condensation polymerisation gcse
Is polyester a condensation polymer?
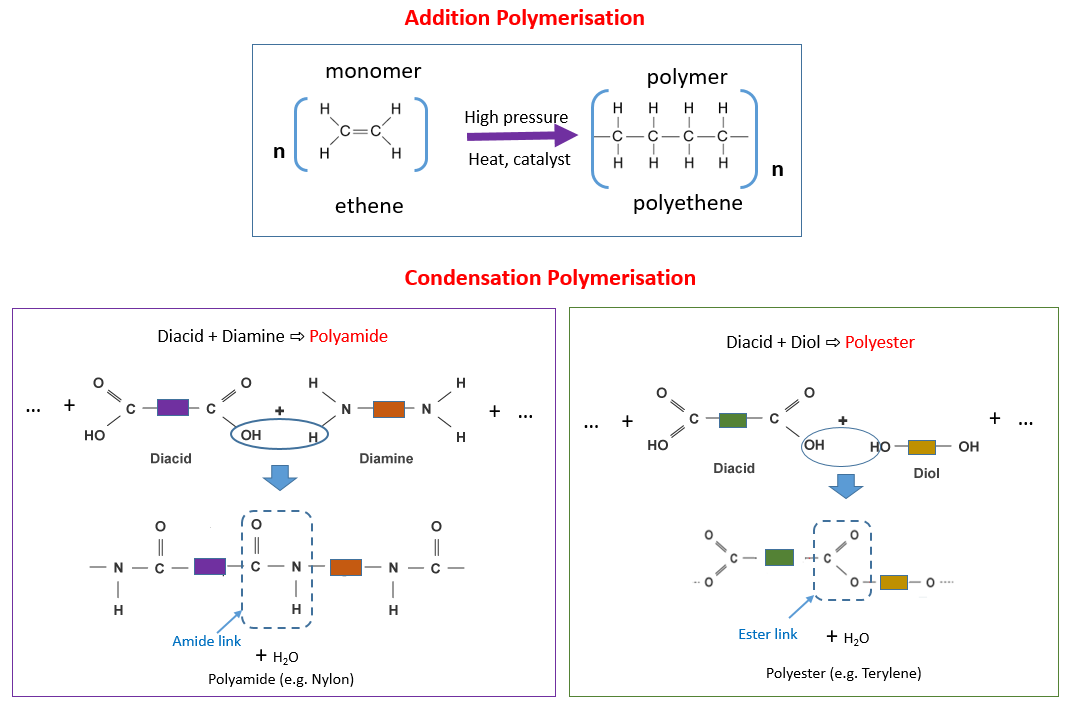
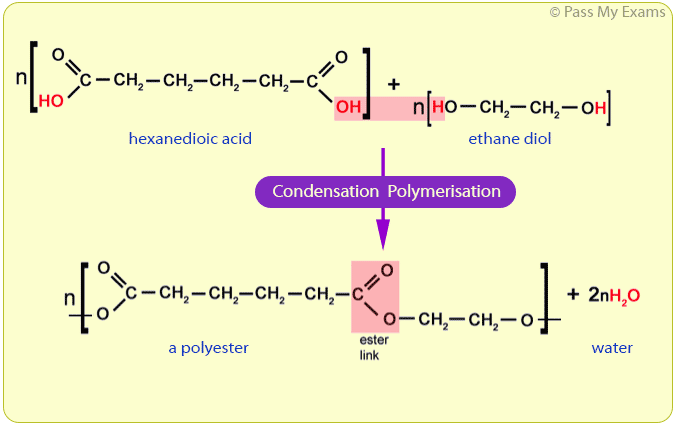
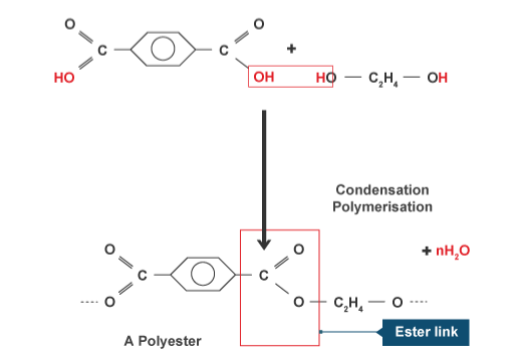
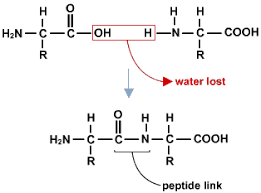
Polyester is a condensation polymer. An example of a condensation polymer is polyester, which is formed from ethane diol and hexanedioic acid. Water is also formed in this reaction. The functional groups of the monomer stay the same. When polymerisation occurs, each repeating unit of the polymer contains a functional group from each monomer.
What is a repeating unit in condensation polymerisation?
In the process of condensation polymerisation, two monomers with different functional groups are used as the repeating unit. Each monomer has the same respective functional group on either end of the molecule. The repeating unit of the polymer will have the same atoms as the monomer. Two products are formed.
What is condensation polymerisation?
In condensation polymerisation, monomers will join together to form polymers. This means that condensation polymerisation is another type of polymerisation. Two different monomers are repeated. In the process of condensation polymerisation, two monomers with different functional groups are used as the repeating unit.
What happens when Polymerisation occurs?
When polymerisation occurs, each repeating unit of the polymer contains a functional group from each monomer. Worked example: Draw the two monomers which have been used to produce the condensation polymer, nylon. Identify where both molecules join together. This will be where the label “amide link” occurs. Separate the molecules where they join.

GCSE Chemistry

Condensation Polymerisation

s=
|
C11.2 Student calculation sheet
C7.3.2 Condensation polymerisation. • C7.3.3 Amino acids. • C7.3.4 DNA (deoxyribonucleic acid) and other naturally occurring polymers. |
|
Condensation Polymerisation Addition Polymerisation Amino acids
Condensation polymerisation involves monomers with two functional groups. When these types of monomers react they join together and usually lose small molecules |
|
Knowledge check
Polymerisation type 1. Polymerisation type 2. Page 3. Organic chemistry in context: © Royal Society of Chemistry. Condensation polymerisation (H) edu.rsc.org. |
|
Untitled
addition polymers and condensation polymers. https://www.savemyexams.co.uk/gcse-chemistry-edexcel-new/revision-notes/polymers/addition- polymerisation/. |
|
Cambridge IGCSE Chemistry Topic 14: Organic chemistry
Condensation polymerisation involves the reaction of two different functional groups to form one long molecule by the removal of a small molecule such as. |
|
1 There are two types of polymerisation addition and condensation
This polymer is made by addition polymerisation. Draw the structural formula of its monomer. [1]. (ii) The two monomers shown below form a nylon which is |
|
Edexcel GCSE Chemistry Topic 9: Separate chemistry 2 - Polymers
Why polyesters are condensation polymers: ? In condensation polymerisation a small molecule is formed as a by-product each time a bond is formed between |
|
Organic Chemistry Revision Questions – Paper 2 Triple only.
condensation polymerisation. • poly(ethene) produced from one monomer wheareas polyester produced from two different monomers. |
|
Edexcel GCSE Chemistry Topic 9 - Physics & Maths Tutor
Why polyesters are condensation polymers: ○ In condensation polymerisation, a small molecule is formed as a by-product each time a bond is formed between |
|
GCSE Chemistry Polymers AQA, OCR, Edexcel Questions
Condensation polymerisation Q6: Define condensation polymerisation (3 marks ) Q7: What must the two monomers have in common for the condensation |
|
GCSE Chemistry Polymers AQA, OCR, Edexcel Answers
Q6: Define condensation polymerisation A= Condensation polymerisation involves monomers with two functional groups (1 mark) When these monomers react |
|
Condensation polymers - Hartismere
These include the polyesters formed from the reaction of a diol with a dicarboxylic acid and the polypeptides formed from the condensation polymerisation of |
|
Condensation Polymerisation Addition Polymerisation Amino acids
Condensation polymerisation involves monomers with two functional groups When these types of monomers react they join together and usually lose small |
|
C11 Polymers Student Bookletpdf - Ark Victoria Academy
addition polymers and condensation polymers It is important https://www savemyexams co uk/gcse-chemistry-edexcel-new/revision-notes/polymers/ addition- |
|
AQA GCSE Organic chemistry 2 (CHEMISTRY ONLY) Reactions of
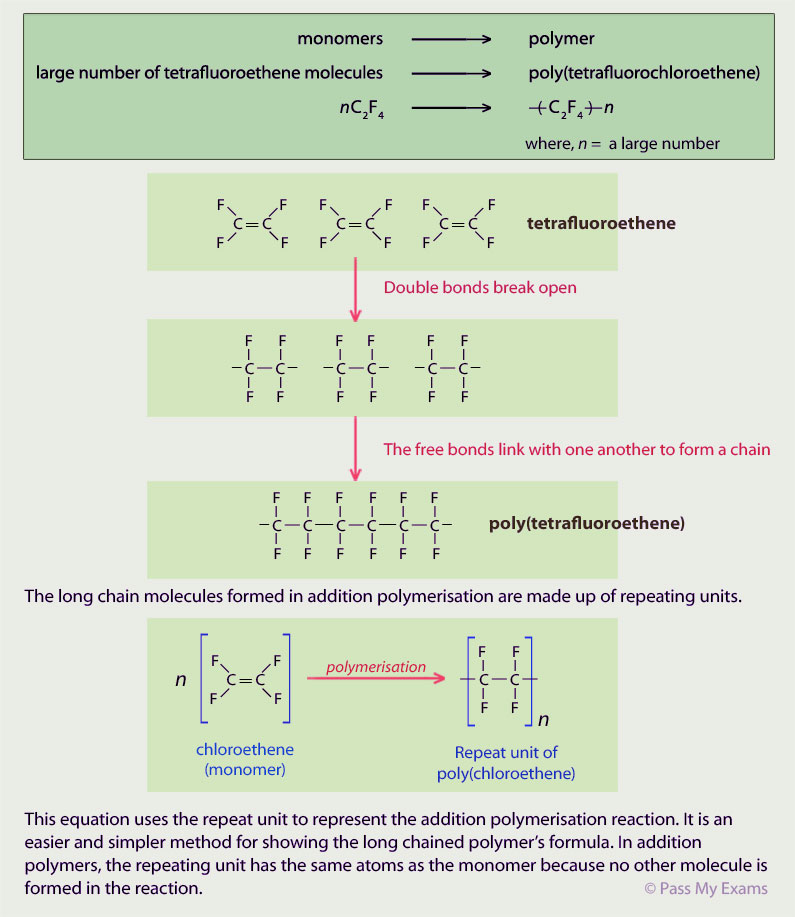
polymers In addition polymers, the repeating unit has the same atoms as the monomer It can be displayed like this: Condensation polymerisation (HT only) |
|
Condensation polymers - Pearson Schools and FE Colleges
Polyamides are condensation polymers, normally formed when a dicarboxylic acid reacts with a diamine, to form a polymer and water Nylon-6,6, used in synthetic |