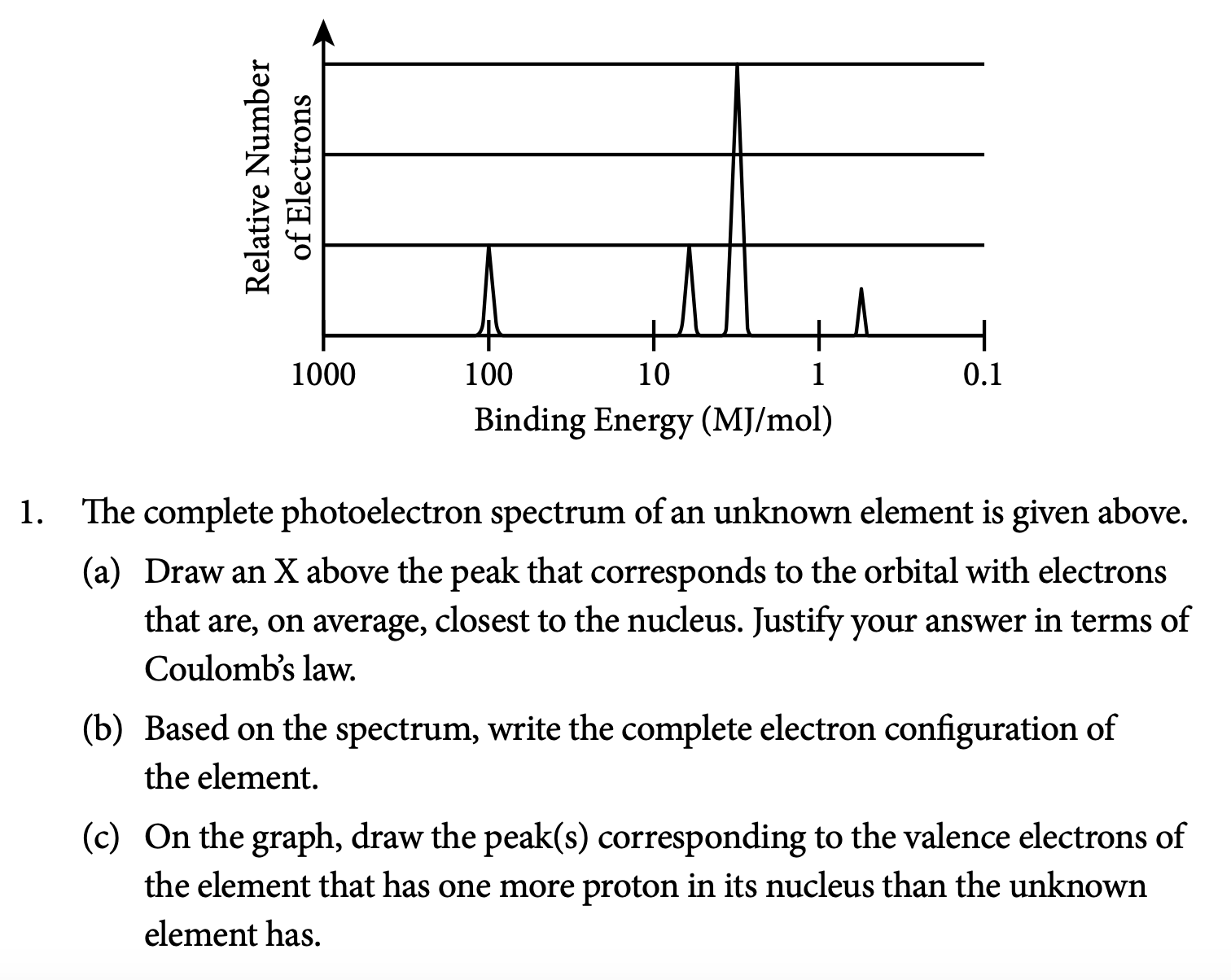
ap chemistry gas laws free response questions
|
AP® CHEMISTRY 2011 SCORING GUIDELINES (Form B)
Question 2 (continued) (b) Consider the three gases in correctly using all the moles of gas in the tank in the Ideal Gas Law to calculate the final pressure |
|
2012 ap® chemistry free-response questions
A sample of a pure gaseous hydrocarbon is introduced into a previously evacuated rigid 1 00 L vessel The pressure of the gas is 0 200 atm at a temperature of |
|
AP* Chemistry GASES
We love to ask question comparing the behavior of ideal and real gases It's not likely you'll be asked an entire free-response gas problem on the real exam in |
|
AP* Gas Law Free Response Questions KEY
AP* Gas Law Free Response Questions KEY (1) AP® is a registered trademark of AP® CHEMISTRY 2002 SCORING GUIDELINES (Form B) Copyright © 2002 by College |
|
AP* Gas Law Free Response Questions
(a) A solution is prepared from 1 0 mole of biphenyl a nonvolatile solute and 49 0 moles of benzene Calculate the vapor pressure of the solution at 20°C (b) |
|
AP* Kinetics Free Response Questions KEY
The rate law shows that the slow reaction must involve one Y consistent with mechanism 3 Mechanisms 1 and 2 would involve both [X] and [Y] in the rate law |
|
AP* Kinetics Free Response Questions
Briefly describe an experiment that can be conducted in order to determine the values of m and n in the rate law for the reaction (d) From the information |
|
2013 AP® CHEMISTRY FREE-RESPONSE QUESTIONS
Answer the following questions involving the stoichiometry and thermodynamics of reactions containing aluminum species 2 Al2O3(l) + 3 C(s) 4 Al(l) + 3 CO2(g) |
|
AP Chem
2004 AP® CHEMISTRY FREE-RESPONSE QUESTIONS (Form B) Answer EITHER Question 2 below OR Question 3 printed on page 8 Only one of these two questions will be |
|
AP Chemistry 2021 Free-Response Questions
Questions 4–7 are short free-response questions that require about 9 minutes each to answer and are worth 4 points each For each question show your work for |
|
AP Chemistry Free-Response Question 7
(d) For a valid explanation: The ideal gas law assumes that gas particles do not experience interparticle attractions As a real gas cools further the |
|
Name: ______ AP Chemistry Gases Practice Problems
Explain your answer c) Compare the average kinetic energies of the gas molecules in the balloons Explain d) Twelve hours after being filled all |
|
2014 apr chemistry free-response questions
2014 APR CHEMISTRY FREE-RESPONSE QUESTIONS CaCO3(s) CaO(s) + CO2 When the reaction stopped the volume of collected gas was 0 0854 L at 0 822 atm and 305 K |
|
Unit 8 Free Response Questions
Explain your answer in terms of kinetic molecular theory 5 Why must all gas law problems involving temperature be solved using the Kelvin (absolute) |
What is the most important rule in chemistry?
The most fundamental concept in chemistry is the law of conservation of mass, which states that there is no detectable change in the quantity of matter during an ordinary chemical reaction.
What is the 15 Boyle's law?
For a fixed mass of an ideal gas kept at a fixed temperature, pressure and volume are inversely proportional.
Boyle's law is a gas law, stating that the pressure and volume of a gas have an inverse relationship.
If volume increases, then pressure decreases and vice versa, when the temperature is held constant.What is Boyle's law? Answer: Boyle's law depicts the relationship between the pressure, volume, and temperature of a gas.
It states that the pressure of a gas is inversely proportional to its volume at a constant temperature.
What are the basic laws of chemistry?
In Chemistry there are Three Fundamental Laws of Chemistry that you need to know: Law of Conservation of Mass, Law of Definite Proportions, Law of Multiple Proportions.
|
AP* Gas Law Free Response Questions KEY
AP* Gas Law Free Response Questions KEY. (1) AP® is a registered trademark of the College Board. The College Board was not involved in the production of and |
|
2021 AP Exam Administration Student Samples: AP Chemistry Free
Free Response Question 7 AP® Chemistry 2021 Scoring Guidelines ... The ideal gas law assumes that gas particles do not experience interparticle ... |
|
AP* Gas Law Free Response Questions
Equilibrium Vapor Pressure of H2O (25°C). 23.8 mm Hg. Page 4. AP* Gas Law Free Response Questions page 4. (a) Calculate the number of moles of hydrogen gas |
|
AP Chemistry 2019 Free-Response Questions
Ideal gas at STP = 22.4 L molí1 2019 AP® CHEMISTRY FREE-RESPONSE QUESTIONS. GO ON TO THE NEXT PAGE ... than the pressure predicted by the ideal gas law. |
|
Untitled
Welcome to your summer packet for AP Chemistry. The FRQs for stoichiometry must be written on a blank sheet of paper with your name. |
|
AP Chemistry 2015 Free-Response Questions
When the reaction stopped the volume of collected gas was 0.0854 L at 0.822 atm and 305 K. (The vapor pressure of water at 305 K is 35.7 torr.) (a) Calculate |
|
AP® CHEMISTRY 2011 SCORING GUIDELINES (Form B)
Visit the College Board on the Web: www.collegeboard.org. Question 2 (continued). (b) Consider the three gases in the tank at 327°C: CH3OH(g) CO(g) |
|
AP Chemistry 2017 Free-Response Questions
Ideal gas at STP = 22.4 L molí1 2017 AP® CHEMISTRY FREE-RESPONSE QUESTIONS ... (a) Chlorine gas Cl2(g) |
|
AP Chemistry 2016 Free-Response Questions
Assume that initially all the gas present in the vessel is C4H6. (b) Use the data plotted in the graphs to determine the order of the reaction with respect to |
|
AP Chemistry 2018 Free-Response Questions
Ideal gas at STP = 22.4 L molí1 Questions 4–7 are short free-response questions that require about 9 ... 2018 AP® CHEMISTRY FREE-RESPONSE QUESTIONS. |
|
AP* Gas Law Free Response Questions KEY
b) 3 points SO2 is the least ideal gas It has the largest size or volume It has the stongest attractive forces ( van der Waals forces or dipole-dipole interactions) |
|
AP Chemistry 2019 Free-Response Questions - AP College Board
Ideal gas at STP = 22 4 L molí1 2019 AP® CHEMISTRY FREE-RESPONSE QUESTIONS GO ON TO THE than the pressure predicted by the ideal gas law |
|
Unit 2 Gas Laws (Chapter 5) Test Set 1 Chapter 5: AP Multiple
Chapter 5: AP Multiple Choice Review Questions (1-16) (between pages 244 and 245 in AP free response (on back of this page): 2012 #2 a-e, 2013 #5 a-b law (Unit 3, Topic 3 4) SAP-7 A 1 The macroscopic properties of ideal gases are |
|
Free response questions - White Station High School AP Chemistry
Equilibrium Vapor Pressure of H2O (25°C) 23 8 mm Hg Page 4 AP* Gas Law Free Response Questions page 4 (a) Calculate the number of moles of hydrogen |
|
AP* Chemistry GASES - Houston ISD
Summer Overview Welcome to your summer packet for AP Chemistry Questions (MCQs) and Section 2: Free Response Questions (FRQs), unless otherwise specified Free-Response Boyle's Gas Law: P1V1 = P2V2 • Scientific Laws – a |
|
AP* Chemistry GASES
AP* Chemistry GASES The gaseous state of matter is the simplest and But you'll thank me later when you're flying through such gas law problems It's not likely you'll be asked an entire free-response gas problem on the real exam in |
|
2013 AP® CHEMISTRY FREE-RESPONSE QUESTIONS
2013 AP® CHEMISTRY FREE-RESPONSE QUESTIONS using the calculated moles of Al from part (a) and 1 point for correctly substituting in the ideal gas law |
|
AP* Chemistry GASES
*AP is a registered trademark of the College Board, which was not involved in the production of, and does not endorse, this product BOYLE'S LAW: “father of chemistry”--the volume of a confined gas is Work stoichiometry problems using your It's not likely you'll be asked an entire free-response gas problem on |
|
AP Chemistry 2018 Free-Response Questions
30 mar 2020 · Kp (gas pressures) 2018 AP® CHEMISTRY FREE-RESPONSE QUESTIONS D 1 than the pressure predicted by the ideal gas law |
|
AP Chemistry task
2012 Free-Response Questions 2012 AP® CHEMISTRY FREE-RESPONSE QUESTIONS pressure of the gas is 0 200 atm at a temperature of 127°C |