types of buffer solution in chemistry
|
Buffers Types of Buffer Solutions Solutions of Single Substance
There are two types of buffers acid buffer and basic buffer • Acid buffer: A buffer solution containing large amounts of a weak acid and its salt with a |
What are the types of buffer solution?
There are several buffer systems in the body.
The most important include: (1) bicarbonate buffer (HCO3–/CO2), (2) haemoglobin buffer (in erythrocytes), (3) phosphate buffer, (4) proteins, and (5) ammonium buffer.What are the 3 types of buffer systems?
Buffers help maintain a stable inconsistent pH level in solutions.
They resist changes in PH when assets or bases are added.
Common buffers include phosphate buffers, Tris buffers (trishydroxymethylaminom ethane buffers that are very commonly used in molecular biology and biochemical applications.) and borate buffers.
|
BUFFER SOLUTIONS
chemistry and in real life and its types . 2- Realize the way by which the buffer stabilizes the pH . 3- Calculate the pH of all sorts of buffer solutions . |
|
Section 19.1. Acid-Base Buffer Solutions
Buffer Calculations: Two types we must be able to handle: (A) Calculate the pH (or pOH [H3O+] |
|
To prepare standard buffer solutions using acetic acid and sodium
Buffer solution is a mixture of weak acid or base wil it's strong salt. Hence buffer solutions are two types: Chemistry Lab Manual.... 19 ... |
|
1 General Chemistry II Jasperse Buffers/Titrations/Solubility. Extra
General Types/Groups of problems: Buffers General p1 Titration Graphs and Recognition p10. What Kind of Solution/pH at End? p2 Titration Calculations. |
|
Scientific Approach and Inquiry Learning Model in the Topic of
Buffer solution is a solution that resists changes in pH when small amounts of acid or base are added. There are two kinds of buffer that are acidic buffers |
|
Acces PDF Preparation Of A Buffer Solution Lab
4 days ago Buffer Solutions: Definition Types |
|
Acces PDF Buffer Solution Lab Report Theecoore
Aug 9 2020 2.4: Buffer Preparation - Chemistry LibreTexts. Buffer Solutions: Definition |
|
File Type PDF Buffer Solution Definition Chemistry (PDF) - covid19
It will unquestionably ease you to look guide Buffer Solution Definition Chemistry as master chemical peeling for darker skin types and examine case. |
|
Analysis of Student Chemistry Learning Difficulties on Buffer
grouping students' learning difficulties into five types includes not difficult difficulty in learning chemistry to understand of buffer solution topic. |
|
Electrochemical Cells and Buffer
When an electrode is dipped in a solution having its reversible ions or water three types of interaction is observed. 1) Cations from the metal surface |
|
BUFFER SOLUTIONS
chemistry and in real life and its types 2- Realize the way by which the buffer stabilizes the pH 3- Calculate the pH of all sorts of buffer solutions 4- Know how |
|
Chapter 17 lecture notes - Cal State LA
A buffer helps a solution maintain its pH when pH of buffered solution adding 0 10 M HCl to 100 mL soln An indicator is a chemical species that changes |
|
Buffer Solutions - UNINOVE
For example, a mixture of acetic acid and sodium acetate acts as a buffer solution with a pH of about 4 75 2 Alkaline (Basic) buffer solution: These are solutions that have a pH above 7 and contain a weak base and one of its salts |
|
The SOLUTION for All of Your Buffer Needs - The Wolfson Centre for
BUFFERS Quality, Consistency, Reliability The SOLUTION for All of Your Buffer Needs tain chemical constituents which act as weak acids or bases in an aqueous solu These solutions may vary with the membrane type and manufacturer |
|
Bufferpdf - Yengage
(b) Basic Buffer: It is the solution of a mixture of a weak base and salt of this In Analytical Chemistry: 0) For example, in the removal of acid radicals such as |
|
Handout on Buffer Solutions - Carnegie Mellon University
6 mar 2001 · Chemistry 09-106: Modern Chemistry II To understand buffer solutions, you need to understand a number of concepts and then tie these together The interesting species in the solution are then HA, A-, H3O+, and OH- |

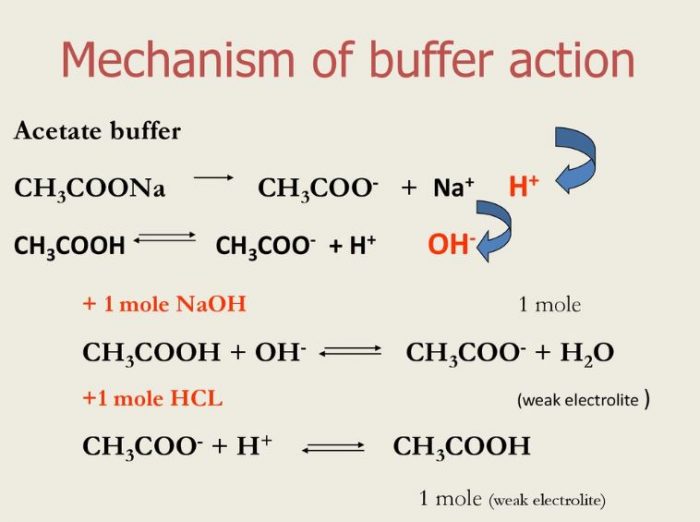

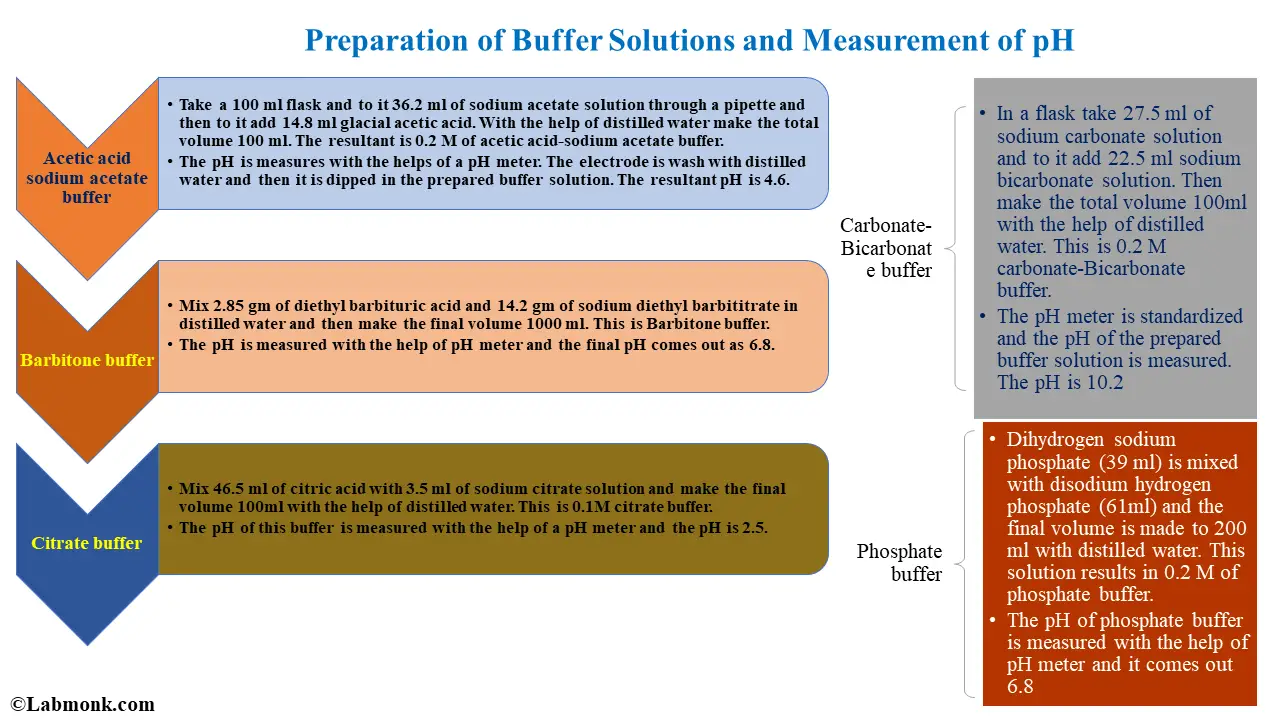








![Revision] Chemistry Part-3-- Hydrogen Economy Hard Water Types Revision] Chemistry Part-3-- Hydrogen Economy Hard Water Types](https://image.slidesharecdn.com/buffers-160329123505/95/buffers-30-638.jpg?cb\u003d1459254923)



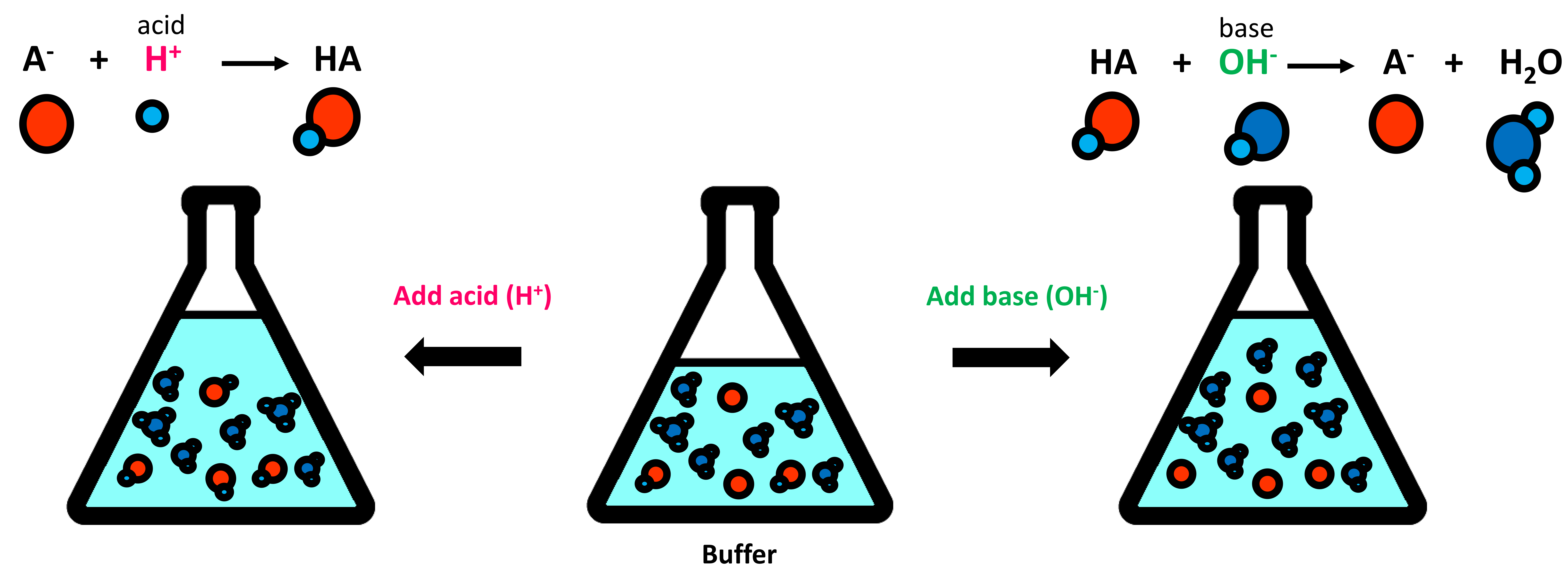






![PDF] BARC Exam Chemistry Syllabus PDF] BARC Exam Chemistry Syllabus](https://ars.els-cdn.com/content/image/3-s2.0-B9780128025055000032-f03-12-9780128025055.gif)







