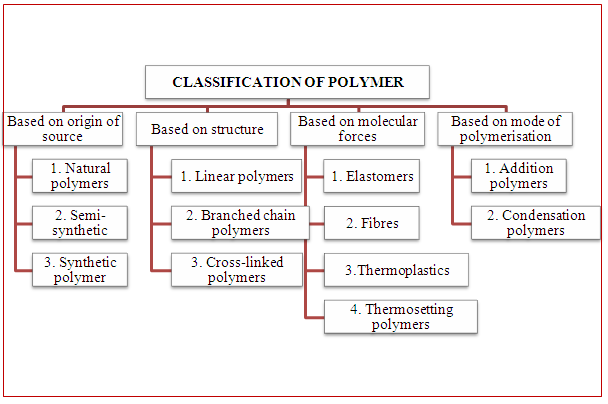
condensation polymerization notes
How many products are formed during a condensation polymerisation reaction?
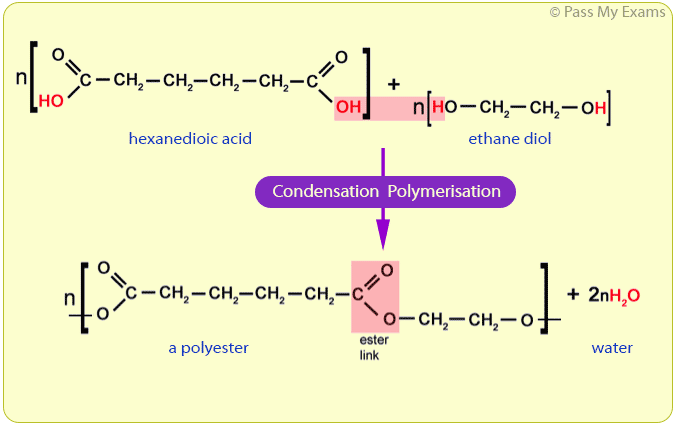
Two products are formed. At the end of the condensation polymerisation reaction, two products are formed. These are the polymer chain and water. The water is formed when the two monomers join together. Using a general equation. There is a general equation for the condensation polymerisation between diols and dicarboxylic acids.
What is a condensation polymer?
These polymerizations often (but not always) occur with loss of a small byproduct, such as water, and generally (but not always) combine two different components in an alternating structure. The polyester Dacron and the polyamide Nylon 66, shown here, are two examples of synthetic condensation polymers, also known as step-growth polymers.

GCSE Chemistry

Polymers

Condensation Polymerisation
|
Detailed Notes - Topic 3.12. Polymerisation - AQA Chemistry A-level
3 Mar 2012 Hydrolysis. Condensation polymers can be broken downback into the constituent molecules by the addition of H2. |
|
Cambridge IGCSE Chemistry Topic 14: Organic chemistry
Notes www.pmt.education. Page 2. Name some typical uses of plastics and of man (a polyester) by condensation polymerization. Formation of nylon: www.pmt ... |
|
National 5 Chemistry Unit 3: Chemistry in Society Plastics Summary
Summary Notes. Name. Page 2. 2 All other natural polymers are condensation polymers. The most common ... |
|
Condensation-polymers.pdf
Condensation polymerisation is therefore a reaction in which many monomers join together to form a polymer with the elimination of a smaller molecule such as |
|
1 3.12. Polymers N Goalby chemrevise.org
In condensation polymerisation there are two different monomers that add The two most common types of condensation polymers are polyesters and polyamides ... |
|
Synthetic polymers - Edexcel IGCSE Chemistry Topic 4: Organic
Notes www.pmt.education. Page 2. 4.44 know that an addition polymer ○ Incondensationpolymerisationasmallmoleculeisformedasa ... |
|
AQA GCSE Chemistry Topic 7: Organic chemistry
Notes. (Content in bold is for Higher Tier only) www.pmt.education. Page 2 ○ They react by condensation polymerisation to produce polypeptides (works the. |
|
Topic 13
condensation polymers because they are the product of condensation reactions. iii) Properties and uses of condensation polymers. Condensation polymers tend ... |
|
Notes - Topic 4.8 Organic Synthesis - WJEC (Wales) Chemistry A-level
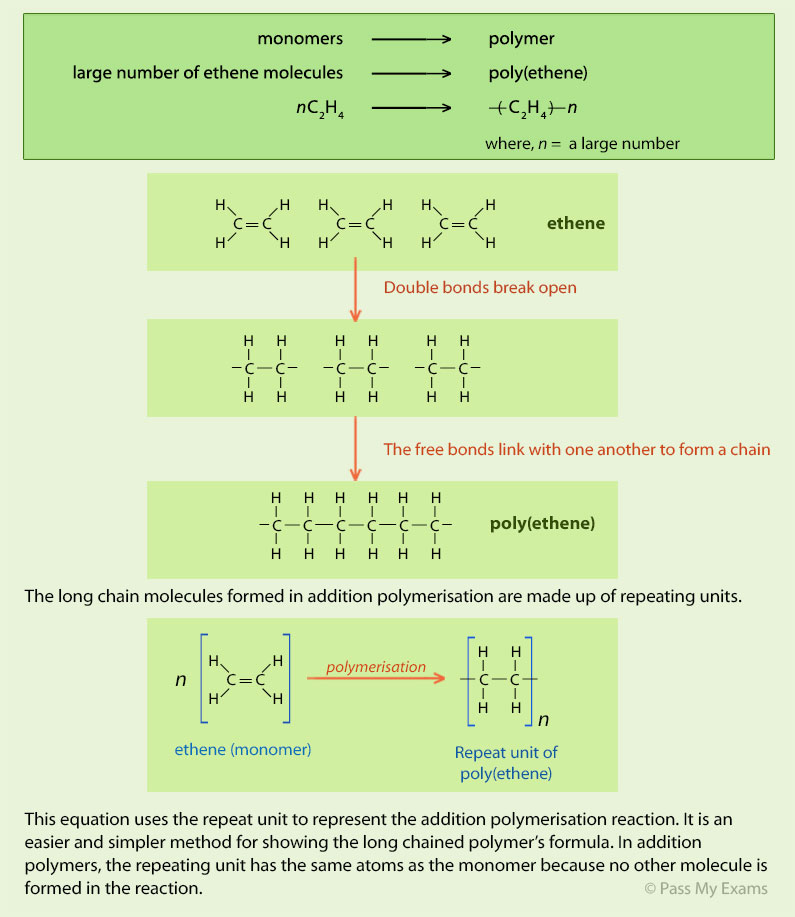
There are two main types of polymer: condensationpolymers and additionpolymers. Addition Polymers. Addition polymers are produced from alkeneswhere the |
|
Edexcel GCSE Chemistry Topic 9: Separate chemistry 2 - Polymers
Notes www.pmt.education. Page 2. 9.17C Recall that… ○ A polymer is a substance of ○ In condensation polymerisation a small molecule is formed as a by ... |
|
STEP-GROWTH (CONDENSATION) POLYMERIZATION
May 13 2020 Examples of polymers prepared by condensation reaction are Kevlar |
|
Cambridge IGCSE Chemistry Topic 14: Organic chemistry
Topic 14: Organic chemistry. Synthetic polymers. Notes www.pmt.education Condensation polymerisation involves the reaction of two different functional. |
|
Detailed Notes - Topic 3.12. Polymerisation - AQA Chemistry A-level
Mar 3 2012 Hydrolysis. Condensation polymers can be ?broken down?back into the constituent molecules by the addition of H?2?. |
|
CIE Chemistry A Level 21 : Polymerisation
CIE Chemistry A Level. 21 : Polymerisation. (A Level only). Notes Condensation polymerisation is the joining together of ?monomers?to form a polymer ... |
|
Polymers
or chain growth polymerisation and condensation or step growth polymerisation. In this type of polymerisation the molecules of the same monomer or. |
|
Untitled
addition polymers and condensation polymers. https://www.savemyexams.co.uk/gcse-chemistry-edexcel-new/revision-notes/polymers/addition- polymerisation/. |
|
Lecture notes on Structure and Properties of Engineering Polymers
Polymerization Reactions and Techniques. Chain-growth polymerization or addition polymerization. Step-growth polymerization or condensation polymerization. |
|
Organic Chemistry: Polymerization Reactions
Condensation Polymerization. ? Monomers combine to form a polymer and a bi-product. Each time a bond forms between monomers small molecules |
|
Organic Chemistry: Polymerization Reactions
Condensation Polymerization. ? Monomers combine to form a polymer and a bi-product. Each time a bond forms between monomers small molecules |
|
National 5 Chemistry Unit 3: Chemistry in Society Plastics Summary
14 draw the structure of a condensation polymer given the monomer structures and vice versa. 15 identify the repeating unit in condensation polymers. |
|
Polymerization PDF
The second type of polymerization reaction produces condensation polymers, which are formed when monomers are linked together with the release of a smaller molecule, such as water or hydrogen chloride In all cases, when polymers are synthesized, the result is a mixture of long chain molecules of varying lengths |
|
Synthetic polymers - Cambridge IGCSE Chemistry Topic 14: Organic
Topic 14: Organic chemistry Synthetic polymers Notes www pmt education Condensation polymerisation involves the reaction of two different functional |
|
Detailed Notes - Topic 312 Polymerisation - AQA Chemistry A-level
3 mar 2012 · Hydrolysis Condensation polymers can be broken downback into the constituent molecules by the addition of H2 O This is because |
|
Polymers Polymers - NCERT
The polymers made by addition polymerisation from two different monomers are termed as copolymers, e g , Buna-S, Buna-N, etc 2 Condensation polymers The |
|
Lecture notes on Structure and Properties of Engineering Polymers
Polymerization Reactions and Techniques Chain-growth polymerization or addition polymerization Step-growth polymerization or condensation polymerization |
|
Condensation polymers - Pearson Schools and FE Colleges
Polyamides are condensation polymers, normally formed when a dicarboxylic acid reacts with a diamine, to form a polymer and water Nylon-6,6, used in synthetic |
|
Lecture36: Introduction To Polymerization Technology - NPTEL
The polymer formed after the reaction (in this case, ethyl benzoate) is called the condensation polymer Page 7 NPTEL – Chemical – Chemical Technology II |
|
MODULE-1 - VSSUT
→ Natural and synthetic polymers → Addition and condensation polymers → Homochain and Heterochain polymer → Organic and inorganic polymers and |