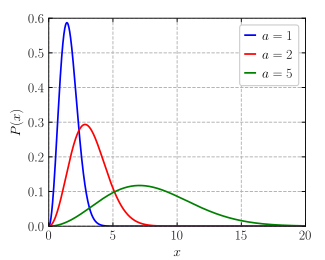
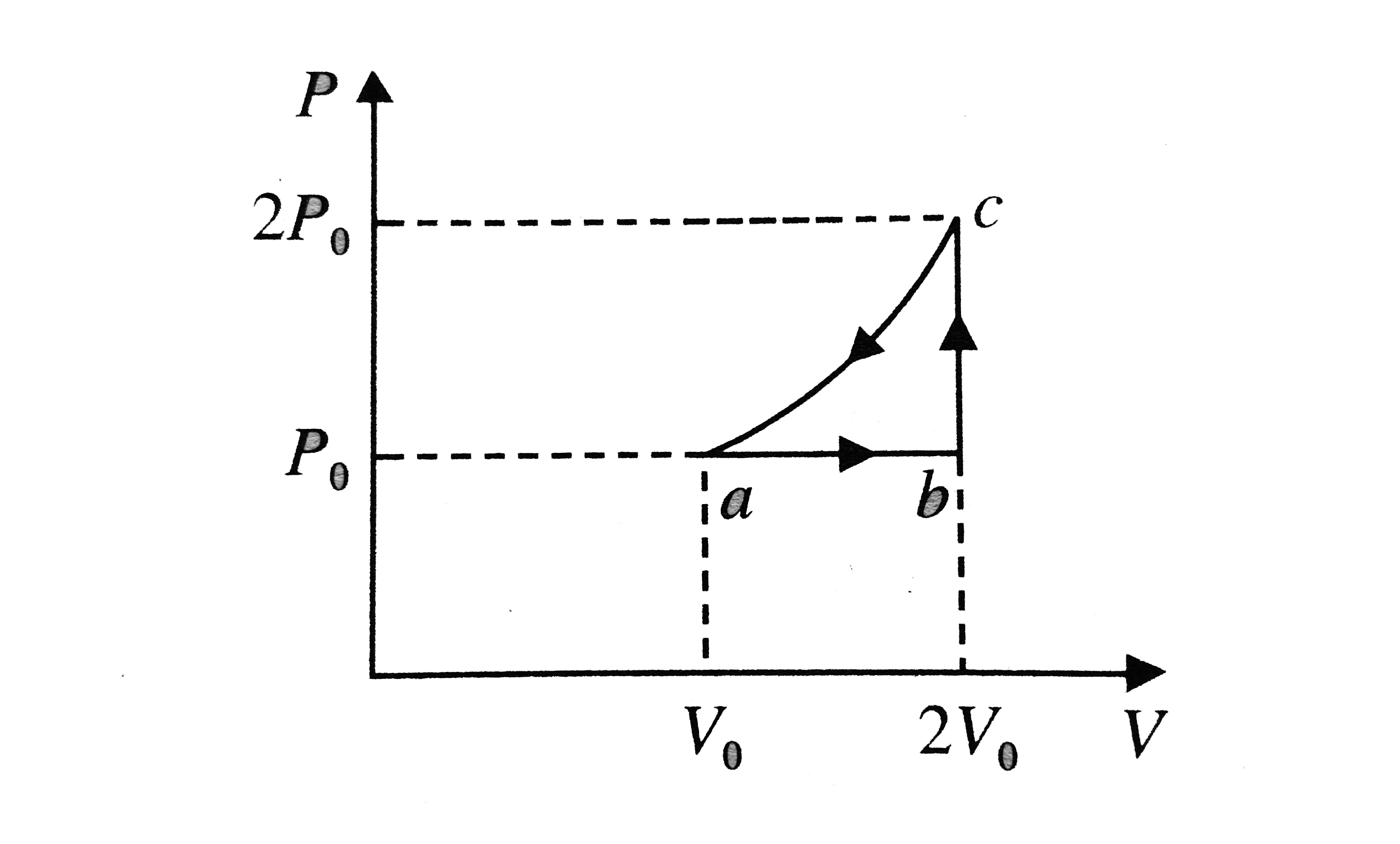
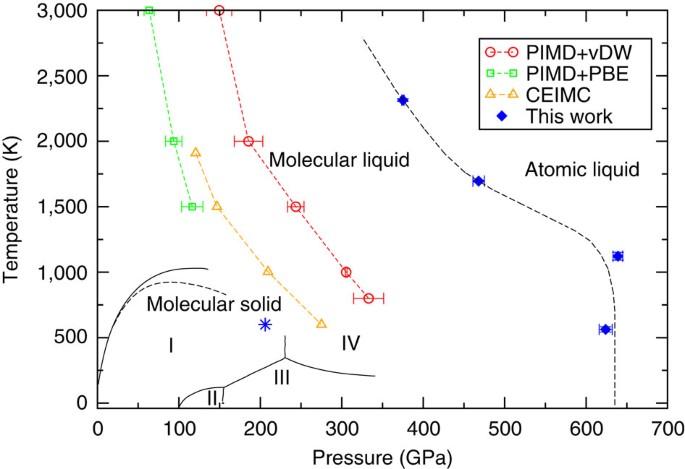
what fraction of its molecules are monatomic
Monatomic compounds are made of a single atom, whereas diatomic compounds are made of molecules with two atoms.
This is the primary distinction between monatomic and diatomic compounds.2 nov. 2020
What are the 7 monatomic elements?
The noble gases exist as monatomic elements:
helium (He)neon (Ne)argon (Ar)krypton (Kr)xenon (Xe)radon (Rn)oganesson (Og)What are the monatomic molecules?
Monoatomic (monatomic): A molecule composed of just one atom, and lacking any covalent bonds.
The noble gases (He, Ne, Ar, Kr, Xe, and Rn) are all monoatomic, whereas most other gases are at least diatomic.
Approximate Composition of the Atmosphere at the Earth's Surface.
How do you identify a monatomic molecule?
The determining factor for whether an element is monoatomic or diatomic is its electron configuration.
Monoatomic elements exist as individual atoms and have stable electron configurations, such as the noble gases.
Diatomic elements, on the other hand, exist as molecules composed of two atoms bonded together.
|
Atomic structural characteristics and dynamical properties in
monatomic metallic liquids via molecular dynamics simulations It is also found that fraction of the selected clusters in liquid Al is. |
|
THE DISSOCIATION OF MOLECULAR HYDROGEN FROM THE
ENTROPIES OF DIATOMIC AND MONATOMIC HYDROGEN. By Graham Edgar. Received December 6 1922. The extent of dissociation- of the hydrogen molecule into atoms at |
|
A Kinetic Model for Gas Mixtures Based on a Fokker-Planck Equation
in the correct Prandtl number for monatomic molecules. helium mixture in the hydrodynamic limit considering different mixture fractions. The paper is. |
|
Homework Week 4
For a polyatomic molecule the vibrational energy is the sum of the temperature |
|
VI. On the law of distribution of molecular velocities and on the
THEORY OF VISCOSITY AND THERMAL CONDUCTION IN A MON ATOMIC GAS. 281 small fraction of the average interval between two encounters. In these circum. |
|
W.J. Nellis S.T.Weir N.C.Holmes M. ROSS A.C.Mitchell U.S.-Japan
The continuous transition from a molecular to monatomic fluid means that temperature measurements indicates a dissociation fraction of about 5% at 140 ... |
|
Lecture 8: Free energy
calculation con rms that for a monatomic ideal gas the chemical potential is the Gibbs free energy per molecule. Note that we found that G = N for the ideal gas |
|
Untitled
An ideal monoatomic gas is compressed (no heat being added or removed in the Hint: the average speed of molecules at given temperature is given by:. |
|
Metallization and Electrical Conductivity of Hydrogen in Jupiter
The large magnetic field is produced in the molecular envelope duced primarily in the monatomic metallic ... pressures differ by a factor of 10). We ob-. |
|
Monatomic and Diatomic Gases
Pressure is proportional to the number of molecules per unit volume (N/V) and to the average translational kinetic energy of the molecules • This equation also |
|
CHAPTER 21 HEAT, WORK, AND THE FIRST - Physics Courses
43 A mixture of monatomic and diatomic gases has specific heat ratio y = 1 52 What fraction of the molecules are monatomic? |
|
TOPIC 1 ELEMENTS, COMPOUNDS AND MIXTURES
distillation of crude oil to obtain various fractions for petrol, lubricating oil, The only elements which occur naturally as diatomic molecules are hydrogen, |
|
Chapter 21 The Kinetic Theory of Gases File
The molecules occupy a negligible volume within the container When energy is added to a monatomic gas in a container with a fixed volume, all of the At a given temperature, the fraction of molecules with speeds exceeding a fixed |
|
LECTURE 11 Classical Ideal Monatomic Gas The ideal gas is
particle ideal monatomic gas is The integrals over the qi's gives a factor of V N The integrals over momenta is the partition function for a single molecule So |
|
Kinetic Theory Of Gases - SelfStudys
How many degrees of freedom have the gas molecules, if under standard molecules Thus For monoatomic gases, Q 71 Find the molar mass and the |
|
Chapter 19: Ideal Gases ( )
(a) The monatomic ideal gas undergoes three processes: (1) 1 1 The fraction of diatomic molecules in the mixture is just the number of diatomic moles divided |
|
Homework Week 4
The vibrational level spacing in the diatomic molecule N2 is 2330 cm−1 At room temperature, what fraction of the N2 molecules are vibrationally excited ( |
|
KINETIC THEORY - NCERT
diatomic molecule dvyanuka, three atoms form a tryanuka or a triatomic molecule ), their properties depending molecules are same, then the fraction of |
|
Chapter4-3439767pdf - HomeworkMarket
This equation is particularly applicable to ideal gases because the molecules are inde- pendent and The Translational Partition Function of an Atom in a Monatomic 12 gives the fraction of molecules in excited vibrational states for several |