ap chemistry chapter 13 properties of solutions practice test
|
AP* Chemistry PROPERTIES OF SOLUTIONS
A chemist is trying to identify a human hormone which controls metabolism by determining its molar mass A sample weighing 0 546 g was dissolved in 15 0 g |
|
Chapter 13 Properties of Solutions
How Does a Solution Form? As a solution forms the solvent pulls solute particles apart and surrounds or solvates them |
|
AP* Chemistry PROPERTIES OF SOLUTIONS
Properties of Solutions 13 Exercise 8 Calculating the Molar Mass by Boiling-Point Elevation A solution was prepared by dissolving 18 00 g glucose in 150 0 g |
|
AP Chemistry Practice Test
A P Chemistry Practice Test - Ch 13: Equilibrium At equilibrium at 25eC a 0 100 M solution of acetic acid has the following concentrations: |
|
AP Chemistry Practice Test: Ch 11 Solutions
A P Chemistry Practice Test: Ch 11 Solutions Choose the one alternative that best completes the statement or answers the question 1) Formation of |
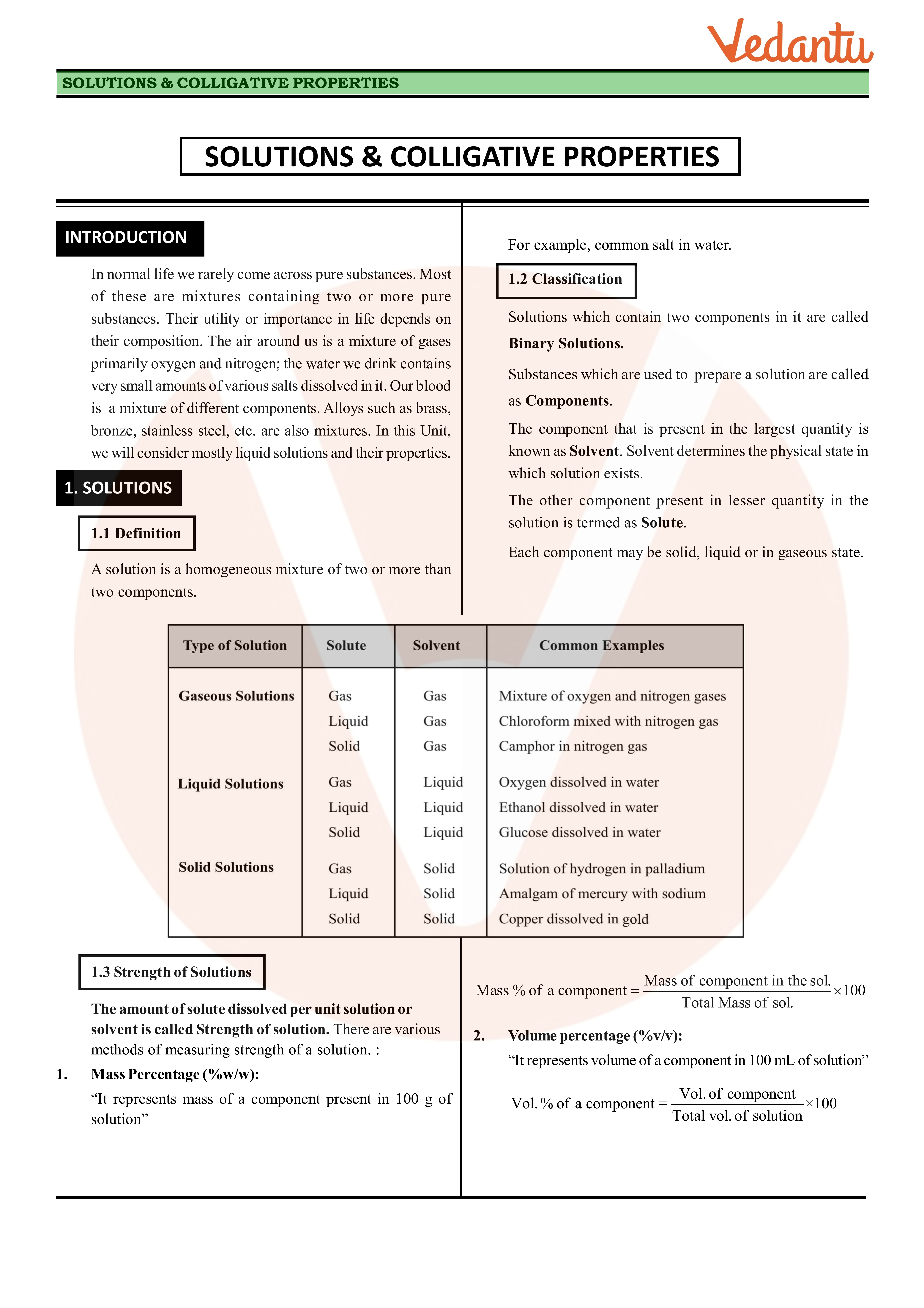
How will you describe the physical properties of solutions?
These properties are called as colligative properties or collective properties.
The colligative properties are vapor-pressure lowering, boiling-point elevation, freezing-point depression, and osmotic pressure.The property of a solution depends only on the concentration of ion/molecule/atom of the solute.
The colligative properties are osmotic pressure, depression in freezing point, boiling point elevation, and relative lowering of vapor pressure.
What are the properties of a solute?
A solute is the substance that dissolves completely in another substance known as a solvent.
When we add a solute to the solvent, it alters general properties of the solvent such as its freezing or boiling point.
These changes in property are respectively called freezing point depression and boiling point elevation.
What are the properties of solution in chemistry?
Properties of Solution
It is a homogeneous mixture.
Its particles are too tiny and have a diameter of less than 1 nm.
The particles are not visible to naked eyes.
Particles don't scatter a beam of light passing through it and hence the path of the light is not visible.
|
Chapter 13 Properties of Solutions.pdf
Chapter 13 – Properties of Solutions Solution formation & Chemical Reactions ... A chemical reaction is more advanced than a simple formation of a ... |
|
A.P. Chemistry Practice Test: Ch. 11 Solutions
A.P. Chemistry Practice Test: Ch. 11 Solutions 13) The concentration of nitrate ion in a solution that contains 0.900 M aluminum nitrate is ______ M. |
|
Chapter 13: Physical Properties of Solutions
Solution: a homogeneous mixture. • Solute: the component that is dissolved in solvent. -- usually in smaller amount. • Solvent: medium (often water) into |
|
Chapter 13 Properties of Solutions
Chapter 13. Properties of Solutions. Chemistry The Central Science |
|
A.P. Chemistry Practice Test - Ch. 13: Equilibrium
A.P. Chemistry Practice Test - Ch. 13: Equilibrium 8) Acetic acid is a weak acid that dissociates into the acetate ion and a proton in aqueous solution:. |
|
AP Chemistry Course and Exam Description - Effective Fall 2022
13 Science Practices. 17 Course Content. 22 Course at a Glance. 27 Unit Guides. 28 Using the Unit Guides. 31 UNIT 1: Atomic Structure and Properties. |
|
1 Chapter 13: Nuclear Magnetic Resonance (NMR) Spectroscopy
Not all nuclei are suitable for NMR. 1H and 13C are the most important NMR active nuclei in organic chemistry. Natural Abundance. 1H |
|
Untitled
Chapter 13 Properties of Solutions Chapter 19 Chemical Thermodynamics. ... This manual Solutions to Exercises in Chemistry: The Central Science |
|
Recommended Chemical Soil Test Procedures
Over the past 30 years the NCR-13 Soil Testing Chapters. 1. Soil Sample Preparation . ... showed that soil properties |
|
Chapter 13
Section 13H Solutions to Practice Exercises In this chapter we exam- ... The earliest analytical methods based on chemical kinetics—which first ap-. |
|
AP* Chemistry PROPERTIES OF SOLUTIONS
Calculate the molarity, mass percent, mole fraction, and molality of ethanol in this solution Exercise 2 Calculating Various Methods of Solution Composition from |
|
Chapter 13 Properties of Solutions
Solutions Chapter 13 Properties of Solutions Chemistry, The Central Science, 10th Solutions The intermolecular forces between solute and solvent particles |
|
Chapter 13 – Properties of Solutions
Chapter 13 – Properties of Solutions Solution formation Chemical Reactions A chemical reaction is more advanced than a simple formation of a solution as Ex: If the boiling point of a sample is 2 3 Celcius above the boiling point of |
|
Untitled - WL Moodle
Chemistry I Chapter 13 Test - Solutions Name 2) A sample of potassium nitrate (49 0 g) is dissolved in 101 g of water at 100 °C, with precautions taken to avoid evaporation of any water B) osmotic properties predict this behavior C) of the |
|
AP Chemistry Practice Test: Ch 11, Solutions
A P Chemistry Practice Test: Ch 11, Solutions 13) The concentration of nitrate ion in a solution that contains 0 900 M aluminum nitrate is ______ M A) 0 450 |
|
CHM 2046 Test 1 Review: Chapter 11, Chapter 12, & Chapter 13
CHM 2046 Test 1 Review: Chapter 11, Chapter 12, Chapter 13 Chapter 11: at 40℃ have properties of both liquid and gas? a 5 1 atm b 39 atm Which of the following would you expect to form a homogenous solution when mixed with BF3 ? source: http://www sciencegeek net/Chemistry/taters/solubility htm 13 |
|
Chpt 13 Solutions - Chemistry 11
Announcements Chapter 13: Start problems in Chapter 13 Chem 11 Exam 1 The Properties of Mixtures: Solutions and Colloids using IMF's--helps one understand the practical) 2 Calculate the vapor pressure lowering, AP, when |
|
Chemistry Chapter Colligative Properties Quiz Key
chapter 13 review ions in aqueous solutions and colligative properties mixed the left, study flashcards on chemistry test 1 chapter 11 at cram com quickly quizlet, colligative properties practice test b student copy of practice test b chapter 13 click chp 13 2013 pdf link to view the file ap chemistry ap chemistry home page |