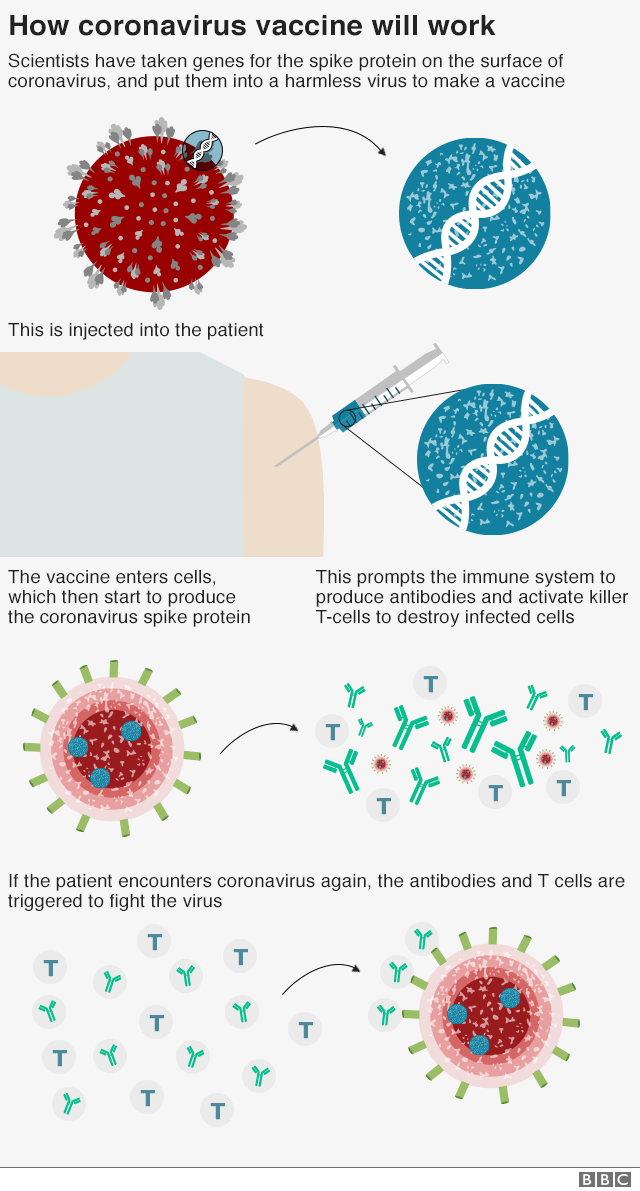
coronavirus vaccine update usa
Is the new COVID vaccine FDA approved?
The FDA has approved and authorized for emergency use updated COVID-19 vaccines (2023-2024 formula) that include a monovalent (single) component that corresponds to the omicron variant XBB.4 oct. 2023
If you plan to visit the U.S., you do not need to be tested or vaccinated for COVID-19.
U.S. citizens going abroad, check with the Department of State for travel advisories.
|
Summary Document for Interim Clinical Considerations for Use of
Sep 13 2022 COVID-19 vaccine products currently approved or authorized in the United States. Pfizer-BioNTech. Age indication. Vaccine composition. |
|
Janssen COVID-19 Vaccine EUA Fact Sheet for Healthcare Providers
May 5 2022 The Janssen COVID-19 Vaccine can cause thrombosis with ... The U.S. Food and Drug Administration (FDA) has issued an ... been updated. |
|
Updates to COVID-19 Vaccine Effectiveness in the United States
Sep 1 2022 cdc.gov/coronavirus. Updates on COVID-19 Vaccine. Effectiveness during Omicron. Ruth Link-Gelles |
|
ACIP COVID-19 Vaccines Work Group-October 30 2020
Oct 30 2020 Phase 1 allocation for COVID-19 vaccine: Work Group considerations. Background ... Vaccine Update: Phase III clinical trials in the U.S.. |
|
COVID-19 Vaccine Frequently Asked Questions September 2 2022
Sep 2 2022 United States today include those to protect against influenza and ... A single booster dose with an updated bivalent COVID-19 vaccine is ... |
|
National Strategy for the COVID-19 Response and Pandemic
Jan 21 2021 Launch a national vaccinations public education campaign. The United States will build public trust through an unprecedented vaccination public ... |
|
ACIP Update: Thrombosis with thrombocytopenia syndrome (TTs
May 12 2021 Update: Thrombosis with thrombocytopenia ... Update on TTS cases ... U.S. COVID-19 vaccine administration by product type and TTS reports to ... |
|
COVID-19 vaccine safety updates: Primary series in children and
Jan 5 2022 U.S. reports to VAERS among children and adolescents ages 5–11 and. 12–15 years after Pfizer-BioNTech COVID-19 vaccination* (as of Dec 19 |
|
COVID-19 Vaccine Healthcare Provider Frequently Asked Questions
Sep 2 2022 protein subunit vaccines used in the United States today include ... A single booster dose with an updated bivalent COVID-19 vaccine is ... |
|
COVID-19 Vaccine Clinical Guidance Summary for Patients with
Mar 4 2021 Updated guidance clarifying the recommendation for a 3-dose primary ... Novavax COVID-19 vaccine in the U.S. and the availability of the ... |
|
COVID-19 Vaccine Update - NCgov
il y a 6 jours · An Emergency Use Authorization (EUA) allows the FDA to get a safe COVID-19 vaccine to you quickly during a public health emergency An independent advisory committee makes sure the vaccines are safe and work before issuing an EUA December 10, 2020, and the EUA was approved on December 11, 2020 |
|
COVID-19 vaccine Safety update—January 2021 - CDC
27 jan 2021 · Disease Control and Prevention (CDC) or the U S Food and Drug Administration Update on anaphylaxis following COVID-19 vaccination |
|
COVID-19: vaccine summary
Data in this summary pertain to COVID-19 vaccines approved by the U S Food and Drug Administration and have been issued an Emergency Use Authorization |
|
Interim COVID-19 Vaccine Provider Guide - Minnesota Department
il y a 2 jours · (www health state mn us/diseases/coronavirus/vaccine/provider html) updated to reflect supplies needed for six doses per vial |
|
COVID-19 Vaccine Frequently Asked Questions March 18 - NJgov
il y a 3 jours · the United States under the Department of Health and Human Services What is New Jersey doing to plan for the COVID-19 vaccine? The Department will continue to update the plan as we receive new information and |
|
February 2021 - European Medicines Agency - europaeu
5 fév 2021 · All published safety updates for COVID-19 Vaccine Moderna are January 2021 at a site in San Diego, California, US with vaccine from the |
|
National Strategy for the COVID-19 Response - The White House
The United States will spare no effort to ensure Americans can get vaccinated quickly minimize the risk of COVID infection, and work with states to update their |