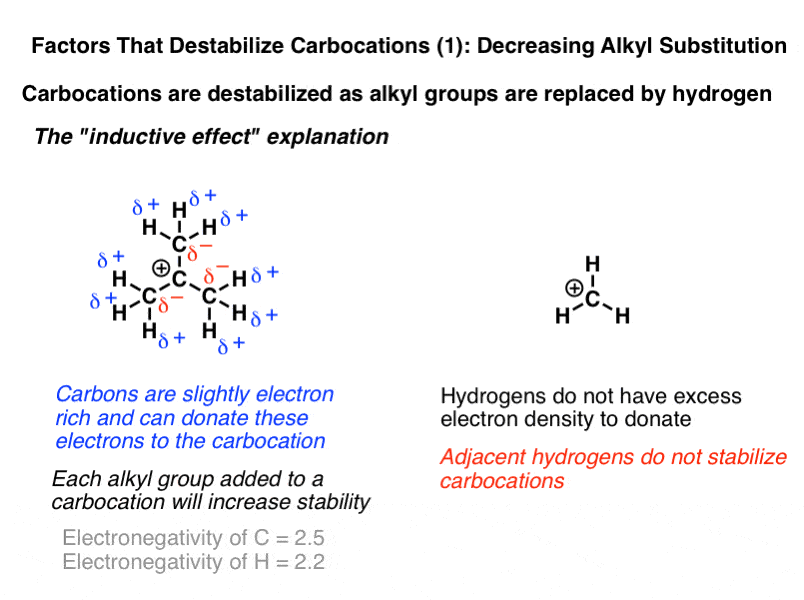
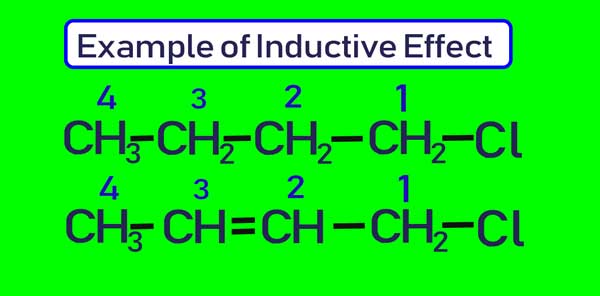
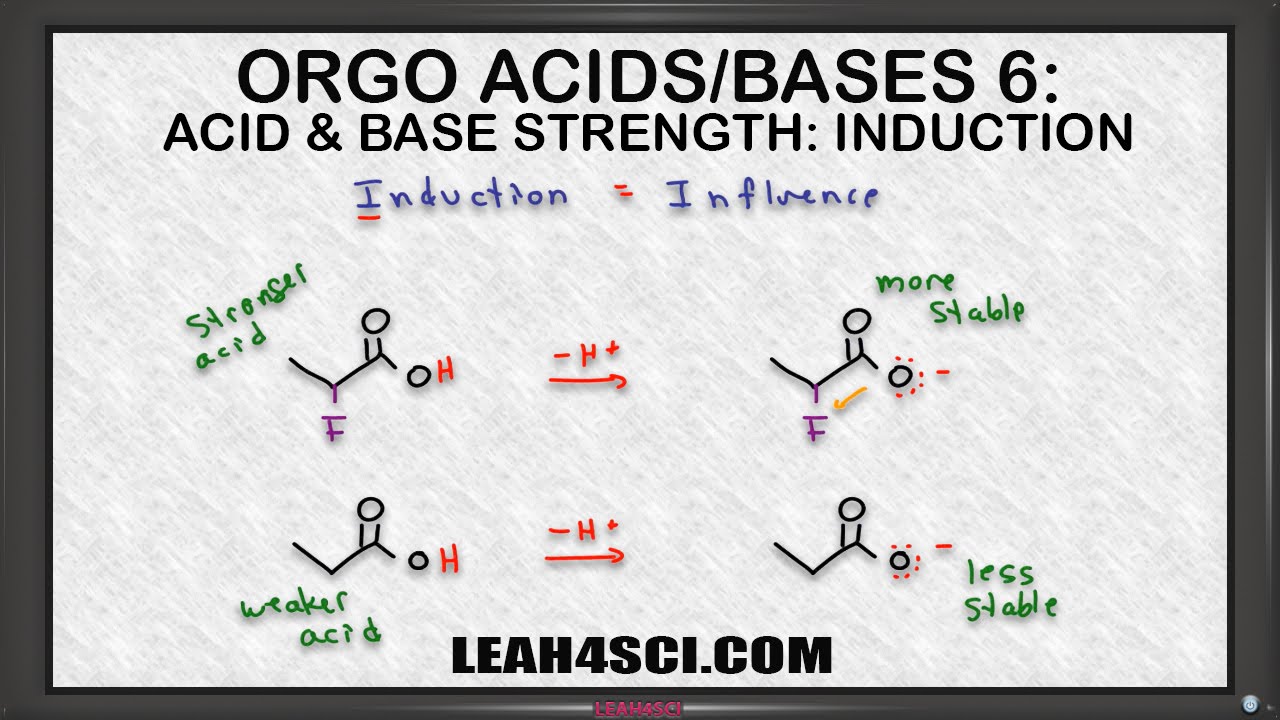
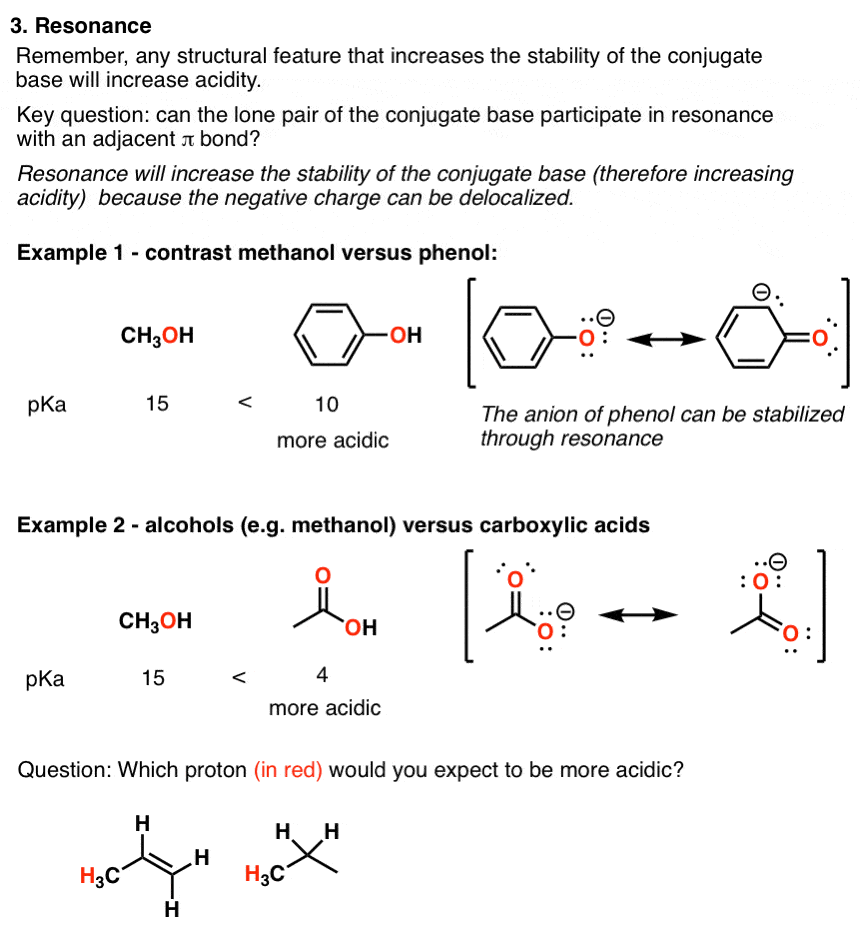
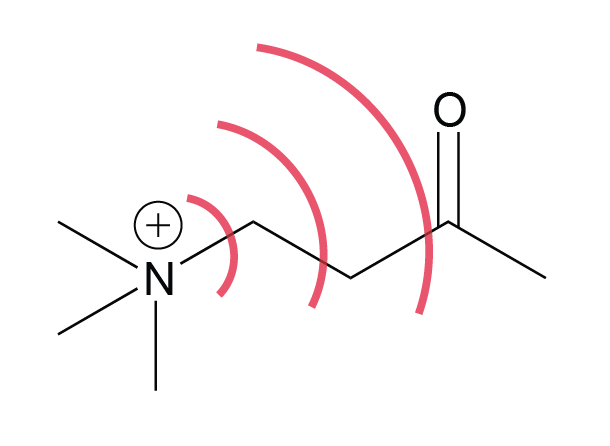
explain inductive effect
|
Example Question involving substituent effects (inductive vs
Discuss and explain the inductive and resonance effects. The methoxy group is electron withdrawing by the inductive effect of the oxygen atom ... |
|
IR Spectroscopy (Module-1I) Lecture Notes: Dr. Diksha Katiyar
three Cartesian coordinates x y and z |
|
Inductive effect is defined as - permanent displacement of shared
permanent displacement of shared electron pair in a carbon chain towards more electronegative atom or group. Page 2. Types of Inductive effect : 1.Negative |
|
The role of resonance and inductive effects in the acidity of
acid group. The above discussionhas shown that the inductive effect of the carbonyl group suffices to explain most of the enhanced acidity. |
|
Chemistry Questions and Answers – Inductive Effect 1
Electronegativity is defined as the power of an atom in a molecule to ______ Explanation: Inductive effect is the permanent electron displacement effect ... |
|
Factors affecting vibrational frequencies and IR Spectroscopy of
4. The Shape of Absorption Bands. 5. Factors Influencing Vibrational Frequencies. 5.1 Effect of Bond Order. 5.2 Resonance and Inductive Electronic Effect. |
|
Inductive effect
11-Mar-2021 The explanation for the above is given by the fact that more charge on an atom decreases stability and less charge on an atom increases. |
|
120 Chapter 24: Phenols. Alcohols contain an OH group bonded to
and stabilizes the negative charge through inductive effects. Page 2. 121. 236. Resonance effect: the benzene ring stabilizes the the phenoxide. |
|
Evidence for a Solid-Electrolyte Inductive Effect in the Superionic
07-Dec-2020 35 that an inductive effect might explain the observed lithium con- ductivity trend in Li10MP2S12 solid-electrolyte inductive effects have been ... |
|
ORGANIC CHEMISTRY – SOME BASIC PRINCIPLES AND
explain the influence of Explain why the names given in the parentheses are incorrect. ... ?-bond is referred to as the inductive effect. |
|
Inductive effect - LS College Muzaffarpur
11 mar 2021 · In chemistry the inductive effect is an effect regarding the transmission of unequal sharing of the bonding electron through a chain of |
|
INDUCTIVE EFFECT POSITIVE NEGATIVE ILLUSTRATIONS
The polarization of a ? bond due to electron withdrawing or electron donating effect of adjacent groups or atoms is called inductive effect |
|
The Inductive Effect in Organic Chemistry - UNL Digital Commons
The present paper is concerned with the ability of alkyl groups to serve as sigma inductive electron donors The “inductive effect” remains an icon of chemical |
|
CONCEPTS IN ORGANIC CHEMISTRY - KEA
Inductive effect is useful in explaining the strength of some organic acids and bases a) Effect of substituent on the acid strength of aliphatic acids HCOOH > |
|
Principles Of Drug Action 1 Spring 2005 Resonance and Induction
Induction or the inductive effect of an atom or functional group is a function of that groups 1) electronegativity 2) bonding order and charge and 3) |
|
Resonance and induction tutorial
Induction or the inductive effect of an atom or functional group is a Resonance may be defined as bonding or sharing of electrons between more than two |
|
Resonance and Inductive Effects
(v) Inductive effects are caused by differences in electronegativity between bonded atoms which leads to a polarisation of the bond (vi) The inductive effects |
|
BSc Chemistry - e-PG Pathshala
MODULE 1: Revisit to Electronic Effects: Inductive and has been demonstrated by the fact that it is commonly used to explain the properties like origin |
|
Types of Inductive Effect Applications Stability - Byjus
The inductive effect refers to the phenomenon wherein a permanent dipole arises in a given molecule due to the unequal sharing of the bonding electrons in |
What is an inductive effect explain?
In chemistry, the inductive effect in a molecule is a local change in the electron density due to electron-withdrawing or electron-donating groups elsewhere in the molecule, resulting in a permanent dipole in a bond.What is inductive effect and types of inductive effect?
The inductive effect is the phenomenon in which a permanent dipole forms in a given molecule due to unequal bonding electron sharing. It also creates a polarised state that lasts longer. There will be two dissimilar atoms in a sigma bond, and the electron density will also not be uniform.What is inductive effect and its characteristics?
The inductive effect, often known as “the -I Effect,” is a gap-established phenomenon in which the pricing of a chemical bond impacts the orientation of adjacent bonds in a molecule, resulting in an eternal kingdom of polarisation. The electron density isn't homogeneous when atoms from two components make a bond.- There are two types of inductive effect i.e. – I effect and +I effect.
|
Inductive Effects
The terms "resonance" and "induction" refer to the electronic effects that atoms or functional groups may have within a compound These effects are defined |
|
Inductive Effect
Inductive Effect • When an electron-donating or an electron-withdrawing group or species is introduced to a carbon chain of atoms their corresponding negative |
|
INDUCTIVE EFFECTS IN A COVALENT BOND Inductive Effect
effects which influence the distribution of electrons in a covalent bond of an For basicities of the amines, again inductive effect can be used to explain the order |
|
INDUCTIVE EFFECT POSITIVE NEGATIVE EXPLANATION
1) Negative inductive effect (-1): The electron withdrawing nature of groups or atoms is But none of them explains all the observed properties of the molecule |
|
Inductive effect and Hyperconjugation
Inductive effects are electronic effects that occur through sigma σ bonds A second explanation for the observed trend in carbocation stability is based on orbital |
|
PDF :8 - BSc Chemistry
Module No and Title Module 1: Revisit to Electronic Effects: Inductive and has been demonstrated by the fact that it is commonly used to explain the properties |
|
Research Article Topological Model on the Inductive Effect - CORE
19 fév 2014 · effects in chemistry [1] According to the International Union of Pure and Applied Chemistry (IUPAC) the inductive effect is defined as |