den160026
|
Package Insert 23andMe® Personal Genome Service® (PGS)
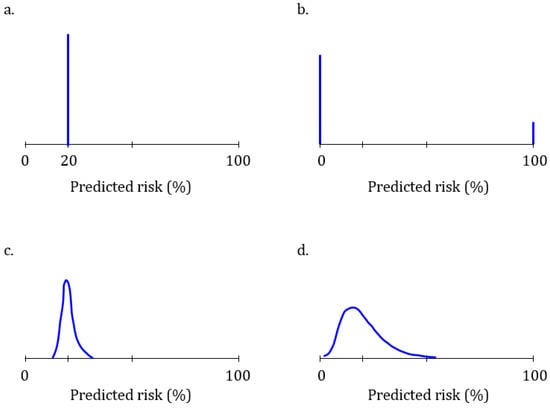
comprehension study for the PGS Genetic Health Risk Test Reports (DEN160026) User comprehension studies were performed to assess how well people understand |
|
Food and Drug Administration
Food and Drug Administration |
|
Den160026pdf
The Decision Summary originally issued April 6 2017 A DEN Number: DEN160026 B Purpose for Submission: De Novo request for the 23andMe Personal Genome |
|
Introduction to FDAs Role in Regulation of Alzheimers Diagnostic
2 mai 2019 · DEN160026: 23andMe Genetic Health Risk Test 1 Detect APOE4 variants in gDNA 2 APOE4 genotype associated with increased risk of late-onset |
|
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
DEN160026 23andMe Personal Genome Service (PGS) Test Evaluation of Automatic Class III Designation – De Novo Request Regulation Number: 21 CFR 866 5950 Regulation Name: Genetic Health Risk |
Consumer-Initiated Test Types
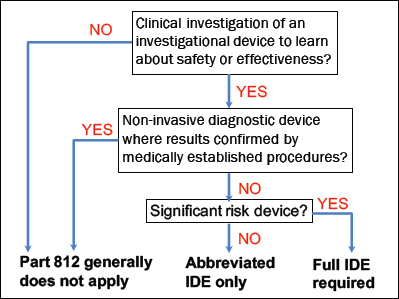
Consumer-initiated (CI) genetic health risk (GHR) tests are marketed to the public and ordered by patients themselves. But not all CI tests are alike. CI tests generally follow one of two pathways to consumers: 1. Direct-to-Consumer(DTC) tests—marketed directly to consumers with no healthcare professional involved. These tests are regulated by the
FDA DTC Authorization Was In-Hand
Just shy of one year after launching their physician-mediated model, in mid-August 2020, Ancestry received an FDA 510(k) approval for their Factor V Leiden Genetic Health Risk Test (K192944). In the 510(k) submission, they demonstrated the test is substantially equivalent to a previously approved device; the 23andMe Personal Genome Service (PGS) Ge
Soundrocket User Comprehension Study
Soundrocket partnered with the AncestryHealth team in the development and user comprehension testingof the Factor V Leiden test report. Through a multi-stage iterative effort, SoundRocket guided improvements to the test report while also designing and implementing a user comprehension study which demonstrated the illusive 90% comprehension in key a
|
1 EVALUATION OF AUTOMATIC CLASS III DESIGNATION FOR The
DEN160026. B. Purpose for Submission: De Novo request for the 23andMe Personal Genome Service (PGS) Genetic Health Risk Test for Hereditary Thrombophilia |
|
1 DEPARTMENT OF HEALTH & HUMAN SERVICES November 02
Nov 2 2017 Re: DEN160026. 23andMe Personal Genome Service (PGS) Test. Evaluation of Automatic Class III Designation – De Novo Request. |
|
The Helix Genetic Health Risk app for late-onset Alzheimers disease
Dec 23 2020 Disease (DEN160026). Similarities and. Differences. Intended Use /. Indications for use. The Helix Genetic. Health Risk App. |
|
K192920
Jan 14 2020 23andMe PGS (DEN140044 |
|
23andMe PGS Genetic Risk Report for Hereditary Prostate Cancer
Jan 6 2022 kit used to generate performance data for DEN140044 |
|
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION
DEN160026. Device Trade. Name. The Helix Genetic Health Risk App for late-onset Alzheimer's disease. 23andMe PGS Genetic Health. Risk Report for Late-onset. |
|
23andMe® Personal Genome Service® (PGS) Package Insert For in
comprehension study for the PGS Genetic Health Risk Test Reports (DEN160026). User comprehension studies were performed to assess how well people understand |
|
510(k) Substantial Equivalence Determination Decision
User comprehension studies were performed to assess the comprehension of the. Genetic Health Risk report. See DEN160026 supportive user comprehension studies. |
|
Proprietary Algorithms for Polygenic Risk: Protecting Scientific
Jun 13 2019 https://www.accessdata.fda.gov/cdrh_docs/pdf16/den160026.pdf (accessed on 10 June 2019). 15. 23 and Me Personal Genome Service Genetic ... |
|
Introduction to FDAs Role in Regulation of Alzheimers Diagnostic
May 2 2019 DEN160026: 23andMe. Genetic Health Risk. Test. 1. Detect APOE4 variants in gDNA. 2. APOE4 genotype associated with. |
|
1 EVALUATION OF AUTOMATIC CLASS III DESIGNATION - FDA
The Decision Summary originally issued April 6, 2017 A DEN Number: DEN160026 B Purpose for Submission: De Novo request for the 23andMe Personal |
|
1 DEPARTMENT OF HEALTH & HUMAN SERVICES - FDA
2 nov 2017 · Re: DEN160026 23andMe Personal Genome Service (PGS) Test Evaluation of Automatic Class III Designation – De Novo Request |
|
Submissions received and TGA response: Review of the regulation
21 nov 2019 · The test is most relevant for people of Ashkenazi Jewish descent 23andMe PGS Genetic Health Risk Test (DEN160026) 23andMe, Inc The |
| [PDF] Genetic Testing for Familial Alzheimer');">PDF |
Genetic Testing for Familial Alzheimers Disease AHS - Blue Cross NC
1 jan 2019 · https://www accessdata fda gov/cdrh_docs/reviews/DEN160026 pdf Fulgent ( 2019) Parkinson-Alzheimer-Dementia NGS Panel Retrieved |
|
Genomics subgroup report - European Medicines Agency - EUROPA
(pgs) genetic health risk test for hereditary thrombophilia 23andME DEN160026 Cystic Fibrosis Illumina MiSeqDx Cystic Fibrosis Clinical Sequencing Assay |
|
Advance Directive - Loyola University Chicago
https://www accessdata fda gov/cdrh_docs/reviews/den160026 pdf (last updated May 02, 2017) 24 Id 25 Timothy Caulfield, The Limits of Personalized |
|
© Copyright 2018 Sarah C Nelson - Scholarly Publishing Services
https://www accessdata fda gov/cdrh_docs/ pdf 16/den160026 pdf FDA (2018a) FDA takes new action to advance the development of reliable and beneficial |
|
The Familial Privacy Implications of Direct to - SSRN Papers
https://www accessdata fda gov/cdrh_docs/ pdf 16/den160026 pdf ) This preprint research paper has not been peer reviewed Electronic copy available at: |