densities of common liquids
How do you calculate the density of a liquid?
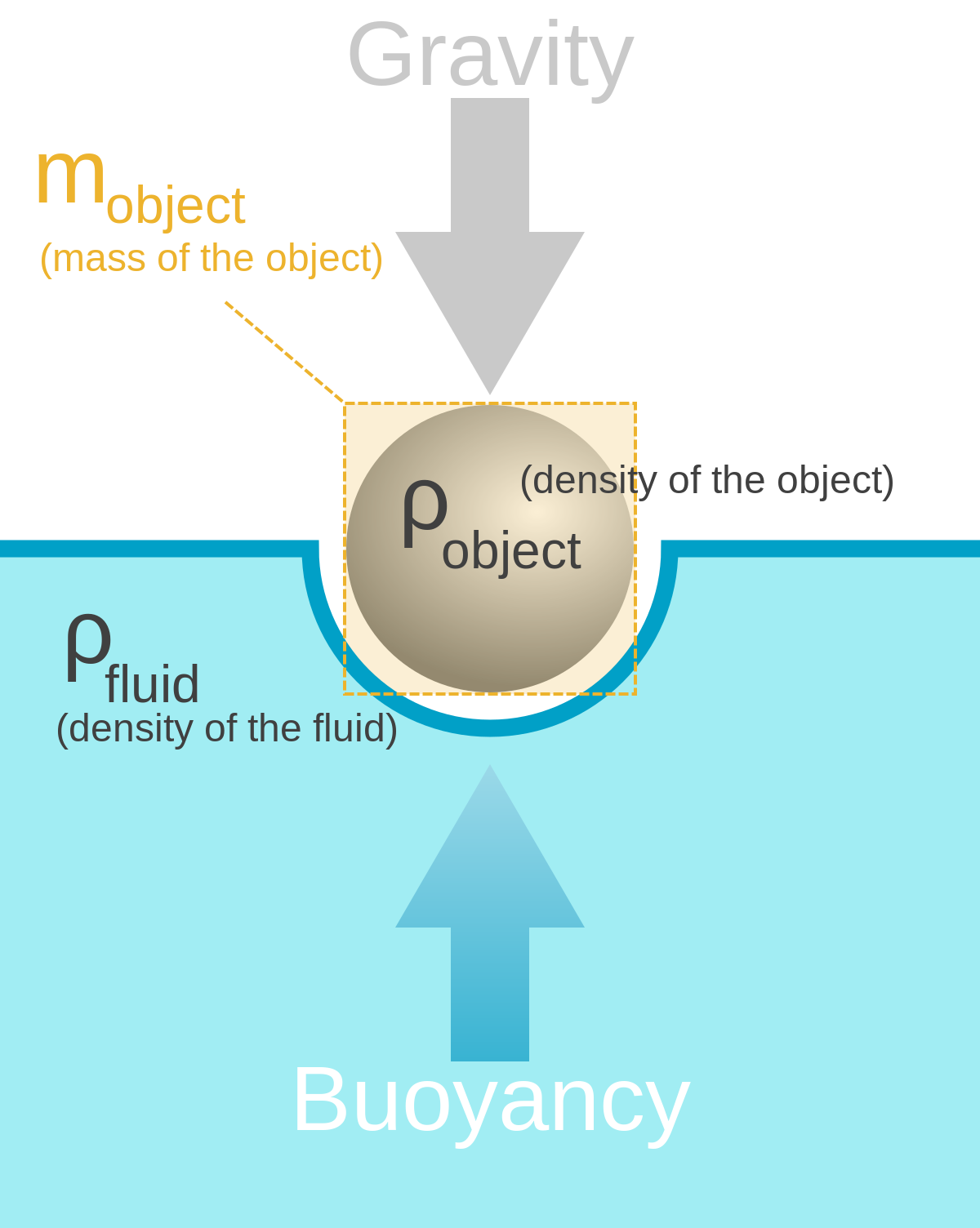
The density of a liquid is typically expressed in units of kilograms per cubic meter (kg/m3) or grams per milliliter (g/mL). The density of a liquid is determined by the mass of the atoms or molecules that make up the substance and the spacing between them.
Why do liquids have different densities?
The liquids must have different densities because the ice cube floats in one but sinks in the other. Explain that since ice floats in water, liquid water must be more dense than ice. Since ice sinks in isopropyl alcohol, alcohol must be less dense than ice.
What is the density of pure water?
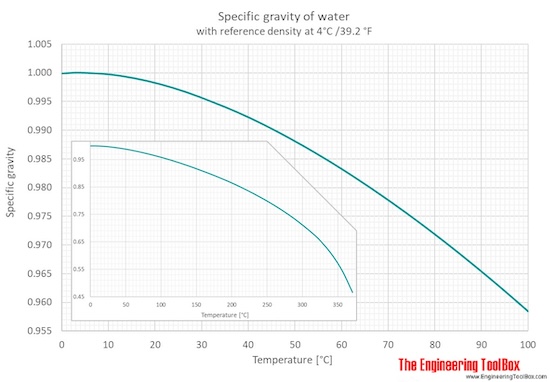
Note that the density of pure water is defined to be 1 gram per cubic centimeter (or, g/ml). Unlike most substances, water is denser as a liquid than as a solid. A consequence is that ice floats on water. Also, pure water is less dense than seawater, so fresh water can float on top of salt water, mixing at the interface.
What is the difference between density and volume?
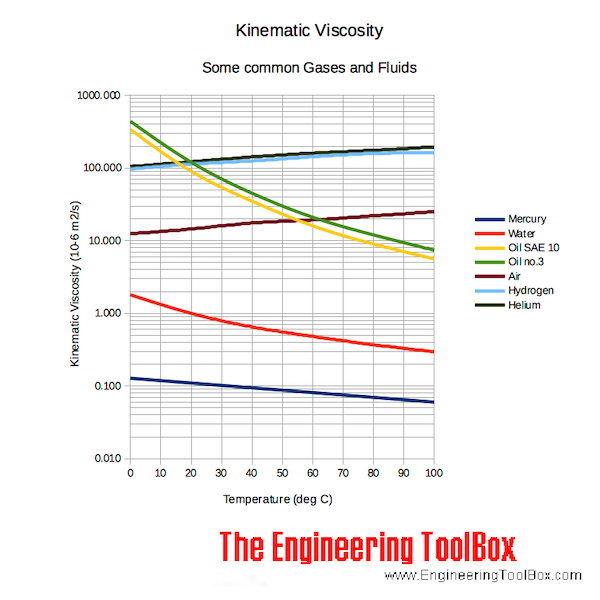
where ρ is the density, m is the mass, and V is the volume. Since gases are free to expand and contract, the densities of the gases vary considerably with temperature, whereas the densities of liquids vary little with temperature. Therefore, the densities of liquids are often treated as constant, with the density equal to the average density.
|
Densities of Some Common Liquids
Liquid Density Column: Sink or Float? Rationale/Lesson Abstract: This is a lesson on floating and sinking of liquids with a demonstration using various |
|
DENSITIES OF COMMON SUBSTANCES Material Density (gm/cm
Material. Density (gm/cm^3). Liquids. Water at 4 C. 1.0000. Water at 20 C. 0.998. Gasoline. 0.70. Diesel Fuel Oil 20-60 C. 0.82-0.95. Kerosene. |
|
7.9 Making things Float and Making a Hydrometer
Use oil and water to demonstrate different densities of liquids. Make a hydrometer using straw and clay and test the density of three different solutions namely |
|
Approximate Densities of Some Common Liquids and Solids
Density and Flotation. In this activity you further observed that materials with a greater density than a given liquid will sink and materials with less. |
|
Density of Liquids and Solids .pdf
To observe the relative densities of some common liquids and solids. • To determine the densities of water an unknown liquid |
|
Ground Water Issue: Light Nonaqueous Phase Liquids
density and inversely proportional to the viscosity of the. LNAPL. The potential mobility of various fluids may be compared using the ratio of density to |
|
General Chemistry I The Densities of Solids and Liquids 1 The
Aug 4 2018 of common convention |
|
Fluid & Gas Properties
Table 1 lists the densities of various liquids. For steam the density is typically read from the steam tables for the de- sired pressure and temperature. |
|
Viscosities and Densities of Some Multi-Component Regular Liquid
Liquid Solutions at Different Temperature Levels The densities and kinematic viscosities of the quinary regular system: benzene (1). |
|
Unit 2(fluids)grd 8 science edits July2010.indd
including viscosity and density and explain them using the particle theory. different liquids and observed objects that sink and float. In. |
|
Densities of Some Common Liquids - Wccusdnet
2 oct 2013 · Liquid Density Column: Sink or Float? Rationale/Lesson Abstract: This is a lesson on floating and sinking of liquids, with a demonstration using |
|
Approximate Densities of Some Common Liquids and Solids, 355
Density and Flotation In this activity you further observed that materials with a greater density than a given liquid will sink, and materials with less density than a |
|
Density of Liquids and Solids pdf
To observe the relative densities of some common liquids and solids • To determine the densities of water, an unknown liquid, a rubber stopper, and |
|
General Chemistry I The Densities of Solids and Liquids 1 The
4 août 2018 · Because of common convention, the mass determined will be called weight The volume of a liquid material may be accurately obtained by the |
|
13 Measurement page 1 136 DENSITY OF LIQUIDS Concepts to
Part 1: Density gradient : The following household liquids vary in density from about 0 9 g/mL to about 1 4 g/mL: vegetable oil, glycerol, water, corn syrup, detergent, and isopropyl alcohol In this activity you will be attempting to rank them in order of increasing density |
|
Liquid densities of eleven hydrocarbons found in - GovInfo
The H«{J*A«Ae measurements on propane are in good accord with these tables 39 Comparison of Eeeults of Various Observers with the Tables, Dana and co- |
|
Science Experiment: Seven Layer Density Column - Purdue Extension
What if you could stack seven different liquids in seven different layers? densities of the liquids used in the column as well as other common liquids ( measured |
|
TRICKS WITH LIQUID DENSITY - QUT
densities Density is defined as mass per unit volume The table below has the density values for some common materials The liquids with a higher density |
|
Layering Liquids - Terrific Science
Can you tell what the density of each object is? Try other objects if you wish Challenge What are the relative densities of various common liquids and solids? |
|
Exploring Density
Common Core State Standard for Mathematical Practice 4 Model with Construct a density column to compare the densities of various liquids and solids |








/hand-pouring-cooking-oil-over-water-in-beaker-98358398-587134265f9b584db3e801a6-5b84778ec9e77c0025b92807.jpg)











/Density-58a280135f9b58819c3188f7.jpg)




:max_bytes(150000):strip_icc()/GettyImages-1195928886-0f2d6e61f76f44efa517d74ffa910226.jpg)