ap chemistry properties of solutions
|
Chapter 12 Physical Properties of Solutions
It turns out that all chemical processes are governed by two factors The first of these is the energy factor In other words does the solution process absorb |
|
11Properties of Solutions
Solutions can be gases liquids or solids as shown in Table 11 1 However we will be concerned in this chapter with the properties of liquid solutions |
|
Solutions
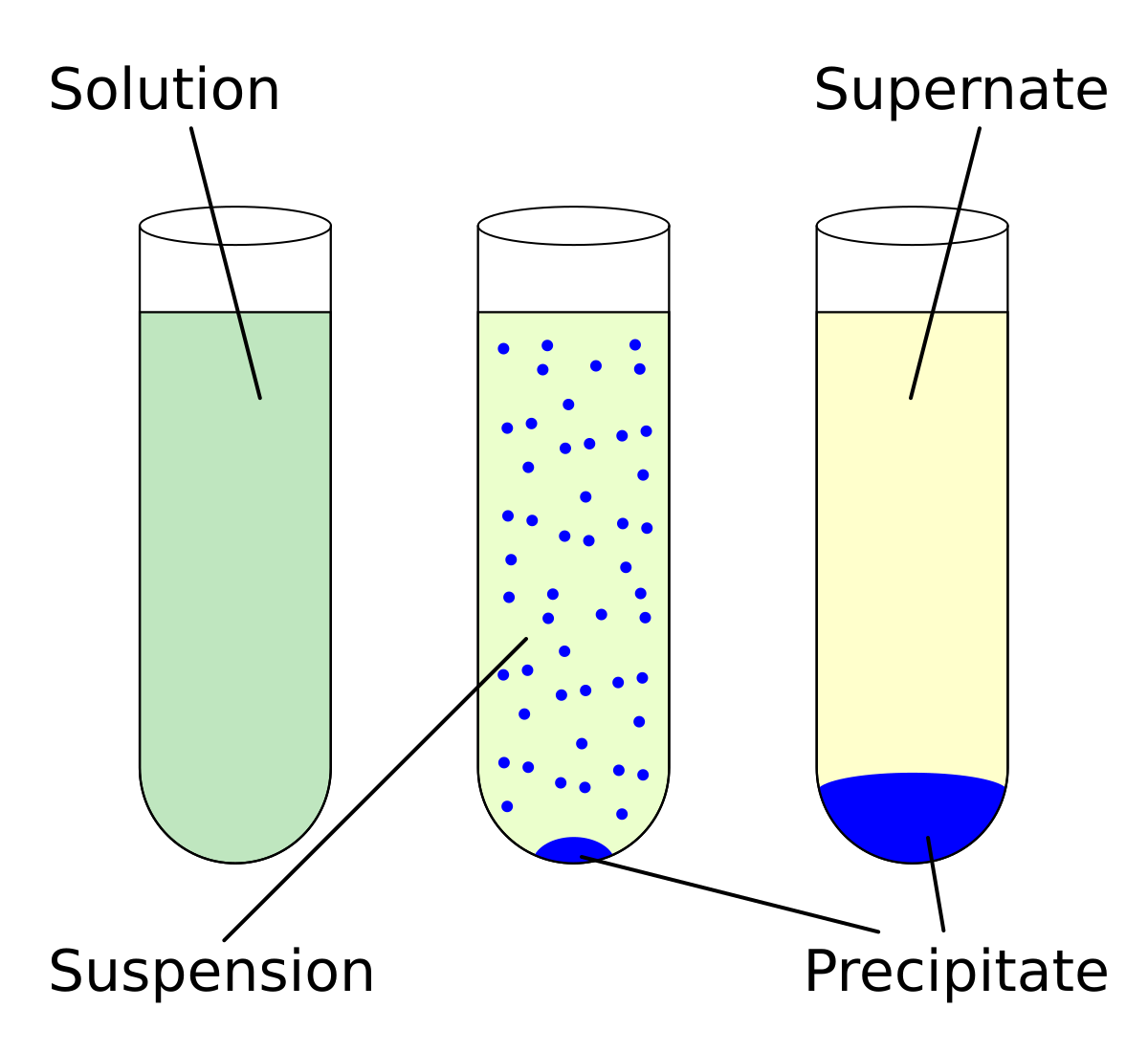
A Molarity is the measure of moles of solute per liter of solution 5 E All of the choices are colligative properties which means they depend only on the |
What are the 5 properties of solution?
Properties of Solution
It is a homogeneous mixture.Its particles are too tiny and have a diameter of less than 1 nm.The particles are not visible to naked eyes.Particles don't scatter a beam of light passing through it and hence the path of the light is not visible.A solute is any substance that is dissolved in a liquid solvent.
A solvent is the liquid that the solute is dissolved in and, together, the solute and solvent make up a solution.
An example of a solution is ocean water.
Ocean water is made up of many dissolved solutes with one of them being salt.
What are the properties of a solution summary?
Properties of a Solution
A solution is a homogeneous mixture.
The constituent particles of a solution are smaller than 10-9 metres in diameter.
Constituent particles of a solution cannot be seen by naked eyes.
Solutions do not scatter a beam of light passing through it.
What are the properties of solutions AP Chem?
These properties include: vapor pressure lowering, boiling-point elevation, freezing-point depression, and osmotic pressure.
|
AP* Chemistry PROPERTIES OF SOLUTIONS
AP* Chemistry. PROPERTIES OF SOLUTIONS. *AP is a registered trademark of the College Board which was not involved in the production of |
|
Properties of Solutions Lecture Outline 13.1 The - AP Chemistry
AP Chemistry: Properties of Solutions. Lecture Outline. 13.1 The Solution Process. A solution is a homogeneous mixture of solute and solvent. Solutions may |
|
Chapter 13: Physical Properties of Solutions
Solution: a homogeneous mixture. • Solute: the component that is dissolved in solvent. -- usually in smaller amount. • Solvent: medium (often water) into |
|
AP* Chemistry PROPERTIES OF SOLUTIONS
AP* Chemistry. PROPERTIES OF SOLUTIONS. *AP is a registered trademark of the College Board which was not involved in the production of |
|
A.P. Chemistry Practice Test: Ch. 11 Solutions
A.P. Chemistry Practice Test: Ch. 11 Solutions 4) A solution with a concentration higher than the solubility is ______. A) is not possible. |
|
AP Chemistry Course and Exam Description - Effective Fall 2022
7 About the AP Chemistry Course 45 UNIT 2: Molecular and Ionic Compound Structure and Properties ... 3.7 Solutions and Mixtures. 3.8 Representations of. |
|
1994 AP Chemistry Exam
Note: For all questions involving solutions and/or chemical equations assume that the system is in pure water and at to enhance its properties as a. |
|
AP Chemistry Scoring Guidelines 2016
A student investigates the enthalpy of solution ?Hsoln |
|
AP Chemistry
K. Solution chemistry. L. Factors affecting solubility. M. Colligative properties. Unit 4: Kinetics Equilibrium |
|
2021 AP Exam Administration Student Samples: AP Chemistry Free
AP®. Chemistry. Sample Student Responses and Scoring Commentary The greater electrical conductivity of the CaSO4 solution relative to the PbSO4. |
|
AP* Chemistry PROPERTIES OF SOLUTIONS
AP* Chemistry PROPERTIES OF SOLUTIONS *AP is a registered trademark of the College Board, which was not involved in the production of, and does not |
|
Chapter 11 – Properties of Solutions
componentA of fraction Mole + = = Chapter 11 – Properties of Solutions 11 1 Solution Composition A Molarity 1 solution of liters solute moles M Molarity = |
|
AP Chemistry: Properties of Solutions Lecture Outline 131 The
A solution is a homogeneous mixture of solute and solvent Solutions may be gases, liquids, or solids Each substance present is a component of the solution The solvent is the component present in the largest amount |
|
Chapter 13 Properties of Solutions
Some solutions form by physical processes and some by chemical processes • Consider: Ni(s) + Colligative properties depend on number of solute particles |
|
AP Chemistry Practice Test: Ch 11, Solutions
A P Chemistry Practice Test: Ch 11, Solutions Name___________________________________ MULTIPLE CHOICE Choose the one alternative that best |
|
Chapter 4 Aqueous Reactions and Solution Stoichiometry
AP Chemistry Chapter 4 Molarity is moles of solute per liter of solution not per liter of solvent • Students 4 1 General Properties of Aqueous Solutions |
|
AP Chemistry Unit 3 Solutions and Mixtures Notes • A - Effingham
: water is the dissolving medium, or ______ • One of most important properties of water is its ______ to many different substances • Water is Polar • |
|
AP* Solution Chemistry Free Response Questions - Waukee
AP* Solution Chemistry Free Response Questions the following phenomena in terms of the chemical and physical properties of the substances involved and |
|
AP CHEMISTRY NOTES 15-1 INTERMOLECULAR - Azle ISD
6 AP CHEMISTRY NOTES 15-3 COLLIGATIVE PROPERTIES SOLUTIONS Molality (m) – the number of moles of solute per kilogram solvent EXAMPLE: How |
















![Lecture 15 Solutions and Colligative Properties - [PDF Document] Lecture 15 Solutions and Colligative Properties - [PDF Document]](https://www.examexxpert.com/ncert-solution/11/chemistry/chapter-3/3.jpeg)


