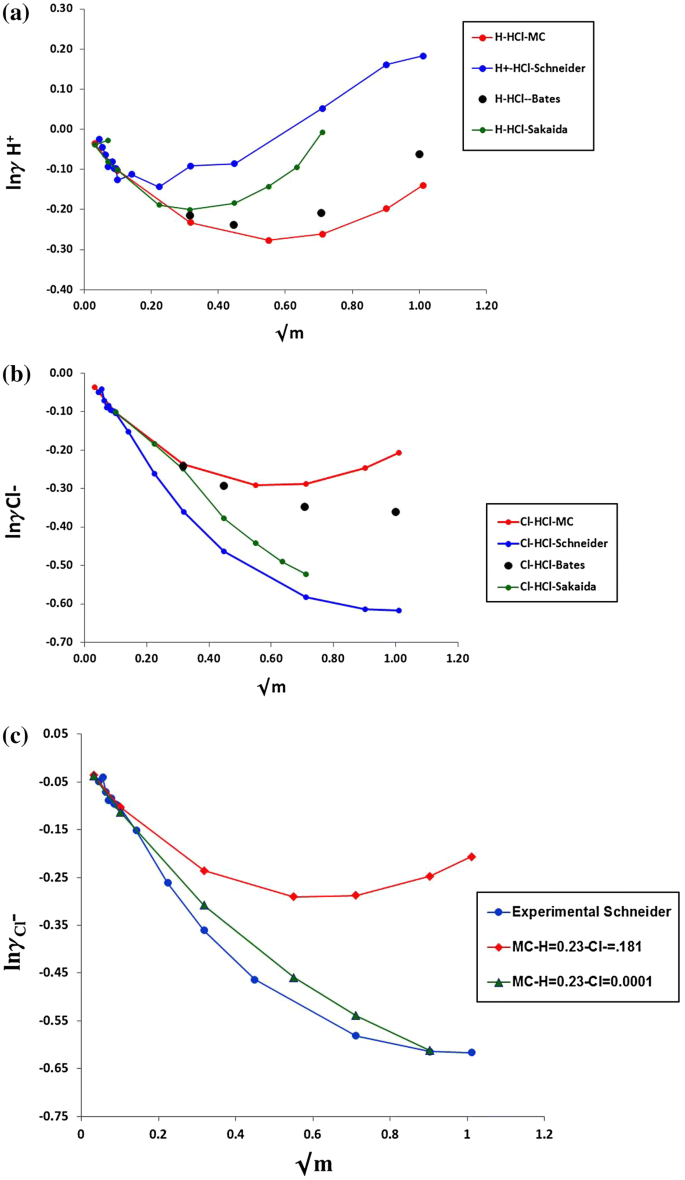
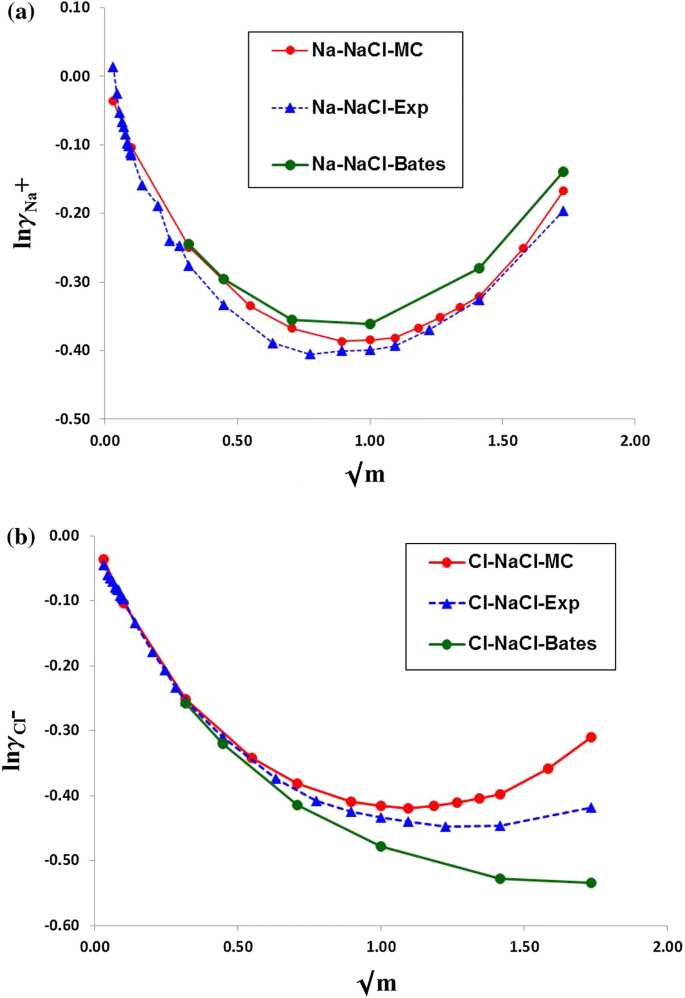
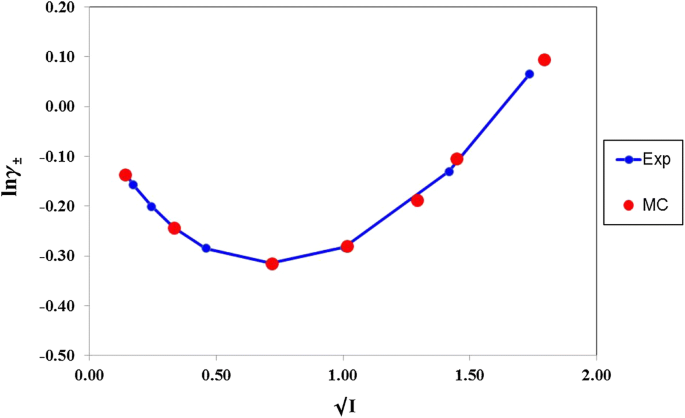
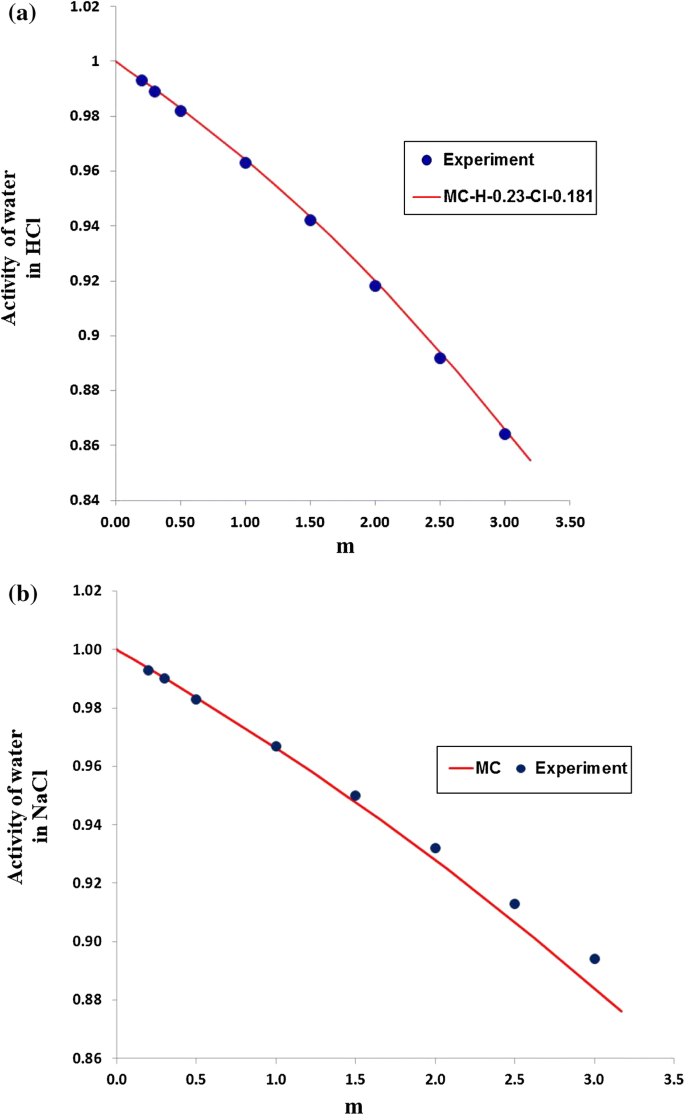
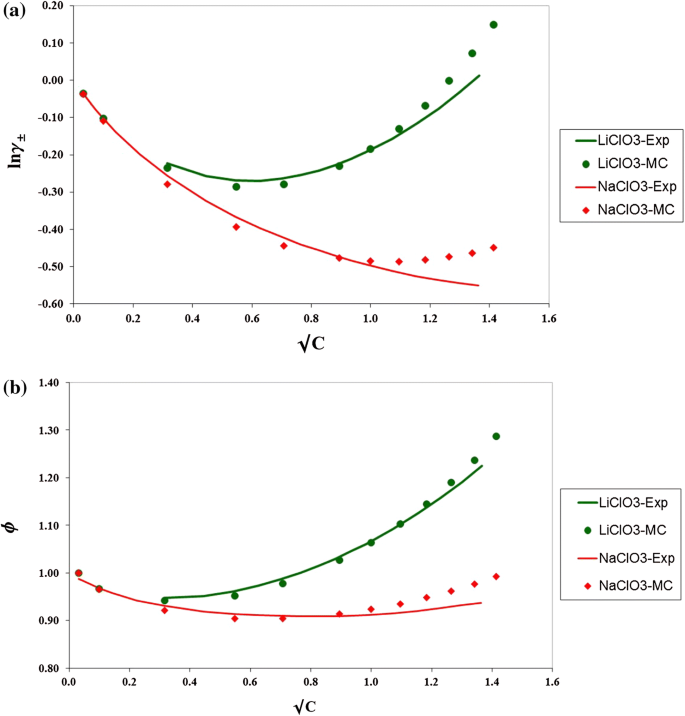
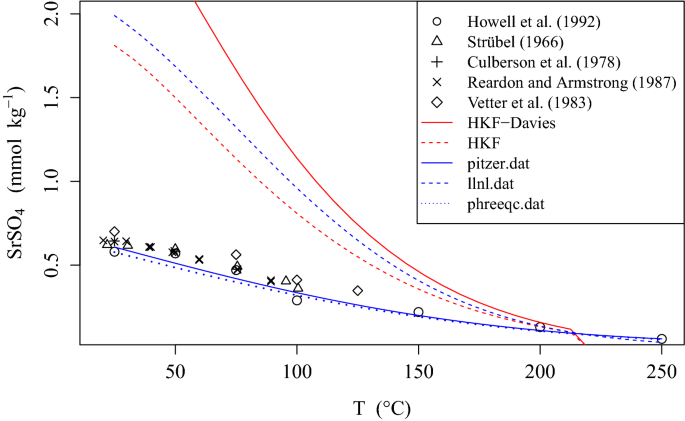
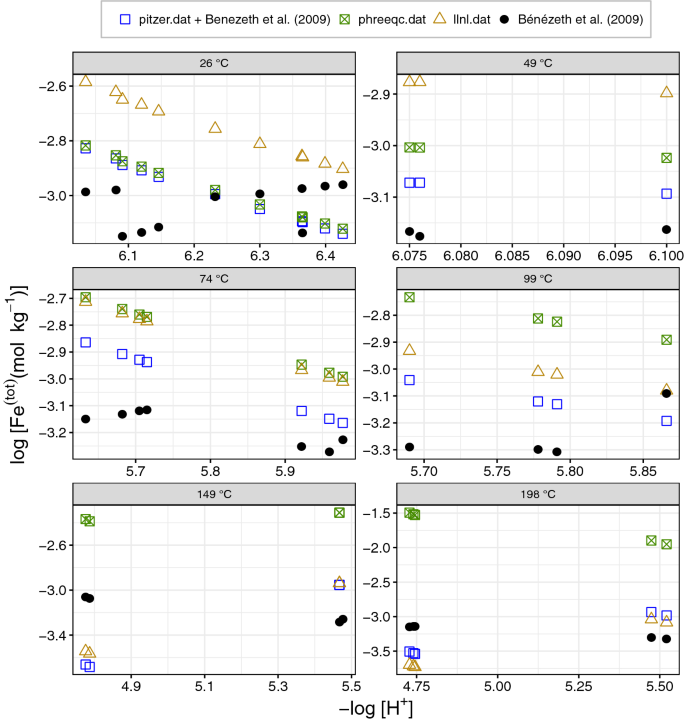
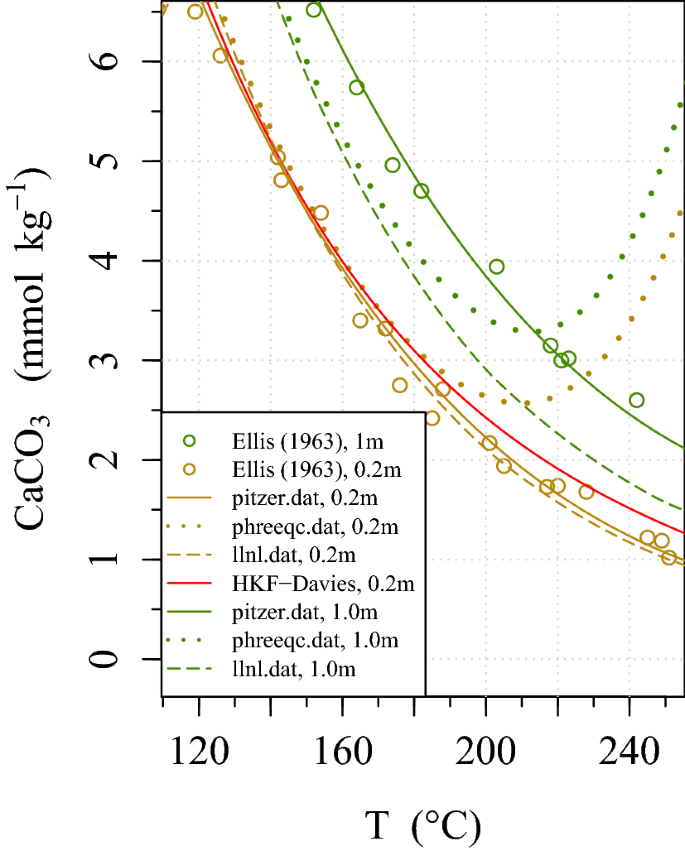
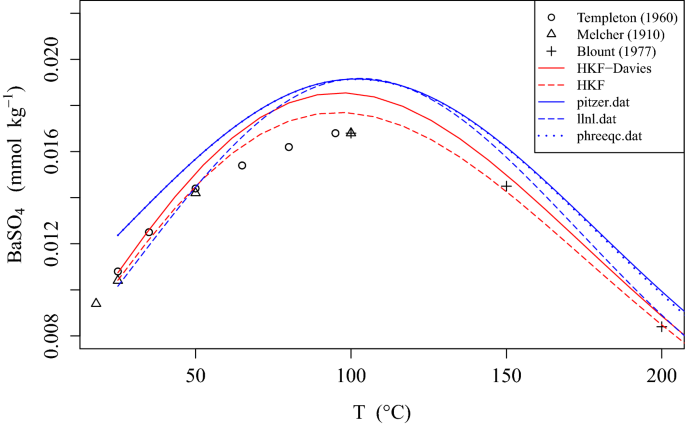
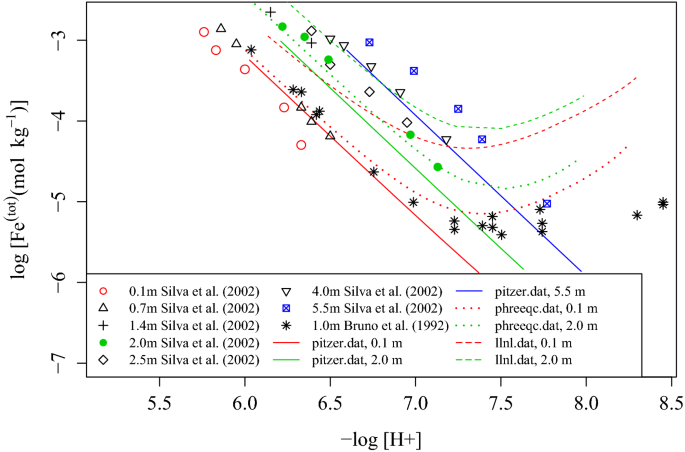
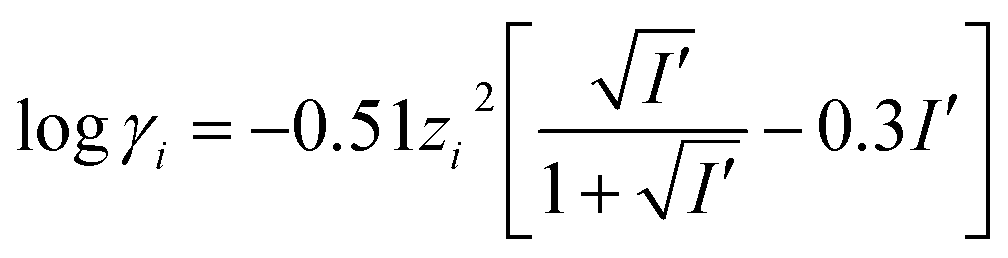davies equation to calculate activity coefficient
|
Chemical Oceanography
Use Davies Equation to calculate activity coefficients for all ions of interest From Davies Equation H+ activity coefficient ln γ = -A Z2 [I0 5/(1 + I0 5) |
|
Chapter 6 Activity Scales and Activity Corrections
11 oct 2004 · This equation predicts that the log of the activity coefficient decreases For example: If we use the Davies equation to calculate γi for |
|
3 Activity Coefficients of Aqueous Species 31 Introduction
The Davies equation predicts a unit activity coefficient for all neutral solute species In EQ3/6 activity coefficients are first calculated from the “raw” |
However, the mean activity coefficient γl can be measured for any salt.
The mean activity coefficient is determined from measurements of such properties of solutions as freezing point, vapor pressure, solubility, and electrical potential.
What is the formula for the mean activity coefficient?
The mean activity coefficient is defiend as follows: γ±=(γ+γ−)1/2.
What is the debye Huckel equation for activity coefficient?
Debye Huckel Limiting Law
To measure the activity a\\[_{C}\\] of an ion C in a solution, the concentration and activity coefficient must be known: a\\[_{C}\\] = γ \\[\\frac{[C]}{[C^{\\Theta}]}\\], This is also called the debye huckel limiting law equation.
How do you calculate the activity coefficient?
In this activity coefficient equation, the activity coefficient is calculated by multiplying the square root of the ionic strength by the total electric charge of both positive and negatively charged ions in solution (times a constant).
|
Chapter 6 Activity Scales and Activity Corrections
11 oct. 2004 6.1 Total Activity Coefficient: Electrostatic Interactions and Ion Complexing ... For example: If we use the Davies equation to calculate ?i. |
|
Chemical Oceanography
Where ?+ is the mean ion activity coefficient Calculate ionic strength from concentrations of ... Use Davies Equation to calculate activity coefficients ... |
|
Activity corrections for ionic equilibria in aqueous solutions
ledge of activity coefficients. estimate single-ion activity coefficients at 25OC e.g. |
|
3. Activity Coefficients of Aqueous Species 3.1. Introduction
els by which to compute the activity coefficients of the solute species and the The Davies equation predicts a unit activity coefficient for all neutral ... |
|
Thermodynamic Consistency in the Modeling of Speciation in Self
5 sept. 2017 the complex formation constants and of the Davies equation to compute the activity coefficients of the species in solution. |
| CNWRA 92-020 MARIANA -- A Simple Chemical Equilibrium |
|
Implementing the Mean Spherical Approximation model in the
30 nov. 2020 Equations like the Davies equation are semi-empirical and do not give ... The first solution to calculate activity coefficients for non-zero ... |
| CNWRA 92-020 MARIANA -- A Simple Chemical Equilibrium |
|
Chemical Oceanography
Where ?+ is the mean ion activity coefficient Calculate ionic strength from concentrations of ... Use Davies Equation to calculate activity coefficients ... |
|
Synergistic use of siderophores and weak organic ligands during
The Davies model has been used extensively in soil studies to calculate activity coefficients of electrolytes at fairly low ionic strengths33. The equation |
|
Chapter 6 Activity Scales and Activity Corrections
11 oct 2004 · 6 6 A 3 Davies Equation 6 6 A 3 Davies Equation log γi = -A zi2 {I1/2 / (1 + I1/2 ) - 0 2 I} ; for I < 0 5 (almost seawater) In this equation all ions of the same charge have the same value of γi there is no ion size parameter monovalent ions = γi divalent ions = γi trivalent ions = γi |
|
Ion Activity, Ion Association and Solubility
Activity coefficient calculations • Implementation in A QUICK REVIEW OF ACTIVITY COEFFICIENT FORMULAE logγ i =−Azi 2 Davies Equation Low ionic |
|
3 Activity Coefficients of Aqueous Species 31 Introduction
els by which to compute the activity coefficients of the solute species and the The Davies equation predicts a unit activity coefficient for all neutral solute |
|
Activity and Concentration
equation • Davies Equation for higher ionic strengths ( |
|
Lecture 6 - SOEST Hawaii
equation Notice that the Davies approximation has no solute- specific size parameterization (ai) whereas mean ionic activity coefficients in MgCl2 solution as a function of Let's calculate true activity of Ca2+ in a 0 05 m solution of CaCl2 at |
|
Chemical Oceanography
Calculate ionic strength from concentrations of all ions in solution using I = 0 5 Σ Z2 m Use Davies Equation to calculate activity coefficients for all ions of interest |
|
MARIANA -- A Simple Chemical Equilibrium Module, Version 10
Activity coefficients can be calculated by the B-dot equation, the Davies equation, or assumed to be equal to one (ideal solution) 2 Page 8 2 THEORY Mariana |