator - International Journal of Scientific and
This concept of generating magnetic field from the permanent magnets became practical only after introducing Neodymium magnets which are much power full |
|
Gibbs Free Energy
27 jan 2010 · How Gibbs Free Energy can be used to determine the direction of a reaction • The meaning and significance of the “mass action ratio” |
|
Free energy generator project report pdf - Squarespace
Free energy generator project report pdf of such a system and to demonstrate the feasibility of the concept of solar magnetohydromynamic energy production |
|
Entropy, free energy - WIT Press
The laws of thermodynamics have a universality of relevance; they encompass widely diverse fields of study that include biology Moreover the concept of |
|
CHAPTER TWO FREE ENERGY CONCEPT - SUST Repository
FREE ENERGY CONCEPT AND LITERATURE REVIEW 2 1 Introduction At this point in time - the early years of the twenty-first century we have reached the |
|
Chemical Thermodynamics SYBSc Concept of Gibbs free energy
2) As entropy, temperature and internal energy are state functions, Helmholtz free energy is also a state function 3) For any chemical reaction, the total internal |
|
Electrostatic Free Energy and Other Properties of States - CORE
Thus, for some brief time after the absorption act, the electrical polarization of the system will not be in equilibrium with the charge distribution This concept |