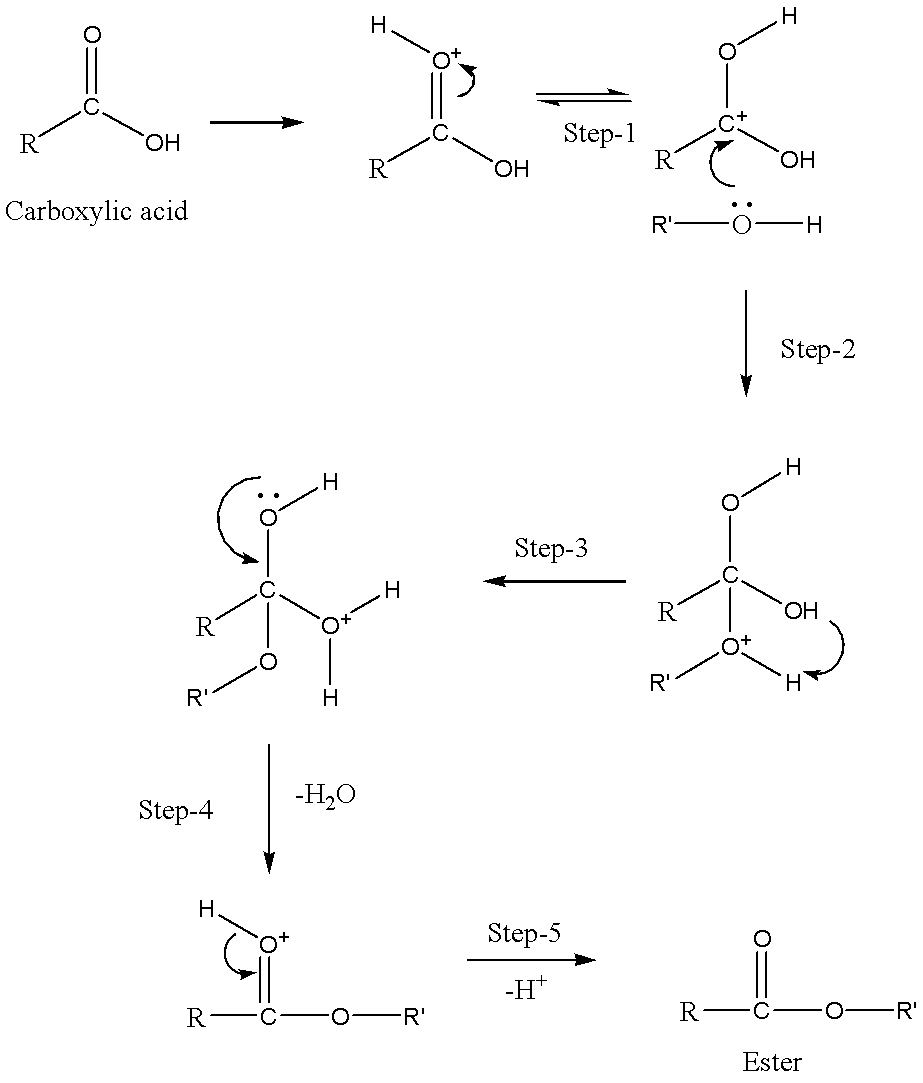
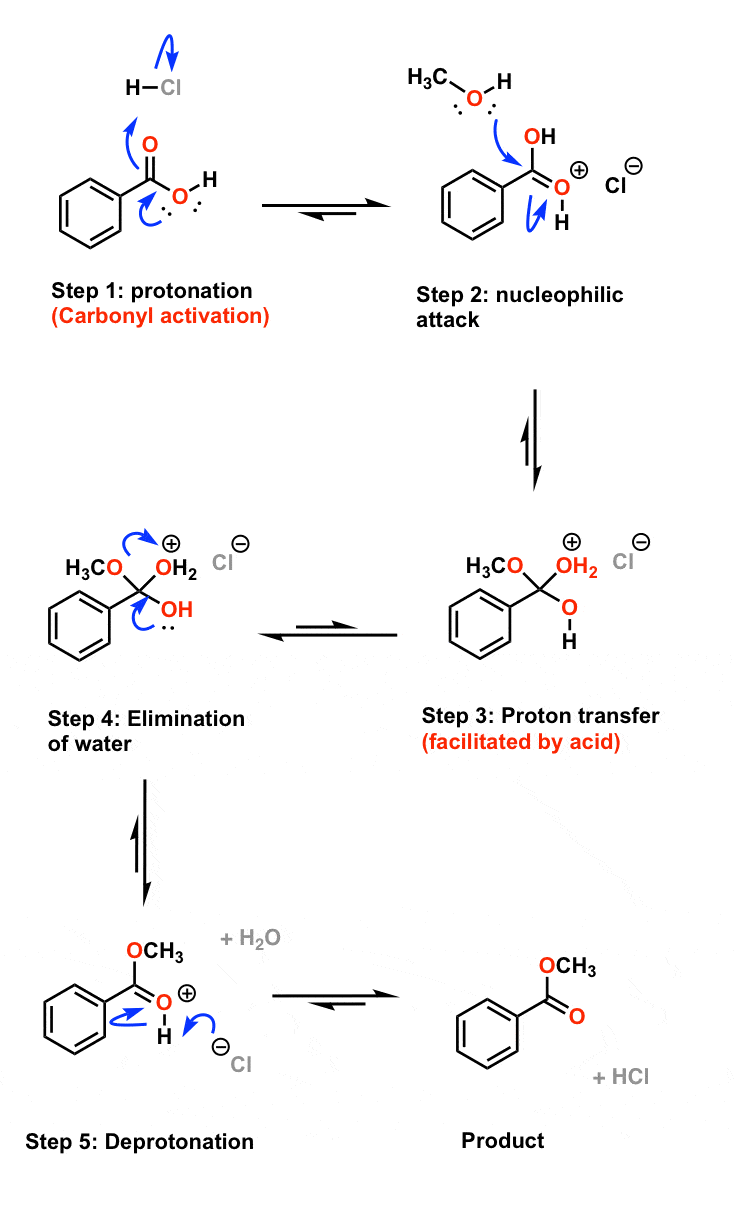
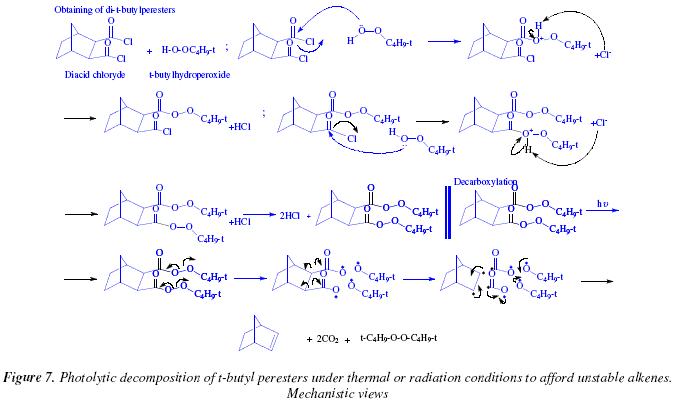
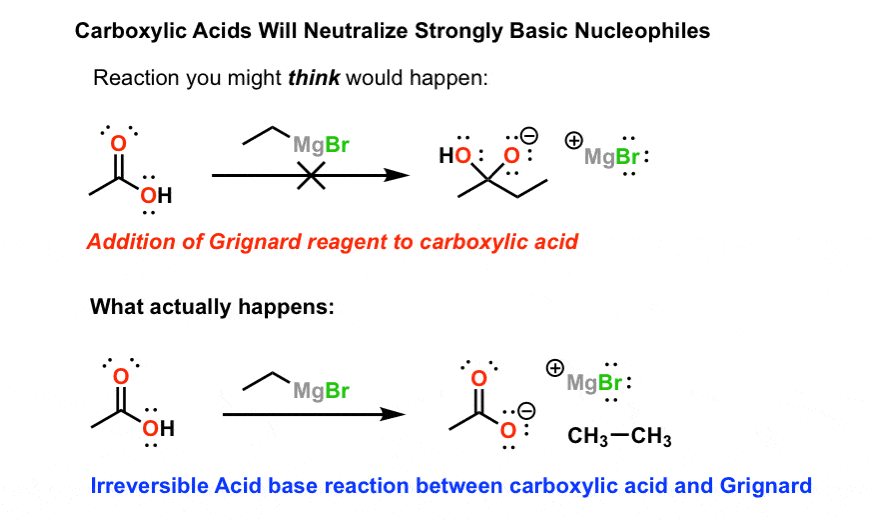
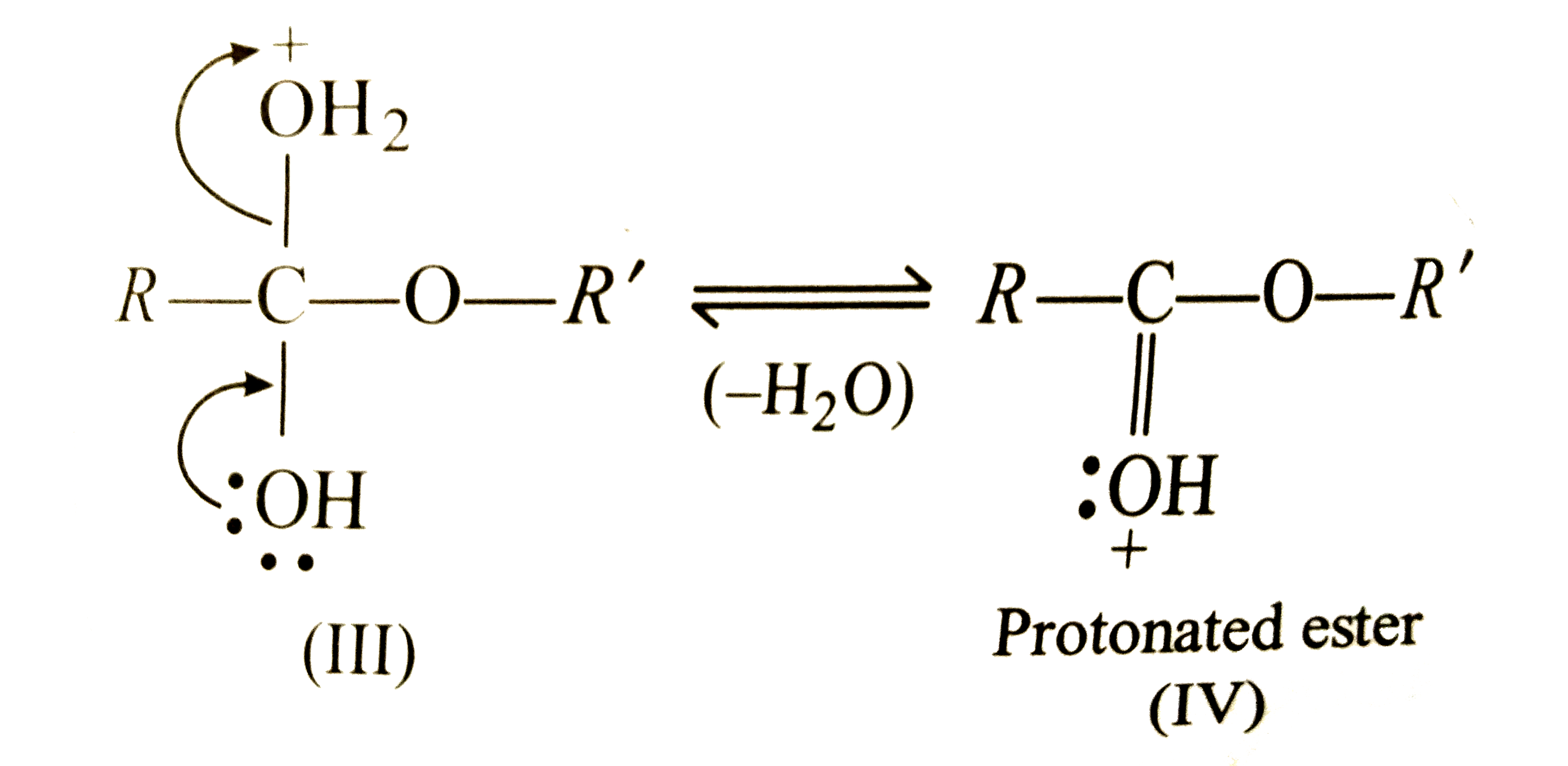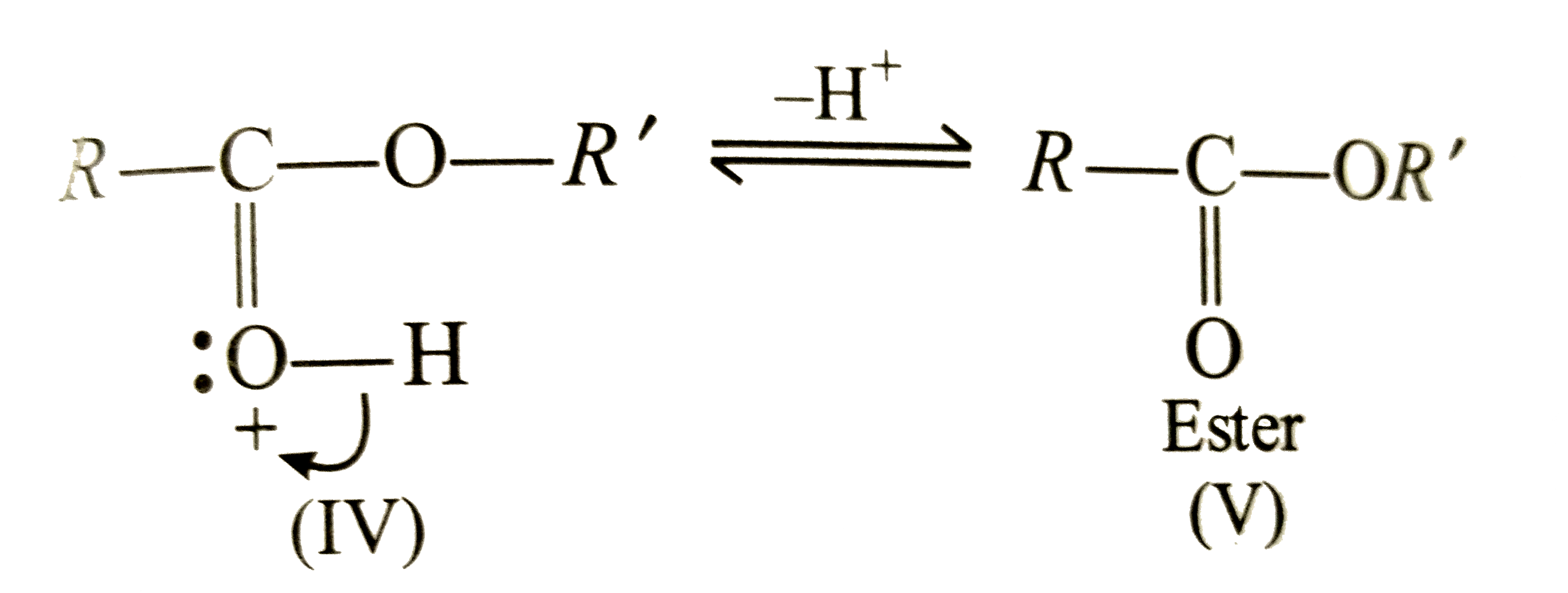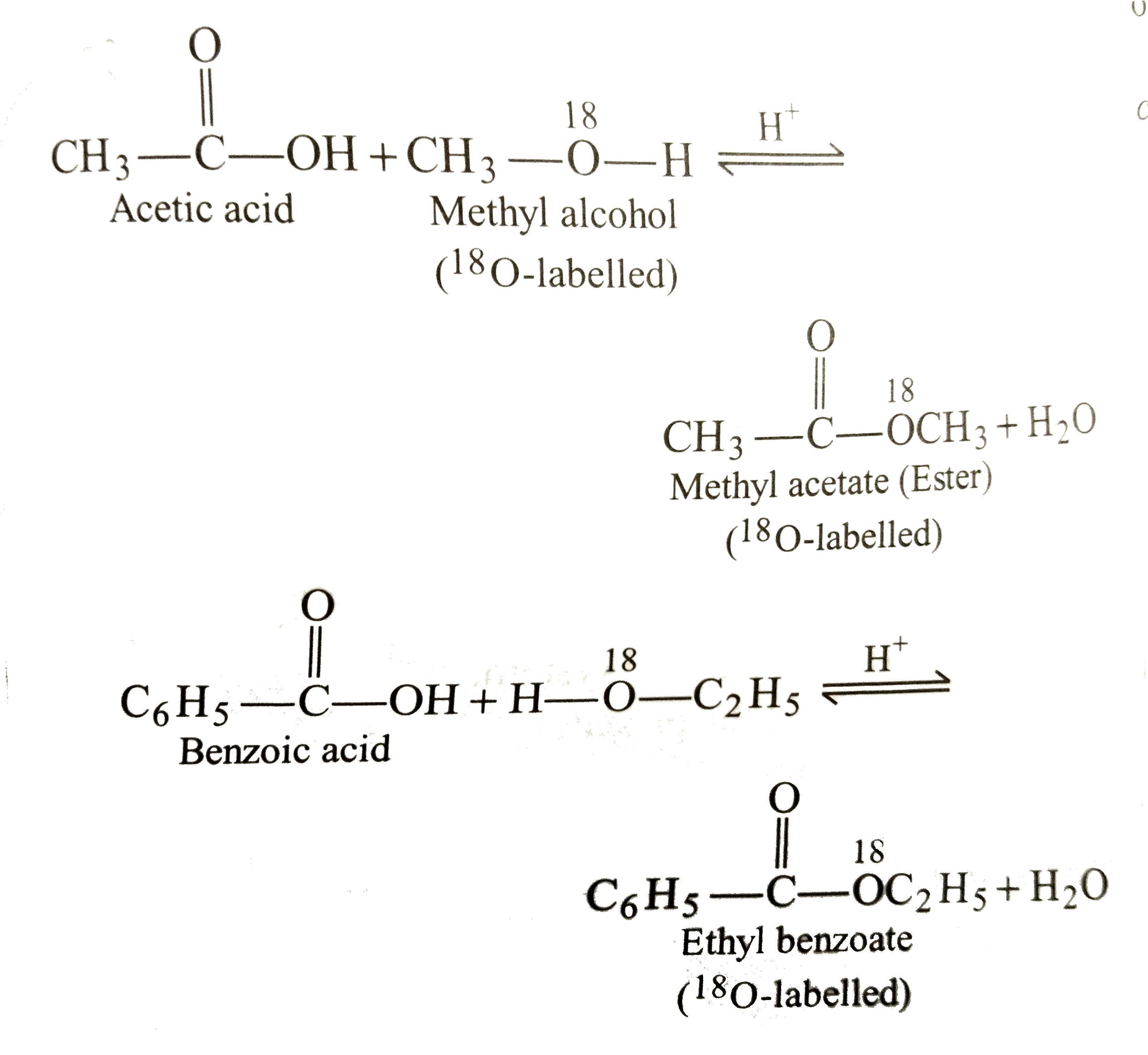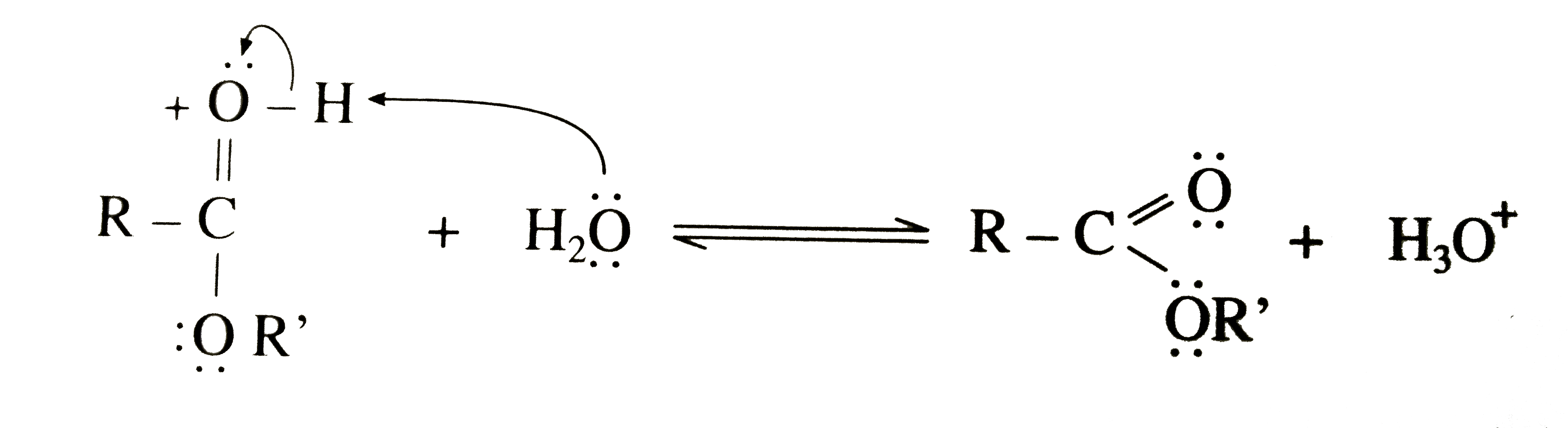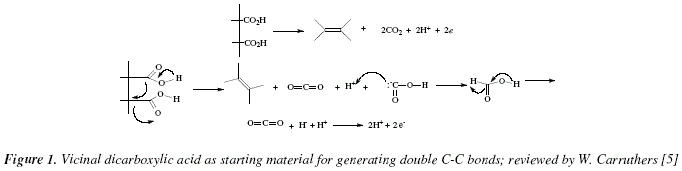explain the mechanism of esterification of carboxylic acid
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to predict the products of ester synthesis and hydrolysis reactions. |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
correlate physical properties and chemical reactions of aldehydes ketones and carboxylic acids |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
explain the mechanism of a few selected reactions of aldehydes describe the uses of aldehydes ... carboxylic acids |
|
Carboxylic acids from limonene oxidation by ozone and OH radicals
mechanisms are suggested to fill-in the knowledge gaps. the anti – CI* is decomposition via the ester or the “hot acid” channel where an energy-rich ... |
|
Studies on the BAL2 mechanism for ester hydrolysis
From mass spectral analysis of the carboxylic acid products it is concluded that the former substrate is hydro- lyzed exclusively by the BAc2 mechanism |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones
Explain reaction mechanisms associated with each type of functional group Carboxylic acid derivatives: • Esters. • Anhydrides. • Acid halides. • Amides. |
|
Www.rsc.org/pccp
Acid-Catalyzed Carboxylic Acid Esterification and Ester Hydrolysis Mechanism: Acylium Ion as a Sharing Active Intermediate via a Spontaneous Trimolecular. |
|
CARBOXYLIC ACIDS
Carboxylic acids are organic compounds containing the carboxyl group (-COOH) wherein the hydroxyl group Mechanism of Esterification with Diazomethane. |
|
An Introduction to Pectins: Structure and Properties
In a l l natural pectins some of the carboxyl groups are i n the methyl ester form. Depending on the isolation conditions |
|
Revisiting the mechanisms of low-temperature base-catalysed ester
If the ester reacts with a different carboxylic acid the ester interchange reaction is called acidolysis |
|
Chapter 5 Carboxylic Acids and Esters - Angelo State University
The simplest way to synthesize an ester is to heat a carboxylic acid with an alcohol or phenol (plus an acid catalyst); the oxygen of the alcohol adds to the |
|
Esterification (Alcohol & Carboxylic acid) - Reactions Mechanism
Esterification is the process of combining an organic acid (RCOOH) with an alcohol (ROH) to form an ester (RCOOR) and water; or a chemical reaction |
|
CARBOXYLIC ACIDS
Diazomethane can be written in two resonance stabilized forms Page 41 Mechanism of Esterification with Diazomethane 45 Although the carboxylate |
|
Write the mechanism involved in the esterification of a carboxylic
Complete answer: We can define esterification as the process which involves combination of an organic acid (RCOOH group) and an alcohol (ROH group) that leads |
|
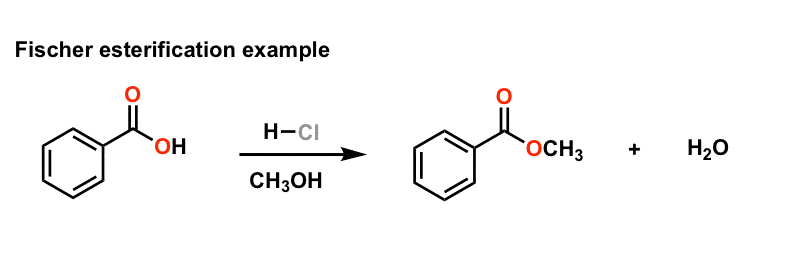
Fischer Esterification - Carboxylic Acid to Ester Under Acidic
16 nov 2022 · The Fischer esterification is the conversion of a carboxylic acid to an ester under acidic conditions It is a robust method for ester |
|
Mechanism for the esterification reaction - Chemguide
This page looks in detail at the mechanism for the formation of esters from carboxylic acids and alcohols in the presence of concentrated sulphuric acid acting |
|
The Esterification of Carboxylic Acid with Alcohol over Hydrous
The mechanism of esterification over hydrous zirconium oxide was also studied in vapor-phase reactions Experimental Materials Commercial reagents were used |
|
Explain the mechanism of esterification of carboxylic acid (Feb 16)
The esterification of carboxylic acids with alcohols is a nucleophilic acylic substitution The Postée : 26 avr 2023 |
|
Explain the mechanism of esterification carboxylic acids - Doubtnut
Mechanism of estrification of carboxylic acids : It is a kind of nucleophilhc acyl substitution The mechanism of esterification involves |
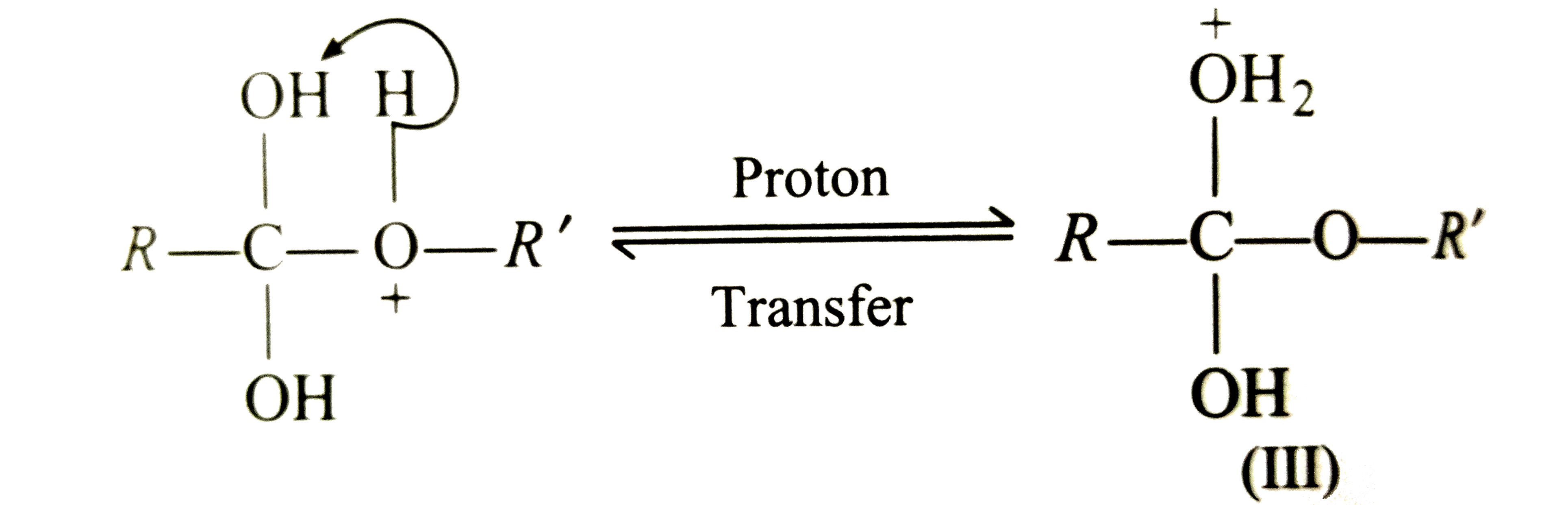
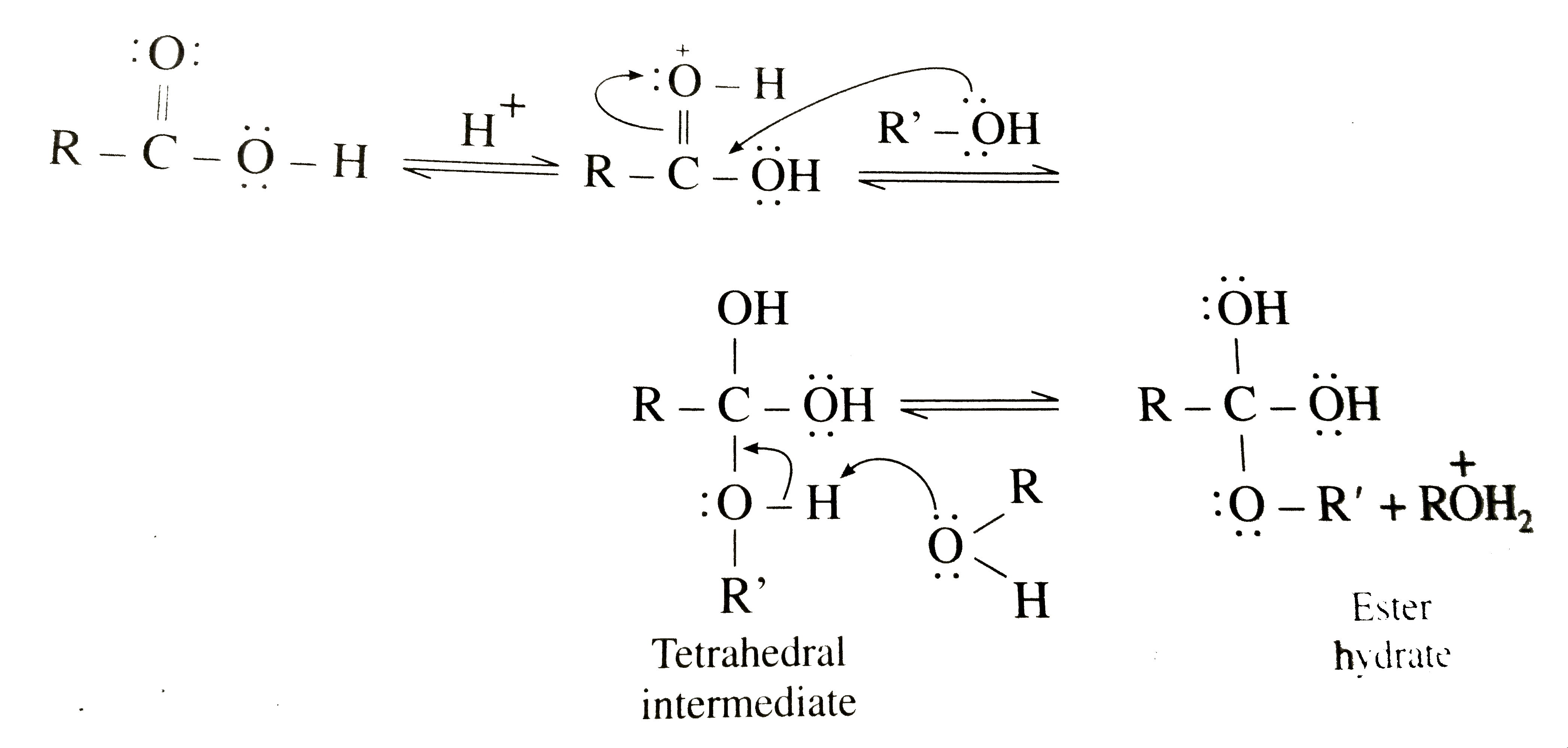
What is the mechanism of esterification of a carboxylic acid?
The esterification of carboxylic acids with alcohols is a nucleophilic acylic substitution . The carboxylic acid is protonated on its carboxyl oxygen atom . Alcohol acts as a nucleophilic and attacks carboxyl carbon . Loss of proton gives ester hydrate.26 avr. 2023What is the mechanism of carboxylic acid?
In general, carboxylic acids undergo a nucleophilic substitution reaction where the nucleophile (-OH) is substituted by another nucleophile (Nu). The carbonyl group (C=O) gets polarized (i.e. there is a charge separation), since oxygen is more electronegative than carbon and pulls the electron density towards itself.What is the mechanism of carboxylic acid alcohol?
Carboxylic acids can react with alcohols to form esters in a process called Fischer esterification. An acid catalyst is required and the alcohol is also used as the reaction solvent. The oxygen atoms are color-coded in the reaction below to help understand the reaction mechanism.- In the first step, the ethanoic acid takes a proton (a hydrogen ion) from the concentrated sulphuric acid. The proton becomes attached to one of the lone pairs on the oxygen which is double-bonded to the carbon.
|
Studies of ester formation on a cellulose matrix - DiVA
The formation of ester cross-links in cellulosic or lignocellulosic esterification is one of the reactions involved in the autoadhesion mechanism The term hornification is used to describe the irreversible structural changes that occur in |
|
211 Introduction Carboxylic Acids 211 Introduction Carboxylic
25 avr 2012 · Carboxylic acid derivatives, such as vinyl acetate, are very common, and they play a How can induction and resonance be used to explain the acidity of a Each step of the Fischer esterification mechanism is equilibrium |
|
Carboxylic acids and their derivatives - Caltech Authors
fatty-acid chains and one phosphate-ester residue One of the Exercise 18-1 Explain why the proton line position of the acidic hydrogen of a car- boxylic acid mechanism occurs in the formation of alkyl chlorides from alcohols and thionyl |
|
Fischer Esterification - MIT OpenCourseWare
Organic Synthesis: Synthesizing an ester from an acid catalyzed reaction of a In the next section are detailed mechanisms for common fragmentation and ionization What is the physical meaning of a negative gyro magnetic constant? |
|
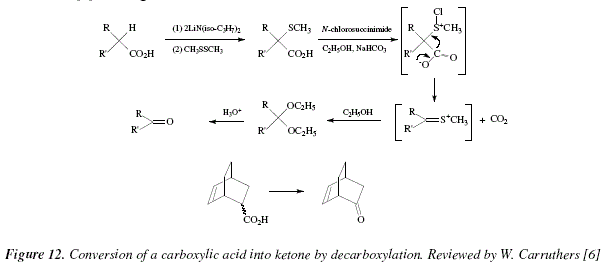
Carboxylic Acid Derivatives Addition/Elimination
The ester is therefore the ethyl ester of benzoic acid, it is called ethyl (from the alcohol) hybridized carbon atom, hence the addition/elimination mechanism To identify what is required to react with the acid chloride to make the amide, |
|
Carboxylic acid deri
O O O carboxylic acid ester amide acid chloride acid anhydride thioester acyl phosphate The mechanism of nucleophilic acyl substitution involves two critical |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
Explain reaction mechanisms associated with each type of functional group Another route to esterification of carboxylic acids involves one of the other types of |