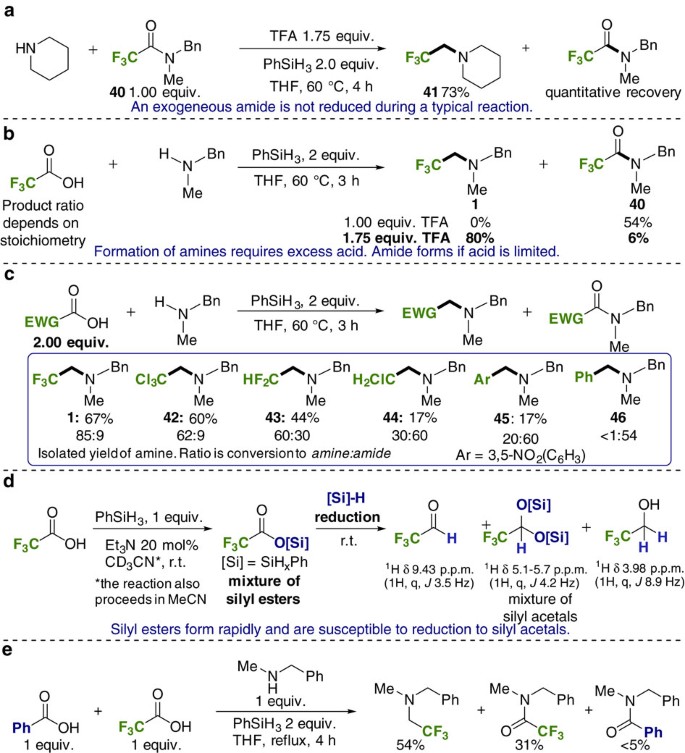
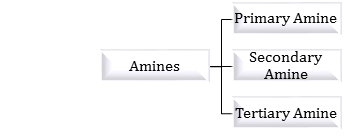
primary amine reaction with water
1°, 2°, and 3° amines can all form hydrogen bonds with water.
Low-molecular weight amines are generally water- soluble.
Higher molecular-weight amines often smell like rotting fish, and are often found in decaying animal tissues.
Are primary amines soluble in water?
Because all three classes of amines can engage in hydrogen bonding with water, amines of low molar mass are quite soluble in water.
What happens when amines react with water?
Water is a neutral molecule.
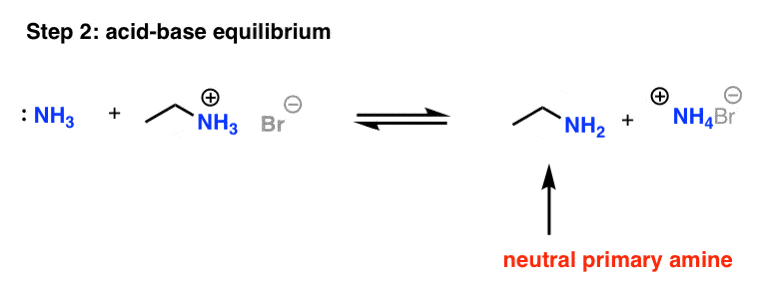
Amines are basic in nature because nitrogen atom contains a lone pair. so, when amines dissolved in water, it accepts the proton from water and forms ammonium ion.
What is the reaction of primary amine?
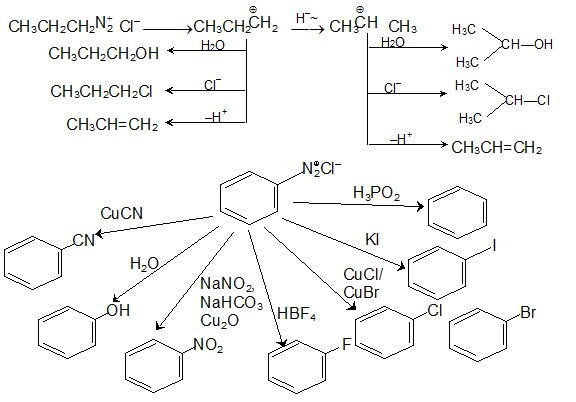
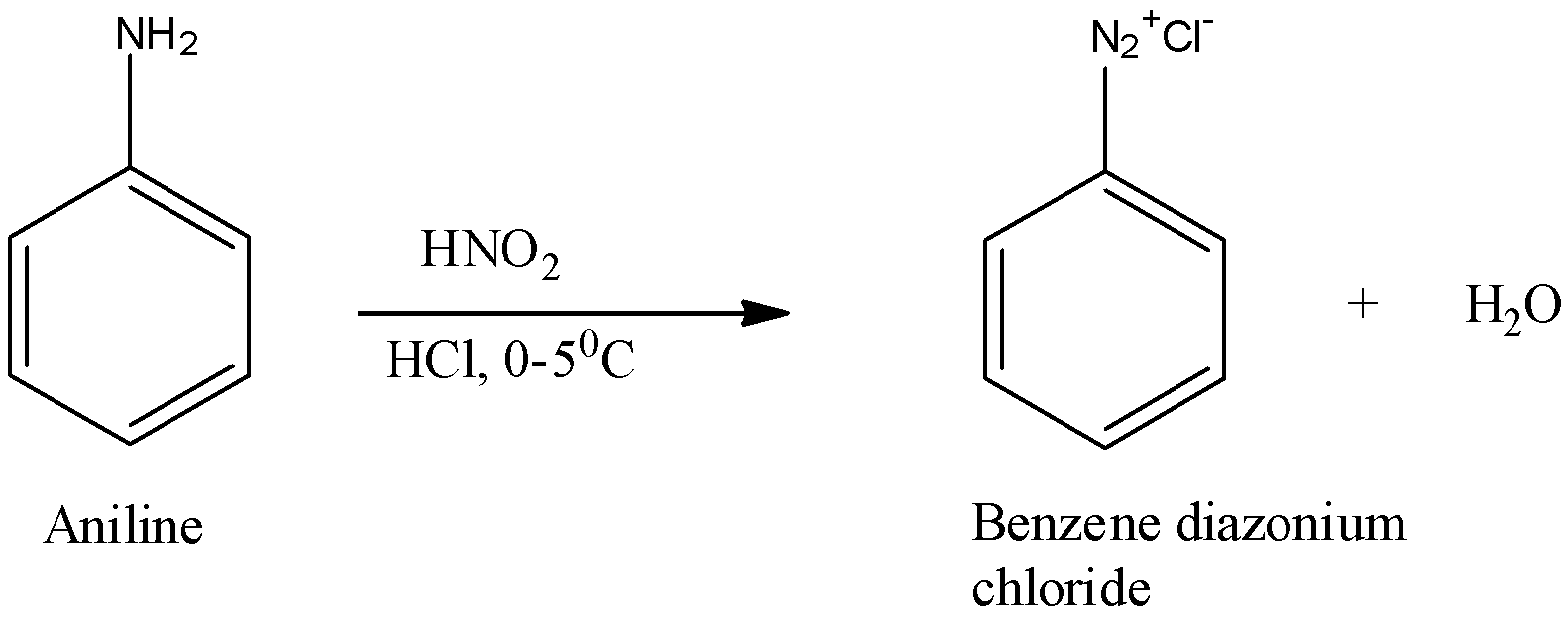
Primary amines react with nitrous acid to yield a diazonium salt, which is highly unstable and degradates into a carbocation that is capable of reaction with any nucleophile in solution.
Therefore, reacting primary amines with nitrous acid leads to a mixture of alcohol, alkenes, and alkyl halides.
|
Reactions of Amines
are more acidic than neutral water/alcohol! 5. Neutral amine can completely deprotonate carboxylic acids but not water or alcohols. 6. Therefore hydroxide can |
|
Chapter 6 Amines and Amides
Chemical Properties of Amines: Basicity. • Amines are weak organic bases. They react with water to produce alkylammonium ions and hydroxide anions:. |
|
Formylation of Amines
10-Jun-2014 The amine and formic acid were heated to 80 °C until the reaction ... Reaction of primary amines with triethyl orthoformate in water. |
|
Reactions of Amines
are more acidic than neutral water/alcohol! 5. Neutral amine can completely deprotonate carboxylic acids but not water or alcohols. 6. Therefore hydroxide can |
|
Testsforfunctionalgroups - inorganiccompounds
iodoform separates warm the reaction mixture in a water bath at Aliphatic as well as aromatic primary amines give carbylamine test in which an amine is ... |
|
Reaction of primary amines with Pt/C catalyst in water under
Reaction of primary amines with Pt/C catalyst in water under microwave irradiation: a convenient synthesis of secondary amines from primary amines. |
|
Analytical Procedures Employing Karl Fischer Reagent. XII. The
The determination of primary amines with few Occasionally the reaction between primary amine ... The water formed from the rapid quantitative reaction ... |
|
19.11 REACTIONS OF ALDEHYDES AND KETONES WITH AMINES
A primary amine is an organic derivative of ammonia in which only one the removal of one of the reaction products (usually water; see Eq. 19.60). |
|
Kinetics reaction of primary and secondary amine group in aqueous
Both DETA and water contribute to the carbamate formation in the primary and secondary amine groups. The rate of CO2 absorbed in the primary amine group is |
|
The Reaction of Primary Amines with 2-Nitramino-?2-13
droxide solution. The residue from the washed and dried ether solution was recrystallized from a mixture of ethanol and water; yield 138 mg. (64%); m. |
|
Reactions of Amines
Reverse Mechanism: Required (deprotonation) • Amines are completely converted to ammonium salts by acids • Ammonium salts are completely neutralized |
|
Kinetics reaction of primary and secondary amine group in aqueous
Both the termolecular and the zwitterion mechanisms were applied to interpret the experimental data and gave identical result Both DETA and water contribute to |
|
CHAPTER 7 AMINES
Amines undergo neutralization reactions with acids to form alkylammonium One example of a molecule containing the piperidine ring is piperine, the primary |
|
AMINES
Primary amine manifests itself by the characteristic odor of iso- nitrile Reaction with Ninhydrin Similar to amino acids, primary aliphatic amines give a very |
|
145 Chapter 24: Amines Amines: Nitrogen containing organic
Primary and secondary amines, likes water and alcohols, can be hydrogen Synthesis of primary amines from the reaction of alkyl halides or tosylates with |
|
NITROGEN CONTAINING COMPOUNDS Amines - Patna Science
(c) Tertiary amines react with nitrous acid to form trialkylammonium nitrite salts which are soluble in water Page 18 This reaction is used as the basis of a test to |
|
Amines
What Are the Reactions of Amines with Acids? 10 6 How Are in Water Ammonia NH3 78 33 very soluble Primary Amines methylamine CH3NH2 95 6 |
|
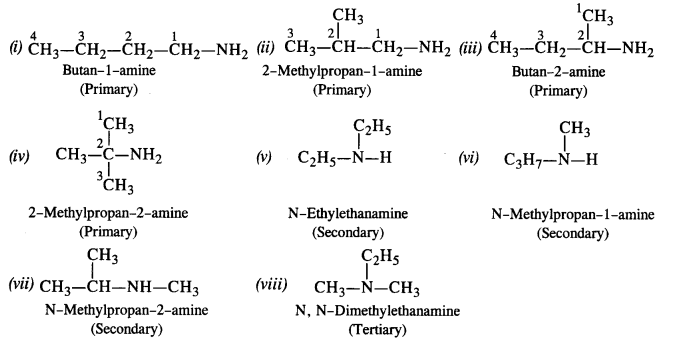
Amines Amines - NCERT
IUPAC system, primary amines are named as alkanamines The name is derived Amines, being basic in nature, react with acids to form salts 13 6 Chemical |
|
Amine Chemistry Tutorial
Tertiary amines have even more limited water solubility since they do not reaction is a base and the resulting protonated amine is its conjugate acid: Based on |


















![PDF] Synthesis of primary amines from secondary and tertiary PDF] Synthesis of primary amines from secondary and tertiary](https://www.vedantu.com/question-sets/e0c37b0a-cc3d-4421-862d-619e81a36eae8319358317465134999.png)