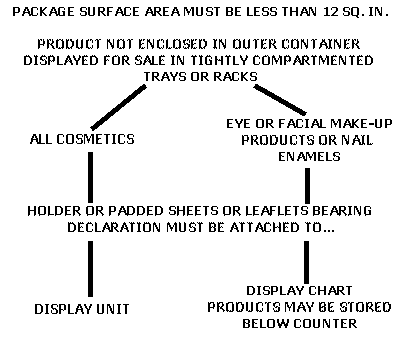
cosmetic labelling requirements
|
Annex I Part 7 COSMETIC LABELING REQUIREMENTS
This document provides guidance for the labelling requirements of cosmetic products They are required to be labelled in accordance with the Regulations |
|
Guidelines on cosmetic product labelling
The following table contains an overview of the compulsory labelling requirements of the Regulation Please see Chapter III for more detailed information on |
What are the EU labeling requirements for cosmetics?
Specifically, the EU Cosmetics Regulation requires cosmetic products to provide the following information on the label or on the packaging: The name and the address of the company (Responsible Person).
An ingredients list, in decreasing order of weight of the ingredients.What are the FDA requirements for product labeling?
All labels must be designed and applied to devices and containers so that the labels will remain in place and legible during the customary conditions of distribution, storage, and use.
Likewise, other labeling, such as user instructions, should remain legible during customary storage and use.Additional labelling requirements for packaged food products, as stipulated in Part VII of the Prevention of Food Adulteration Rules, 1955, and the Standards of Weights and Measures (Packaged Commodities) Rules of 1977, require that the labels contain the following information: Name, trade name, or description.
What is required on a cosmetic label?
The FDA has issued regulations which cover the labeling requirements for cosmetics, which are located in the Code of Federal Regulations Title 21, Sections 700 - 740.
In brief, the label for a cosmetic must contain: Identity of the product (what it is) Net Contents (how much is in the package)
|
Guidelines for Cosmetic Advertising and Labelling Claims
The basis for the regulatory authority for the. Cosmetics Division comes from the Food and Drugs Act and Cosmetic Regulations. Advertising Standards Canada (ASC). |
|
Canada.ca
The Cosmetic Regulations to the Food and. Drugs Act allow a designated Health Canada inspector to inspect: — cosmetic products. — locations where cosmetics are |
|
Food and Drug Administration Cosmetic Labeling Guide
The cosmetics marketed in the United States whether they are manufactured here or are imported from abroad |
|
Canada.ca
The information in this guide has been prepared to provide an overview of the INCI labelling requirements of the Cosmetics Regulations under the Food and Drugs |
|
LABELING REQUIREMENTS - Singapore
that the cosmetic products comply with the Regulations before they supply the product. Labels or labeling statements must be in English and legible. The |
|
APPENDIX II ASEAN COSMETIC LABELING REQUIREMENTS A
Sep 4 2007 This document provides guidance for the labeling requirements of cosmetic products to which Article 6 of the ASEAN Cosmetic. Directive 05/01/ ... |
|
Annex I Part 7 COSMETIC LABELING REQUIREMENTS
COSMETIC LABELING REQUIREMENTS. 1. OBJECTIVE. This document provides guidance for the labelling requirements of cosmetic products. They are required to be |
|
Guidelines on cosmetic product labelling
labelling requirements in other horizontal regulations that apply to cosmetic products. Page 4. Page 4 of 32. II. QUICK GUIDE FOR COSMETIC PRODUCT LABELLING IN. |
|
Summary Chart: Labelling Requirements for Cosmetics in
Summary Chart: Labelling Requirements for Cosmetics in Pressurized Containers. Definition/. Criteria. Aerosol. Flammability. Tested according to CCCR Schedule |
|
Guidelines for the Nonprescription and Cosmetic Industry Regarding
How do you determine what advertising and labelling claims are properly representing your product according to legislative or regulatory requirements? |
|
Guidelines on cosmetic product labelling
governing the composition labelling and packaging of finished cosmetic products labelling requirements in other |
|
Criteria for the demarcation of cosmetic products from therapeutic
Thus Food Legislation |
|
APPENDIX II ASEAN COSMETIC LABELING REQUIREMENTS A
04.09.2007 This document provides guidance for the labeling requirements of cosmetic products to which Article 6 of the ASEAN Cosmetic. Directive 05/01/ ... |
|
LABELING REQUIREMENTS - Singapore
that the cosmetic products comply with the Regulations before they supply the product. Labels or labeling statements must be in English and legible. |
|
Food and Drug Administration Cosmetic Labeling Guide
The raw materials used as ingredients of cosmetic products are by law also cosmetics. Page 4. Cosmetic Labeling Guide. Page 4. In section 701.20 of Title 21 of |
|
COSMETIC INGREDIENT LABELLING IN THE EUROPEAN UNION
These guidelines are intended to provide information on the requirements of the Cosmetics. Directive concerning ingredient labelling and on those aspects of the |
|
34. Do we need to reflect the Manufacturers name and address on
The ASEAN Cosmetic Labeling Requirement requires the name and address of the company or person responsible for placing the product in the local market on the |
|
EPA NZ
In New Zealand cosmetics (see page 2 for a general The cosmetic standard has an overall requirement ... ingredients |
|
ASEAN Cosmetic Labeling Requirements Guidelines
This document provides guidance for labelling requirements of cosmetic products to which Article. 5 of the ASEAN Cosmetic Directive 05/01/ACCSQPWG apply. |
|
Annex I, part 7 COSMETIC LABELING REQUIREMENTS - NPRA
7 fév 2017 · Guidelines for Control of Cosmetic Products in Malaysia-1st Revision This document provides guidance for the labelling requirements of |
|
Labelling Requirements for Cosmetics - Oils4life
Labelling Requirements for Cosmetics All cosmetics placed on the market in the UK must be labelled in English All cosmetic products must be labelled with the |
|
Labelling - European Commission
The Cosmetic Toiletry and Perfumery Association represents cosmetic manufacturers Mandatory labelling requirements must be dealt with by sector specific |
|
Cosmetic Product Labelling - EU - Herrco Cosmetics
Article 19 of the EU Cosmetics Regulation (EC) No 1223/2009 specifies the labelling requirements for finished cosmetic products placed on the market anywhere |
|
Common Labeling Requirements
This document provides guidance for labelling requirements of cosmetic products to which Article 5 of the ASEAN Cosmetic Directive 05/01/ACCSQPWG apply |
|
A The ASEAN Cosmetic Labeling Requirement require
Meanwhile, you would need to plan how to ensure that your product labels comply with the ASEAN Cosmetic Labeling requirements by January 2008 F Claims |